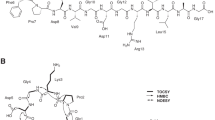
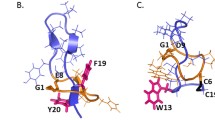
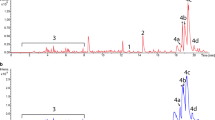
Abstract
Lasso peptides produced by bacteria have a very unique cyclic structure (“lasso” structure) and are resistant to protease. To date, a number of lasso peptides have been isolated from proteobacteria and actinobacteria. Many lasso peptides exhibit various biological activities, such as antibacterial activity, and are expected to have various applications. Based on study of genome mining, large numbers of biosynthetic gene cluster of lasso peptides are revealed to distribute over genomes of proteobacteria and actinobacteria. However, the biosynthetic gene clusters are cryptic in most cases. Therefore, the combination of genome mining and heterologous production is efficient method for the production of lasso peptides. To utilize lasso peptide as fine chemical, there have been several attempts to add new function to lasso peptide by genetic engineering. Currently, a more efficient lasso peptide production system is being developed to harness cryptic biosynthetic gene clusters of lasso peptide. In this review, the overview of lasso peptide study is discussed.


Similar content being viewed by others
References
Adelman K, Yuzenkova J, La Porta A, Zenkin N, Lee J, Lis JT, Borukhov S, Wang MD, Severinov K (2004) Molecular mechanism of transcription inhibition by peptide antibiotic microcin J25. Mol Cell 14:753–762. https://doi.org/10.1016/j.molcel.2004.05.017
Agrawal P, Khater S, Gupta M, Sain N, Mohanty D (2017) RiPPMiner: a bioinformatics resource for deciphering chemical structures of RiPPs based on prediction of cleavage and cross-links. Nucleic Acids Res 45:W80–W88. https://doi.org/10.1093/nar/gkx408
Allen CD, Chen MY, Trick AY, Le DT, Ferguson AL, Link AJ (2016) Thermal unthreading of the lasso peptides astexin-2 and astexin-3. ACS Chem Biol 11:3043–3051. https://doi.org/10.1021/acschembio.6b00588
Allen CD, Link AJ (2016) Self-assembly of catenanes from lasso peptides. J Am Chem Soc 138:14214–14217. https://doi.org/10.1021/jacs.6b09454
Bayro MJ, Mukhopadhyay J, Swapna GV, Huang JY, Ma LC, Sineva E, Dawson PE, Montelione GT, Ebright RH (2003) Structure of antibacterial peptide microcin J25: a 21-residue lariat protoknot. J Am Chem Soc 125:12382–12383. https://doi.org/10.1021/ja036677e
Blin K, Shaw S, Steinke K, Villebro R, Ziemert N, Lee SY, Medema MH, Weber T (2019) antiSMASH 5.0: updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res 47:W81–W87. https://doi.org/10.1093/nar/gkz310
Braffman NR, Piscotta FJ, Hauver J, Campbell EA, Link AJ, Darst SA (2019) Structural mechanism of transcription inhibition by lasso peptides microcin J25 and capistruin. Proc Natl Acad Sci USA 116:1273–1278. https://doi.org/10.1073/pnas.1817352116
Bratovanov EV, Ishida K, Heinze B, Pidot SJ, Stinear TP, Hegemann JD, Marahiel MA, Hertweck C (2020) Genome mining and heterologous expression reveal two distinct families of lasso peptides highly conserved in endofungal bacteria. ACS Chem Biol 15:1169–1176. https://doi.org/10.1021/acschembio.9b00805
Chekan JR, Koos JD, Zong C, Maksimov MO, Link AJ, Nair SK (2016) Structure of the lasso peptide isopeptidase identifies a topology for processing threaded substrates. J Am Chem Soc 138:16452–16458. https://doi.org/10.1021/jacs.6b10389
Cheung-Lee WL, Cao L, Link AJ (2019) Pandonodin: a proteobacterial lasso peptide with an exceptionally long C-terminal tail. ACS Chem Biol 14:2783–2792. https://doi.org/10.1021/acschembio.9b00676
Cheung-Lee WL, Link AJ (2019) Genome mining for lasso peptides: past, present, and future. J Ind Microbiol Biotechnol 46:1371–1379. https://doi.org/10.1007/s10295-019-02197-z
Cheung-Lee WL, Parry ME, Jaramillo Cartagena A, Darst SA, Link AJ (2019) Discovery and structure of the antimicrobial lasso peptide citrocin. J Biol Chem 294:6822–6830. https://doi.org/10.1074/jbc.RA118.006494
Cheung-Lee WL, Parry ME, Zong C, Cartagena AJ, Darst SA, Connell ND, Russo R, Link AJ (2020) Discovery of ubonodin, an antimicrobial lasso peptide active against members of the Burkholderia cepacia complex. ChemBioChem 21:1335–1340. https://doi.org/10.1002/cbic.201900707
Cheung WL, Chen MY, Maksimov MO, Link AJ (2016) Lasso peptide biosynthetic protein LarB1 binds both leader and core peptide regions of the precursor protein LarA. ACS Cent Sci 2:702–709. https://doi.org/10.1021/acscentsci.6b00184
Cheung WL, Pan SJ, Link AJ (2010) Much of the microcin J25 leader peptide is dispensable. J Am Chem Soc 132:2514–2515. https://doi.org/10.1021/ja910191u
Chokekijchai S, Kojima E, Anderson S, Nomizu M, Tanaka M, Machida M, Date T, Toyota K, Ishida S, Watanabe K, Yoshioka H, Roller PP, Murakami K, Mitsuya H (1995) NP-06: a novel anti-human immunodeficiency virus polypeptide produced by a Streptomyces species. Antimicrob Agents Chemother 39:2345–2347. https://doi.org/10.1128/aac.39.10.2345
Constantine KL, Friedrichs MS, Detlefsen D, Nishio M, Tsunakawa M, Furumai T, Ohkuma H, Oki T, Hill S, Bruccoleri RE, Lin PF, Mueller L (1995) High-resolution solution structure of siamycin II: novel amphipathic character of a 21-residue peptide that inhibits HIV fusion. J Biomol NMR 5:271–286. https://doi.org/10.1007/BF00211754
Cortes-Albayay C, Jarmusch SA, Trusch F, Ebel R, Andrews BA, Jaspars M, Asenjo JA (2020) Downsizing class II lasso peptides: genome mining-guided isolation of huascopeptin containing the first Gly1-Asp7 macrocycle. J Org Chem 85:1661–1667. https://doi.org/10.1021/acs.joc.9b02231
Delgado MA, Rintoul MR, Farias RN, Salomon RA (2001) Escherichia coli RNA polymerase is the target of the cyclopeptide antibiotic microcin J25. J Bacteriol 183:4543–4550. https://doi.org/10.1128/JB.183.15.4543-4550.2001
Detlefsen DJ, Hill SE, Volk KJ, Klohr SE, Tsunakawa M, Furumai T, Lin PF, Nishio M, Kawano K, Oki T, Lee MS (1995) Siamycins I and II, new anti-HIV-1 peptides: II. Sequence analysis and structure determination of siamycin I. J Antibiot (Tokyo) 48:1515–1517. https://doi.org/10.7164/antibiotics.48.1515
DiCaprio AJ, Firouzbakht A, Hudson GA, Mitchell DA (2019) Enzymatic reconstitution and biosynthetic investigation of the lasso peptide fusilassin. J Am Chem Soc 141:290–297. https://doi.org/10.1021/jacs.8b09928
Ducasse R, Yan KP, Goulard C, Blond A, Li Y, Lescop E, Guittet E, Rebuffat S, Zirah S (2012) Sequence determinants governing the topology and biological activity of a lasso peptide, microcin J25. ChemBioChem 13:371–380. https://doi.org/10.1002/cbic.201100702
Elashal HE, Cohen RD, Elashal HE, Zong C, Link AJ, Raj M (2018) Cyclic and lasso peptides: sequence determination, topology analysis, and rotaxane formation. Angew Chem Int Ed Engl 57:6150–6154. https://doi.org/10.1002/anie.201801299
Elsayed SS, Trusch F, Deng H, Raab A, Prokes I, Busarakam K, Asenjo JA, Andrews BA, van West P, Bull AT, Goodfellow M, Yi Y, Ebel R, Jaspars M, Rateb ME (2015) Chaxapeptin, a lasso peptide from extremotolerant Streptomyces leeuwenhoekii strain C58 from the hyperarid atacama desert. J Org Chem 80:10252–10260. https://doi.org/10.1021/acs.joc.5b01878
Esumi Y, Suzuki Y, Itoh Y, Uramoto M, Kimura K, Goto M, Yoshihama M, Ichikawa T (2002) Propeptin, a new inhibitor of prolyl endopeptidase produced by Microbispora. II. Determination of chemical structure. J Antibiot (Tokyo) 55:296–300. https://doi.org/10.7164/antibiotics.55.296
Fage CD, Hegemann JD, Nebel AJ, Steinbach RM, Zhu S, Linne U, Harms K, Bange G, Marahiel MA (2016) Structure and mechanism of the sphingopyxin I lasso peptide isopeptidase. Angew Chem Int Ed Engl 55:12717–12721. https://doi.org/10.1002/anie.201605232
Feng Z, Ogasawara Y, Nomura S, Dairi T (2018) Biosynthetic gene cluster of a D-tryptophan-containing lasso peptide, MS-271. ChemBioChem 19:2045–2048. https://doi.org/10.1002/cbic.201800315
Ferguson AL, Zhang S, Dikiy I, Panagiotopoulos AZ, Debenedetti PG, Link AJ (2010) An experimental and computational investigation of spontaneous lasso formation in microcin J25. Biophys J 99:3056–3065. https://doi.org/10.1016/j.bpj.2010.08.073
Frechet D, Guitton JD, Herman F, Faucher D, Helynck G, Monegier du Sorbier B, Ridoux JP, James-Surcouf E, Vuilhorgne M (1994) Solution structure of RP 71955, a new 21 amino acid tricyclic peptide active against HIV-1 virus. Biochemistry 33:42–50. https://doi.org/10.1021/bi00167a006
Gavrish E, Sit CS, Cao S, Kandror O, Spoering A, Peoples A, Ling L, Fetterman A, Hughes D, Bissell A, Torrey H, Akopian T, Mueller A, Epstein S, Goldberg A, Clardy J, Lewis K (2014) Lassomycin, a ribosomally synthesized cyclic peptide, kills Mycobacterium tuberculosis by targeting the ATP-dependent protease ClpC1P1P2. Chem Biol 21:509–518. https://doi.org/10.1016/j.chembiol.2014.01.014
Gomez-Escribano JP, Castro JF, Razmilic V, Jarmusch SA, Saalbach G, Ebel R, Jaspars M, Andrews B, Asenjo JA, Bibb MJ (2019) Heterologous expression of a cryptic gene cluster from Streptomyces leeuwenhoekii C34(T) yields a novel lasso peptide, leepeptin. Appl Environ Microbiol 85:e01752–e11719. https://doi.org/10.1128/AEM.01752-19
Hegemann JD, De Simone M, Zimmermann M, Knappe TA, Xie X, Di Leva FS, Marinelli L, Novellino E, Zahler S, Kessler H, Marahiel MA (2014) Rational improvement of the affinity and selectivity of integrin binding of grafted lasso peptides. J Med Chem 57:5829–5834. https://doi.org/10.1021/jm5004478
Hegemann JD, Fage CD, Zhu S, Harms K, Di Leva FS, Novellino E, Marinelli L, Marahiel MA (2016) The ring residue proline 8 is crucial for the thermal stability of the lasso peptide caulosegnin II. Mol Biosyst 12:1106–1109. https://doi.org/10.1039/c6mb00081a
Hegemann JD, Schwalen CJ, Mitchell DA, van der Donk WA (2018) Elucidation of the roles of conserved residues in the biosynthesis of the lasso peptide paeninodin. Chem Commun (Camb) 54:9007–9010. https://doi.org/10.1039/c8cc04411b
Hegemann JD, Zimmermann M, Xie X, Marahiel MA (2013) Caulosegnins I-III: a highly diverse group of lasso peptides derived from a single biosynthetic gene cluster. J Am Chem Soc 135:210–222. https://doi.org/10.1021/ja308173b
Hegemann JD, Zimmermann M, Xie X, Marahiel MA (2015) Lasso peptides: an intriguing class of bacterial natural products. Acc Chem Res 48:1909–1919. https://doi.org/10.1021/acs.accounts.5b00156
Hegemann JD, Zimmermann M, Zhu S, Klug D, Marahiel MA (2013) Lasso peptides from proteobacteria: genome mining employing heterologous expression and mass spectrometry. Biopolymers 100:527–542. https://doi.org/10.1002/bip.22326
Hegemann JD, Zimmermann M, Zhu S, Steuber H, Harms K, Xie X, Marahiel MA (2014) Xanthomonins I–III: a new class of lasso peptides with a seven-residue macrolactam ring. Angew Chem Int Ed Engl 53:2230–2234. https://doi.org/10.1002/anie.201309267
Helynck G, Dubertret C, Mayaux JF, Leboul J (1993) Isolation of RP 71955, a new anti-HIV-1 peptide secondary metabolite. J Antibiot (Tokyo) 46:1756–1757. https://doi.org/10.7164/antibiotics.46.1756
Hudson GA, Mitchell DA (2018) RiPP antibiotics: biosynthesis and engineering potential. Curr Opin Microbiol 45:61–69. https://doi.org/10.1016/j.mib.2018.02.010
Inokoshi J, Koyama N, Miyake M, Shimizu Y, Tomoda H (2016) Structure-activity analysis of gram-positive bacterium-producing lasso peptides with anti-mycobacterial activity. Sci Rep 6:30375. https://doi.org/10.1038/srep30375
Inokoshi J, Matsuhama M, Miyake M, Ikeda H, Tomoda H (2012) Molecular cloning of the gene cluster for lariatin biosynthesis of Rhodococcus jostii K01-B0171. Appl Microbiol Biotechnol 95:451–460. https://doi.org/10.1007/s00253-012-3973-8
Iwatsuki M, Koizumi Y, Gouda H, Hirono S, Tomoda H, Omura S (2009) Lys17 in the 'lasso' peptide lariatin A is responsible for anti-mycobacterial activity. Bioorg Med Chem Lett 19:2888–2890. https://doi.org/10.1016/j.bmcl.2009.03.033
Iwatsuki M, Tomoda H, Uchida R, Gouda H, Hirono S, Omura S (2006) Lariatins, antimycobacterial peptides produced by Rhodococcus sp. K01-B0171, have a lasso structure. J Am Chem Soc 128:7486–7491. https://doi.org/10.1021/ja056780z
Iwatsuki M, Uchida R, Takakusagi Y, Matsumoto A, Jiang CL, Takahashi Y, Arai M, Kobayashi S, Matsumoto M, Inokoshi J, Tomoda H, Omura S (2007) Lariatins, novel anti-mycobacterial peptides with a lasso structure, produced by Rhodococcus jostii K01-B0171. J Antibiot (Tokyo) 60:357–363. https://doi.org/10.1038/ja.2007.48
Jeanne Dit Fouque K, Bisram V, Hegemann JD, Zirah S, Rebuffat S, Fernandez-Lima F (2019) Structural signatures of the class III lasso peptide BI-32169 and the branched-cyclic topoisomers using trapped ion mobility spectrometry-mass spectrometry and tandem mass spectrometry. Anal Bioanal Chem 411:6287–6296. https://doi.org/10.1007/s00216-019-01613-8
Kaczmarczyk A, Vorholt JA, Francez-Charlot A (2013) Cumate-inducible gene expression system for sphingomonads and other alphaproteobacteria. Appl Environ Microbiol 79:6795–6802. https://doi.org/10.1128/AEM.02296-13
Katahira R, Yamasaki M, Matsuda Y, Yoshida M (1996) MS-271, a novel inhibitor of calmodulin-activated myosin light chain kinase from Streptomyces sp.-II. Solution structure of MS-271: characteristic features of the "lasso' structure. Bioorg Med Chem 4:121–129. https://doi.org/10.1016/0968-0896(95)00176-x
Kaweewan I, Hemmi H, Komaki H, Harada S, Kodani S (2018) Isolation and structure determination of a new lasso peptide specialicin based on genome mining. Bioorg Med Chem 26:6050–6055. https://doi.org/10.1016/j.bmc.2018.11.007
Kaweewan I, Ohnishi-Kameyama M, Kodani S (2017) Isolation of a new antibacterial peptide achromosin from Streptomyces achromogenes subsp. achromogenes based on genome mining. J Antibiot (Tokyo) 70:208–211. https://doi.org/10.1038/ja.2016.108
Kersten RD, Yang YL, Xu Y, Cimermancic P, Nam SJ, Fenical W, Fischbach MA, Moore BS, Dorrestein PC (2011) A mass spectrometry-guided genome mining approach for natural product peptidogenomics. Nat Chem Biol 7:794–802. https://doi.org/10.1038/nchembio.684
Kimura K, Kanou F, Takahashi H, Esumi Y, Uramoto M, Yoshihama M (1997) Propeptin, a new inhibitor of prolyl endopeptidase produced by Microbispora. I. Fermentation, isolation and biological properties. J Antibiot (Tokyo) 50:373–378. https://doi.org/10.7164/antibiotics.50.373
Kimura K, Yamazaki M, Sasaki N, Yamashita T, Negishi S, Nakamura T, Koshino H (2007) Novel propeptin analog, propeptin-2, missing two amino acid residues from the propeptin C-terminus loses antibiotic potency. J Antibiot (Tokyo) 60:519–523. https://doi.org/10.1038/ja.2007.66
Knappe TA, Linne U, Robbel L, Marahiel MA (2009) Insights into the biosynthesis and stability of the lasso peptide capistruin. Chem Biol 16:1290–1298. https://doi.org/10.1016/j.chembiol.2009.11.009
Knappe TA, Linne U, Xie X, Marahiel MA (2010) The glucagon receptor antagonist BI-32169 constitutes a new class of lasso peptides. FEBS Lett 584:785–789. https://doi.org/10.1016/j.febslet.2009.12.046
Knappe TA, Linne U, Zirah S, Rebuffat S, Xie X, Marahiel MA (2008) Isolation and structural characterization of capistruin, a lasso peptide predicted from the genome sequence of Burkholderia thailandensis E264. J Am Chem Soc 130:11446–11454. https://doi.org/10.1021/ja802966g
Knappe TA, Manzenrieder F, Mas-Moruno C, Linne U, Sasse F, Kessler H, Xie X, Marahiel MA (2011) Introducing lasso peptides as molecular scaffolds for drug design: engineering of an integrin antagonist. Angew Chem Int Ed Engl 50:8714–8717. https://doi.org/10.1002/anie.201102190
Kodani S, Hemmi H, Miyake Y, Kaweewan I, Nakagawa H (2018) Heterologous production of a new lasso peptide brevunsin in Sphingomonas subterranea. J Ind Microbiol Biotechnol 45:983–992. https://doi.org/10.1007/s10295-018-2077-6
Kodani S, Inoue Y, Suzuki M, Dohra H, Suzuki T, Hemmi H, Ohnishi-Kameyama M (2017) Sphaericin, a lasso peptide from the rare actinomycete Planomonospora sphaerica. Eur J Org Chem 2017:1177–1183. https://doi.org/10.1002/ejoc.201601334
Koos JD, Link AJ (2019) Heterologous and in vitro reconstitution of fuscanodin, a lasso peptide from Thermobifida fusca. J Am Chem Soc 141:928–935. https://doi.org/10.1021/jacs.8b10724
Kunakom S, Eustaquio AS (2020) Heterologous production of lasso peptide capistruin in a Burkholderia host. ACS Synth Biol 9:241–248. https://doi.org/10.1021/acssynbio.9b00438
Kuroha M, Hemmi H, Ohnishi-Kameyama M, Kodani S (2017) Isolation and structure determination of a new lasso peptide subterisin from Sphingomonas subterranea. Tetrahedron Lett 58:3429–3432. https://doi.org/10.1016/j.tetlet.2017.07.064
Kuznedelov K, Semenova E, Knappe TA, Mukhamedyarov D, Srivastava A, Chatterjee S, Ebright RH, Marahiel MA, Severinov K (2011) The antibacterial threaded-lasso peptide capistruin inhibits bacterial RNA polymerase. J Mol Biol 412:842–848. https://doi.org/10.1016/j.jmb.2011.02.060
Li Y, Ducasse R, Zirah S, Blond A, Goulard C, Lescop E, Giraud C, Hartke A, Guittet E, Pernodet JL, Rebuffat S (2015) Characterization of sviceucin from Streptomyces provides insight into enzyme exchangeability and disulfide bond formation in lasso peptides. ACS Chem Biol 10:2641–2649. https://doi.org/10.1021/acschembio.5b00584
Lin PF, Samanta H, Bechtold CM, Deminie CA, Patick AK, Alam M, Riccardi K, Rose RE, White RJ, Colonno RJ (1996) Characterization of siamycin I, a human immunodeficiency virus fusion inhibitor. Antimicrob Agents Chemother 40:133–138. https://doi.org/10.1128/AAC.40.1.133
Maksimov MO, Koos JD, Zong C, Lisko B, Link AJ (2015) Elucidating the specificity determinants of the AtxE2 lasso peptide isopeptidase. J Biol Chem 290:30806–30812. https://doi.org/10.1074/jbc.M115.694083
Maksimov MO, Link AJ (2013) Discovery and characterization of an isopeptidase that linearizes lasso peptides. J Am Chem Soc 135:12038–12047. https://doi.org/10.1021/ja4054256
Maksimov MO, Link AJ (2014) Prospecting genomes for lasso peptides. J Ind Microbiol Biotechnol 41:333–344. https://doi.org/10.1007/s10295-013-1357-4
Maksimov MO, Pan SJ, Link AJ (2012) Lasso peptides: structure, function, biosynthesis, and engineering. Nat Prod Rep 29:996–1006. https://doi.org/10.1039/c2np20070h
Maksimov MO, Pelczer I, Link AJ (2012) Precursor-centric genome-mining approach for lasso peptide discovery. Proc Natl Acad Sci USA 109:15223–15228. https://doi.org/10.1073/pnas.1208978109
Martin-Gomez H, Linne U, Albericio F, Tulla-Puche J, Hegemann JD (2018) Investigation of the biosynthesis of the lasso peptide chaxapeptin using an E. coli-based production system. J Nat Prod 81:2050–2056. https://doi.org/10.1021/acs.jnatprod.8b00392
Mathavan I, Zirah S, Mehmood S, Choudhury HG, Goulard C, Li Y, Robinson CV, Rebuffat S, Beis K (2014) Structural basis for hijacking siderophore receptors by antimicrobial lasso peptides. Nat Chem Biol 10:340–342. https://doi.org/10.1038/nchembio.1499
Metelev M, Arseniev A, Bushin LB, Kuznedelov K, Artamonova TO, Kondratenko R, Khodorkovskii M, Seyedsayamdost MR, Severinov K (2017) Acinetodin and klebsidin, RNA polymerase targeting lasso peptides produced by human isolates of Acinetobacter gyllenbergii and Klebsiella pneumoniae. ACS Chem Biol 12:814–824. https://doi.org/10.1021/acschembio.6b01154
Metelev M, Tietz JI, Melby JO, Blair PM, Zhu L, Livnat I, Severinov K, Mitchell DA (2015) Structure, bioactivity, and resistance mechanism of streptomonomicin, an unusual lasso peptide from an understudied halophilic actinomycete. Chem Biol 22:241–250. https://doi.org/10.1016/j.chembiol.2014.11.017
Mevaere J, Goulard C, Schneider O, Sekurova ON, Ma H, Zirah S, Afonso C, Rebuffat S, Zotchev SB, Li Y (2018) An orthogonal system for heterologous expression of actinobacterial lasso peptides in Streptomyces hosts. Sci Rep 8:8232. https://doi.org/10.1038/s41598-018-26620-0
Morishita Y, Chiba S, Tsukuda E, Tanaka T, Ogawa T, Yamasaki M, Yoshida M, Kawamoto I, Matsuda Y (1994) RES-701-1, a novel and selective endothelin type B receptor antagonist produced by Streptomyces sp. RE-701. I. Characterization of producing strain, fermentation, isolation, physico-chemical and biological properties. J Antibiot (Tokyo) 47:269–275. https://doi.org/10.7164/antibiotics.47.269
Ochou M, Saito M, Kurusu Y (2008) Characterization of two compatible small plasmids from Sphingobium yanoikuyae. Biosci Biotechnol Biochem 72:1130–1133. https://doi.org/10.1271/bbb.70813
Ogawa T, Ochiai K, Tanaka T, Tsukuda E, Chiba S, Yano K, Yamasaki M, Yoshida M, Matsuda Y (1995) RES-701-2, -3 and -4, novel and selective endothelin type B receptor antagonists produced by Streptomyces sp. I. Taxonomy of producing strains, fermentation, isolation, and biochemical properties. J Antibiot (Tokyo) 48:1213–1220. https://doi.org/10.7164/antibiotics.48.1213
Pan SJ, Cheung WL, Fung HK, Floudas CA, Link AJ (2011) Computational design of the lasso peptide antibiotic microcin J25. Protein Eng Des Sel 24:275–282. https://doi.org/10.1093/protein/gzq108
Pan SJ, Cheung WL, Link AJ (2010) Engineered gene clusters for the production of the antimicrobial peptide microcin J25. Protein Expr Purif 71:200–206. https://doi.org/10.1016/j.pep.2009.12.010
Pan SJ, Link AJ (2011) Sequence diversity in the lasso peptide framework: discovery of functional microcin J25 variants with multiple amino acid substitutions. J Am Chem Soc 133:5016–5023. https://doi.org/10.1021/ja1109634
Pan SJ, Rajniak J, Cheung WL, Link AJ (2012) Construction of a single polypeptide that matures and exports the lasso peptide microcin J25. ChemBioChem 13:367–370. https://doi.org/10.1002/cbic.201100596
Pan SJ, Rajniak J, Maksimov MO, Link AJ (2012) The role of a conserved threonine residue in the leader peptide of lasso peptide precursors. Chem Commun (Camb) 48:1880–1882. https://doi.org/10.1039/c2cc17211a
Pavlova O, Mukhopadhyay J, Sineva E, Ebright RH, Severinov K (2008) Systematic structure-activity analysis of microcin J25. J Biol Chem 283:25589–25595. https://doi.org/10.1074/jbc.M803995200
Piscotta FJ, Tharp JM, Liu WR, Link AJ (2015) Expanding the chemical diversity of lasso peptide MccJ25 with genetically encoded noncanonical amino acids. Chem Commun (Camb) 51:409–412. https://doi.org/10.1039/c4cc07778d
Potterat O, Stephan H, Metzger JW, Gnau V, Zähner H, Jung G (1994) Aborycin—a tricyclic 21-peptide antibiotic Isolated from Streptomyces griseoflavus. Liebigs Ann Chem 1994:741–743. https://doi.org/10.1002/jlac.199419940716
Potterat O, Wagner K, Gemmecker G, Mack J, Puder C, Vettermann R, Streicher R (2004) BI-32169, a bicyclic 19-peptide with strong glucagon receptor antagonist activity from Streptomyces sp. J Nat Prod 67:1528–1531. https://doi.org/10.1021/np040093o
Romano M, Fusco G, Choudhury HG, Mehmood S, Robinson CV, Zirah S, Hegemann JD, Lescop E, Marahiel MA, Rebuffat S, De Simone A, Beis K (2018) Structural basis for natural product selection and export by bacterial ABC transporters. ACS Chem Biol 13:1598–1609. https://doi.org/10.1021/acschembio.8b00226
Rosengren KJ, Blond A, Afonso C, Tabet JC, Rebuffat S, Craik DJ (2004) Structure of thermolysin cleaved microcin J25: extreme stability of a two-chain antimicrobial peptide devoid of covalent links. Biochemistry 43:4696–4702. https://doi.org/10.1021/bi0361261
Salomon RA, Farias RN (1992) Microcin 25, a novel antimicrobial peptide produced by Escherichia coli. J Bacteriol 174:7428–7435. https://doi.org/10.1128/jb.174.22.7428-7435.1992
Sanchez-Hidalgo M, Martin J, Genilloud O (2020) Identification and heterologous expression of the biosynthetic gene cluster encoding the lasso peptide humidimycin, a caspofungin activity potentiator. Antibiotics (Basel) 9:67. https://doi.org/10.3390/antibiotics9020067
Santos-Aberturas J, Chandra G, Frattaruolo L, Lacret R, Pham TH, Vior NM, Eyles TH, Truman AW (2019) Uncovering the unexplored diversity of thioamidated ribosomal peptides in actinobacteria using the RiPPER genome mining tool. Nucleic Acids Res 47:4624–4637. https://doi.org/10.1093/nar/gkz192
Shao M, Ma J, Li Q, Ju J (2019) Identification of the anti-infective aborycin biosynthetic gene cluster from deep-sea-derived Streptomyces sp. SCSIO ZS0098 enables production in a heterologous host. Mar Drugs 17:127. https://doi.org/10.3390/md17020127
Solbiati JO, Ciaccio M, Farias RN, Gonzalez-Pastor JE, Moreno F, Salomon RA (1999) Sequence analysis of the four plasmid genes required to produce the circular peptide antibiotic microcin J25. J Bacteriol 181:2659–2662. https://doi.org/10.1128/JB.181.8.2659-2662.1999
Son S, Jang M, Lee B, Hong YS, Ko SK, Jang JH, Ahn JS (2018) Ulleungdin, a lasso peptide with cancer cell migration inhibitory activity discovered by the genome mining approach. J Nat Prod 81:2205–2211. https://doi.org/10.1021/acs.jnatprod.8b00449
Sugai S, Ohnishi-Kameyama M, Kodani S (2017) Isolation and identification of a new lasso peptide cattlecin from Streptomyces cattleya based on genome mining. Appl Biol Chem 60:163–167. https://doi.org/10.1007/s13765-017-0268-x
Takasaka N, Kaweewan I, Ohnishi-Kameyama M, Kodani S (2017) Isolation of a new antibacterial peptide actinokineosin from Actinokineospora spheciospongiae based on genome mining. Lett Appl Microbiol 64:150–157. https://doi.org/10.1111/lam.12693
Tietz JI, Schwalen CJ, Patel PS, Maxson T, Blair PM, Tai HC, Zakai UI, Mitchell DA (2017) A new genome-mining tool redefines the lasso peptide biosynthetic landscape. Nat Chem Biol 13:470–478. https://doi.org/10.1038/nchembio.2319
Tsunakawa M, Hu SL, Hoshino Y, Detlefson DJ, Hill SE, Furumai T, White RJ, Nishio M, Kawano K, Yamamoto S, Fukagawa Y, Oki T (1995) Siamycins I and II, new anti-HIV peptides: I. Fermentation, isolation, biological activity and initial characterization. J Antibiot (Tokyo) 48:433–434. https://doi.org/10.7164/antibiotics.48.433
Um S, Kim YJ, Kwon H, Wen H, Kim SH, Kwon HC, Park S, Shin J, Oh DC (2013) Sungsanpin, a lasso peptide from a deep-sea streptomycete. J Nat Prod 76:873–879. https://doi.org/10.1021/np300902g
Valiante V, Monteiro MC, Martin J, Altwasser R, El Aouad N, Gonzalez I, Kniemeyer O, Mellado E, Palomo S, de Pedro N, Perez-Victoria I, Tormo JR, Vicente F, Reyes F, Genilloud O, Brakhage AA (2015) Hitting the caspofungin salvage pathway of human-pathogenic fungi with the novel lasso peptide humidimycin (MDN-0010). Antimicrob Agents Chemother 59:5145–5153. https://doi.org/10.1128/AAC.00683-15
Vincent PA, Delgado MA, Farias RN, Salomon RA (2004) Inhibition of Salmonella enterica serovars by microcin J25. FEMS Microbiol Lett 236:103–107. https://doi.org/10.1016/j.femsle.2004.05.027
Weber W, Fischli W, Hochuli E, Kupfer E, Weibel EK (1991) Anantin—a peptide antagonist of the atrial natriuretic factor (ANF). I. Producing organism, fermentation, isolation and biological activity. J Antibiot (Tokyo) 44:164–171. https://doi.org/10.7164/antibiotics.44.164
Wilson KA, Kalkum M, Ottesen J, Yuzenkova J, Chait BT, Landick R, Muir T, Severinov K, Darst SA (2003) Structure of microcin J25, a peptide inhibitor of bacterial RNA polymerase, is a lassoed tail. J Am Chem Soc 125:12475–12483. https://doi.org/10.1021/ja036756q
Wyss DF, Lahm HW, Manneberg M, Labhardt AM (1991) Anantin—a peptide antagonist of the atrial natriuretic factor (ANF). II. Determination of the primary sequence by NMR on the basis of proton assignments. J Antibiot (Tokyo) 44:172–180. https://doi.org/10.7164/antibiotics.44.172
Yagi A, Uchida R, Hamamoto H, Sekimizu K, Kimura KI, Tomoda H (2017) Anti-mycobacterium activity of microbial peptides in a silkworm infection model with Mycobacterium smegmatis. J Antibiot (Tokyo) 70:685–690. https://doi.org/10.1038/ja.2017.23
Yamasaki M, Yano K, Yoshida M, Matsuda Y, Yamaguchi K (1994) RES-701-1, a novel and selective endothelin type B receptor antagonist produced by Streptomyces sp. RE-701. II. Determination of the primary sequence. J Antibiot (Tokyo) 47:276–280. https://doi.org/10.7164/antibiotics.47.276
Yan KP, Li Y, Zirah S, Goulard C, Knappe TA, Marahiel MA, Rebuffat S (2012) Dissecting the maturation steps of the lasso peptide microcin J25 in vitro. ChemBioChem 13:1046–1052. https://doi.org/10.1002/cbic.201200016
Yano K, Toki S, Nakanishi S, Ochiai K, Ando K, Yoshida M, Matsuda Y, Yamasaki M (1996) MS-271, a novel inhibitor of calmodulin-activated myosin light chain kinase from Streptomyces sp.-I. Isolation, structural determination and biological properties of MS-271. Bioorg Med Chem 4:115–120. https://doi.org/10.1016/0968-0896(95)00175-1
Yuzenkova J, Delgado M, Nechaev S, Savalia D, Epshtein V, Artsimovitch I, Mooney RA, Landick R, Farias RN, Salomon R, Severinov K (2002) Mutations of bacterial RNA polymerase leading to resistance to microcin j25. J Biol Chem 277:50867–50875. https://doi.org/10.1074/jbc.M209425200
Zhang C, Seyedsayamdost MR (2020) CanE, an iron/2-oxoglutarate-dependent lasso peptide hydroxylase from Streptomyces canus. ACS Chem Biol 15:890–894. https://doi.org/10.1021/acschembio.0c00109
Zhu S, Fage CD, Hegemann JD, Mielcarek A, Yan D, Linne U, Marahiel MA (2016) The B1 protein guides the biosynthesis of a lasso peptide. Sci Rep 6:35604. https://doi.org/10.1038/srep35604
Zhu S, Fage CD, Hegemann JD, Yan D, Marahiel MA (2016) Dual substrate-controlled kinase activity leads to polyphosphorylated lasso peptides. FEBS Lett 590:3323–3334. https://doi.org/10.1002/1873-3468.12386
Zhu S, Hegemann JD, Fage CD, Zimmermann M, Xie X, Linne U, Marahiel MA (2016) Insights into the unique phosphorylation of the lasso peptide paeninodin. J Biol Chem 291:13662–13678. https://doi.org/10.1074/jbc.M116.722108
Zimmermann M, Hegemann JD, Xie X, Marahiel MA (2013) The astexin-1 lasso peptides: biosynthesis, stability, and structural studies. Chem Biol 20:558–569. https://doi.org/10.1016/j.chembiol.2013.03.013
Zimmermann M, Hegemann JD, Xie X, Marahiel MA (2014) Characterization of caulonodin lasso peptides revealed unprecedented N-terminal residues and a precursor motif essential for peptide maturation. Chem Sci 5:4032–4043. https://doi.org/10.1039/c4sc01428f
Zong C, Cheung-Lee WL, Elashal HE, Raj M, Link AJ (2018) Albusnodin: an acetylated lasso peptide from Streptomyces albus. Chem Commun (Camb) 54:1339–1342. https://doi.org/10.1039/c7cc08620b
Zong C, Maksimov MO, Link AJ (2016) Construction of lasso peptide fusion proteins. ACS Chem Biol 11:61–68. https://doi.org/10.1021/acschembio.5b00745
Zong C, Wu MJ, Qin JZ, Link AJ (2017) Lasso peptide benenodin-1 is a thermally actuated [1] rotaxane switch. J Am Chem Soc 139:10403–10409. https://doi.org/10.1021/jacs.7b04830
Zyubko T, Serebryakova M, Andreeva J, Metelev M, Lippens G, Dubiley S, Severinov K (2019) Efficient in vivo synthesis of lasso peptide pseudomycoidin proceeds in the absence of both the leader and the leader peptidase. Chem Sci 10:9699–9707. https://doi.org/10.1039/c9sc02370d
Acknowledgements
Research of lasso peptide in Kodani’s laboratory was supported by the Japan Society for the Promotion of Science by Grants-in-aids (Grant number 20K05848) and the Sumitomo Foundation (Grant number 180074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kodani, S., Unno, K. How to harness biosynthetic gene clusters of lasso peptides. J Ind Microbiol Biotechnol 47, 703–714 (2020). https://doi.org/10.1007/s10295-020-02292-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-020-02292-6