Abstract
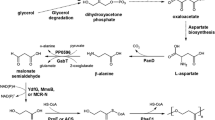
The biological production of 3-hydroxypropionic acid (3-HP) has attracted significant attention because of its industrial importance. The low titer, yield and productivity, all of which are related directly or indirectly to the toxicity of 3-HP, have limited the commercial production of 3-HP. The aim of this study was to identify and select a 3-HP tolerant Escherichia coli strain among nine strains reported to produce various organic acids efficiently at high titer. When transformed with heterologous glycerol dehydratase, reactivase and aldehyde dehydrogenase, all nine E. coli strains produced 3-HP from glycerol but the level of 3-HP production, protein expression and activities of the important enzymes differed significantly according to the strain. Two E. coli strains, W3110 and W, showed higher levels of growth than the others in the presence of 25 g/L 3-HP. In the glycerol fed-batch bioreactor experiments, the recombinant E. coli W produced a high level of 3-HP at 460 ± 10 mM (41.5 ± 1.1 g/L) in 48 h with a yield of 31 % and a productivity of 0.86 ± 0.05 g/L h. In contrast, the recombinant E. coli W3110 produced only 180 ± 8.5 mM 3-HP (15.3 ± 0.8 g/L) in 48 h with a yield and productivity of 26 % and 0.36 ± 0.02 g/L h, respectively. This shows that the tolerance to and the production of 3-HP differ significantly among the well-known, similar strains of E. coli. The titer and productivity obtained with E. coli W were the highest reported thus far for the biological production of 3-HP from glycerol by E. coli.





Similar content being viewed by others
References
Arasu MV, Kumar V, Ashok S, Song H, Rathnasingh C, Lee HJ, Seung D, Park S (2011) Isolation and characterization of the new Klebsiella pneumoniae J2B strain showing improved growth characteristics with reduced lipopolysaccharide formation. Biotechnol Bioprocess Eng 16:1134–1143
Arasu MV, Sarkar R, Sekar BS, Kumar V, Rathnasingh C, Choi J, Song H, Seung D, Park S (2013) Isolation of a novel Pseudomonas species SP2 producing vitamin B12 under aerobic condition. Biotechnol Bioprocess Eng 18(1):43–51
Ashok S, Mohan Raj S, Ko Y, Sankaranarayanan M, Zhou S, Kumar V, Park S (2013) Effect of puuC overexpression and nitrate addition on glycerol metabolism and anaerobic 3-hydroxypropionic acid production in recombinant Klebsiella pneumoniae ΔglpK ΔdhaT. Metab Eng 15:10–24. doi:10.1016/j.ymben.2012.09.004
Ashok S, Sankaranarayanan M, Ko Y, Jae KE, Ainala SK, Kumar V, Park S (2013) Production of 3-hydroxypropionic acid from glycerol by recombinant Klebsiella pneumoniae ΔdhaT ΔyqhD which can produce vitamin B12 naturally. Biotechnol Bioeng 110(2):511–524. doi:10.1002/bit.24726
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72(1):248–254
Datta R, Tsai SP, Bonsignore P, Moon SH, Frank JR (1995) Technological and economic potential of poly (lactic acid) and lactic acid derivatives. FEMS Microbiol Rev 16(2–3):221–231
De Mey M, De Maeseneire S, Soetaert W, Vandamme E (2007) Minimizing acetate formation in E. coli fermentations. J Ind Microbiol Biotechnol 34(11):689–700
Dien B, Nichols N, Bothast R (2001) Recombinant Escherichia coli engineered for production of l-lactic acid from hexose and pentose sugars. J Ind Microbiol Biotechnol 27(4):259–264
Huang Y, Li Z, Shimizu K, Ye Q (2013) Co-production of 3-hydroxypropionic acid and 1,3-propanediol by Klebseilla pneumoniae expressing aldH under microaerobic conditions. Bioresour Technol 128:505–512. doi:10.1016/j.biortech.2012.10.143
Kirk RE, Othmer DF (1951) Encyclopedia of chemical technology, vol 6. Interscience, New York
Ko Y, Ashok S, Zhou S, Kumar V, Park S (2012) Aldehyde dehydrogenase activity is important to the production of 3-hydroxypropionic acid from glycerol by recombinant K. pneumoniae. Process Biochem 47(7):1135–1143. doi:10.1016/j.procbio.2012.04.007
Kumar V, Sankaranarayanan M, Durgapal M, Zhou S, Ko Y, Ashok S, Sarkar R, Park S (2013) Simultaneous production of 3-hydroxypropionic acid and 1,3-propanediol from glycerol using resting cells of the lactate dehydrogenase-deficient recombinant K. pneumoniae overexpressing an aldehyde dehydrogenase. Bioresour Technol 135:555–563. doi:10.1016/j.biortech.2012.11.018
Kumar V, Ashok S, Park S (2013) Recent advances in biological production of 3-hydroxypropionic acid. Biotechnol Adv 31(6):945–961
Kumar V, Sankaranarayanan M, Jae KE, Durgapal M, Ashok S, Ko Y, Sarkar R, Park S (2012) Co-production of 3-hydroxypropionic acid and 1,3-propanediol from glycerol using resting cells of recombinant K. pneumoniae J2B strain overexpressing aldehyde dehydrogenase. Appl Microbiol Biotechnol 96(2):373–383. doi:10.1007/s00253-012-4187-9
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Lynch M, Gill R, Warnecke-Lipscomb T (2011) Method for producing 3-hydroxypropionic acid and other products. WO Patent 2,011,038,364
Mazumdar S, Clomburg JM, Gonzalez R (2010) Escherichia coli strains engineered for homofermentative production of d-lactic acid from glycerol. Appl Environ Microbiol 76(13):4327–4336
Miller JH (1972) Experiments in molecular genetics, vol 60. Cold Spring Harbor Laboratory Cold Spring Harbor, New York
Raj SM, Rathnasingh C, Jo J-E, Park S (2008) Production of 3-hydroxypropionic acid from glycerol by a novel recombinant E. coli BL21 strain. Process Biochem 43(12):1440–1446. doi:10.1016/j.procbio.2008.04.027
Raj SM, Rathnasingh C, Jung W-C, Selvakumar E, Park S (2010) A novel NAD+ -dependent aldehyde dehydrogenase encoded by the puuC gene of K. pneumoniae DSM 2026 that utilizes 3-hydroxypropionaldehyde as a substrate. Biotechnol Bioprocess Eng 15(1):131–138. doi:10.1007/s12257-010-0030-2
Rathnasingh C, Raj SM, Jo JE, Park S (2009) Development and evaluation of efficient recombinant E. coli strains for the production of 3-hydroxypropionic acid from glycerol. Biotechnol Bioeng 104(4):729–739. doi:10.1002/bit.22429
Rathnasingh C, Raj SM, Lee Y, Catherine C, Ashok S, Park S (2012) Production of 3-hydroxypropionic acid via malonyl-CoA pathway using recombinant E. coli strains. J Biotechnol 157(4):633–640. doi:10.1016/j.jbiotec.2011.06.008
Sambrook J, Russell DW (2001) Molecular cloning: a laboratory manual (3-volume set)
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Slater SC, Voige W, Dennis D (1988) Cloning and expression in E. coli of the Alcaligenes eutrophus H16 poly-β-hydroxybutyrate biosynthetic pathway. J Bacteriol 170(10):4431–4436
Stincone A, Daudi N, Rahman AS, Antczak P, Henderson I, Cole J, Johnson MD, Lund P, Falciani F (2011) A systems biology approach sheds new light on E. coli acid resistance. Nucl Acids Res 39(17):7512–7528
Vemuri G, Altman E, Sangurdekar D, Khodursky A, Eiteman M (2006) Overflow metabolism in E. coli during steady-state growth: transcriptional regulation and effect of the redox ratio. Appl Environ Microbiol 72(5):3653–3661
Wang Y, Tian T, Zhao J, Wang J, Yan T, Xu L, Liu Z, Garza E, Iverson A, Manow R (2012) Homofermentative production of d-lactic acid from sucrose by a metabolically engineered E. coli. Biotechnol Lett 34(11):2069–2075
Warnecke-Lipscomb T, Lynch M, Gill R (2010) Methods, systems and compositions for increased microorganism tolerance to and production of 3-hydroxypropionic acid (3-HP). WO Patent 2,010,011,874
Warnecke T, Gill RT (2005) Organic acid toxicity, tolerance, and production in E. coli biorefining applications. Microb Cell Fact 4:25. doi:10.1186/1475-2859-4-25
Warnecke TE, Lynch MD, Karimpour-Fard A, Lipscomb ML, Handke P, Mills T, Ramey CJ, Hoang T, Gill RT (2010) Rapid dissection of a complex phenotype through genomic-scale mapping of fitness altering genes. Metab Eng 12(3):241–250. doi:10.1016/j.ymben.2009.12.002
Warnecke TE, Lynch MD, Karimpour-Fard A, Sandoval N, Gill RT (2008) A genomics approach to improve the analysis and design of strain selections. Metab Eng 10(3–4):154–165. doi:10.1016/j.ymben.2008.04.004
Warnecke TE, Lynch MD, Lipscomb ML, Gill RT (2012) Identification of a 21 amino acid peptide conferring 3-hydroxypropionic acid stress-tolerance to E. coli. Biotechnol Bioeng 109(5):1347–1352. doi:10.1002/bit.24398
Zhou S, Catherine C, Rathnasingh C, Somasundar A, Park S (2013) Production of 3-hydroxypropionic acid from glycerol by recombinant P. denitrificans. Biotechnol Bioeng 110(12):3177–3187. doi:10.1002/bit.24980
Zhou S, Causey T, Hasona A, Shanmugam K, Ingram L (2003) Production of optically pure d-lactic acid in mineral salts medium by metabolically engineered E. coli W3110. Appl Environ Microbiol 69(1):399–407
Acknowledgement
This study was supported financially by the Korean Ministry of Education, Science, and Technology through the Advanced Biomass R&D Center (ABC; Grant No. 2010-0029799), KAIST, Korea. The authors are grateful for the financial assistance provided by the BK21 Plus Program for Advanced Chemical Technology at Pusan National University. In addition, Dr. Ashok Somasundar is grateful for the financial support provided by “2014 Post-Doc. Development Program of Pusan National University”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sankaranarayanan, M., Ashok, S. & Park, S. Production of 3-hydroxypropionic acid from glycerol by acid tolerant Escherichia coli . J Ind Microbiol Biotechnol 41, 1039–1050 (2014). https://doi.org/10.1007/s10295-014-1451-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1451-2