Abstract
Plant organs are repetitively generated at the shoot apical meristem (SAM) in recognizable patterns. This phenomenon, known as phyllotaxis, has long fascinated scientists from different disciplines. While we have an enriched body of knowledge on phyllotactic patterns, parameters, and transitions, only in the past 20 years, however, have we started to identify genes and elucidate genetic pathways that involved in phyllotaxis. In this review, I first summarize the classical knowledge of phyllotaxis from a morphological perspective. I then discuss recent advances in the regulation of phyllotaxis, from a molecular genetics perspective. I show that the morphological beauty of phyllotaxis we appreciate is the manifestation of many regulators, in addition to the critical role of auxin as a patterning signal, exerting their respective effects in a coordinated fashion either directly or indirectly in the SAM.




Modified from Douady and Couder (1996b) with permission from Elsevier. d divergence angle formed by primordia 1 and 2, R0 radius of the shoot apical meristem (SAM), r1 distance from primordium 1 to the center of the SAM, r2: distance from primordium 2 to the center of the SAM, ψ/2 half conic angle


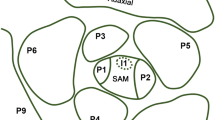
Modified from Reinhardt et al. (2003) with permission from Springer Nature. c Auxin distribution in Arabidopsis inflorescence meristem visualized by DII-VENUS (green, absence of auxin). Inset: overlay with autofluorescence signal (red). Reprinted from Vernoux et al. (2011). d Simulated SAM exhibiting a spiral pattern. Higher to lower auxin concentration is indicated by brighter to darker green. PIN1 localization is indicated by red. Reprinted from Smith et al. (2006), copyright (2006) National Academy of Sciences. e, f Conceptual model of polar auxin transport determining the position of organ formation. Auxin is transported acropetally toward the SAM (e) and accumulates in young primordia (f) under the guidance of polarly localized PIN1 (light blue). Polar auxin transport is indicated by blue arrows. Auxin transport from inner cells to the epidermal cells is indicated by green arrows. Auxin gradient is indicated by red. Insets: depicted area in the SAM. Modified from Reinhardt (2005) with permission from Elsevier. I incipient primordia, P visible primordia, SAM shoot apical meristem


Similar content being viewed by others
References
Abe K, Osakabe K, Ishikawa Y, Tagiri A, Yamanouchi H, Takyuu T, Yoshioka T, Ito T, Kobayashi M, Shinozaki K, Ichikawa H, Toki S (2009) Inefficient double-strand DNA break repair is associated with increased fasciation in Arabidopsis BRCA2 mutants. J Exp Bot 60:2751–2761
Abley K, Sauret-Gueto S, Maree AF, Coen E (2016) Formation of polarity convergences underlying shoot outgrowths. Elife 5:e18165
Adler I (1974) A model of contact pressure in phyllotaxis. J Theor Biol 45:1–79
Adler I, Barabé D, Jean RV (1997) A history of the study of phyllotaxis. Ann Bot 80:231–244
Aida M, Ishida T, Tasaka M (1999) Shoot apical meristem and cotyledon formation during Arabidopsis embryogenesis: interaction among the CUP-SHAPED COTYLEDON and SHOOT MERISTEMLESS genes. Development 126:1563–1570
Airy H (1873) On leaf-arrangement. Proc R Soc Lond B 21:176–179
Alexander DL, Mellor EA, Langdale JA (2005) CORKSCREW1 defines a novel mechanism of domain specification in the maize shoot. Plant Physiol 138:1396–1408
Amagai Y, Martinek P, Kuboyama T, Watanabe N (2017) Microsatellite mapping of the Scr1 locus conferring screwed spike rachis to modify the spatial orientation of spikelets in Triticum aestivum L. Genet Resour Crop Evol 64:1569–1579
Anastasiou E, Kenz S, Gerstung M, MacLean D, Timmer J, Fleck C, Lenhard M (2007) Control of plant organ size by KLUH/CYP78A5-dependent intercellular signaling. Dev Cell 13:843–856
Bainbridge K, Guyomarc’h S, Bayer E, Swarup R, Bennett M, Mandel T, Kuhlemeier C (2008) Auxin influx carriers stabilize phyllotactic patterning. Genes Dev 22:810–823
Banasiak A, Zagórska-Marek B (2006) Signals flowing from mature tissues to shoot apical meristem affect phyllotaxis in coniferous shoot. Acta Soc Bot Pol 75:113–121
Barabé D, Lacroix C (2020) Phyllotactic patterns: a multidisciplinary approach. World Scientific, Singapore
Barabé D, Bourque L, Yin X, Lacroix C (2010) Phyllotaxis of the palm Euterpe oleracea Mart. at the level of the shoot apical meristem. Botany 88:528–536
Bartrina I, Otto E, Strnad M, Werner T, Schmulling T (2011) Cytokinin regulates the activity of reproductive meristems, flower organ size, ovule formation, and thus seed yield in Arabidopsis thaliana. Plant Cell 23:69–80
Bayer EM, Smith RS, Mandel T, Nakayama N, Sauer M, Prusinkiewicz P, Kuhlemeier C (2009) Integration of transport-based models for phyllotaxis and midvein formation. Genes Dev 23:373–384
Besnard F, Refahi Y, Morin V, Marteaux B, Brunoud G, Chambrier P, Rozier F, Mirabet V, Legrand J, Laine S, Thevenon E, Farcot E, Cellier C, Das P, Bishopp A, Dumas R, Parcy F, Helariutta Y, Boudaoud A, Godin C, Traas J, Guedon Y, Vernoux T (2014) Cytokinin signalling inhibitory fields provide robustness to phyllotaxis. Nature 505:417–421
Bhatia N, Bozorg B, Larsson A, Ohno C, Jonsson H, Heisler MG (2016) Auxin acts through MONOPTEROS to regulate plant cell polarity and pattern phyllotaxis. Curr Biol 26:3202–3208
Bichet A, Desnos T, Turner S, Grandjean O, Hofte H (2001) BOTERO1 is required for normal orientation of cortical microtubules and anisotropic cell expansion in Arabidopsis. Plant J 25:137–148
Bolle F (1939) Theorie der blattstellung. Verh Bot Prov Brandenb 79:152–192
Bommert P, Lunde C, Nardmann J, Vollbrecht E, Running M, Jackson D, Hake S, Werr W (2005) Thick tassel dwarf1 encodes a putative maize ortholog of the Arabidopsis CLAVATA1 leucine-rich repeat receptor-like kinase. Development 132:1235–1245
Bommert P, Nagasawa NS, Jackson D (2013) Quantitative variation in maize kernel row number is controlled by the FASCIATED EAR2 locus. Nat Genet 45:334–337
Brand U, Fletcher JC, Hobe M, Meyerowitz EM, Simon R (2000) Dependence of stem cell fate in Arabidopsis on a feedback loop regulated by CLV3 activity. Science 289:617–619
Braun A (1835) Dr. Carl Schimper’s vorträge über die möglichkeit eines wissen-schaftlichen verstandnis der blattstellung, nebst andeutung de hauptsächlichen blattstellungsgesetze und insbesondere der neuentdeckten gesetze der aneinanderreihung von cyclen verschiedener maasse. Flora 18:145–191
Bravais L, Bravais A (1837) Essai sur la disposition des feuilles curvisériées. Ann Sci Nat Bot (2) 7:42–110; 193–221; 291–348; 8:11–42
Braybrook SA, Peaucelle A (2013) Mechano-chemical aspects of organ formation in Arabidopsis thaliana: the relationship between auxin and pectin. PLoS ONE 8:e57813
Bringmann M, Li E, Sampathkumar A, Kocabek T, Hauser MT, Persson S (2012) POM-POM2/cellulose synthase interacting1 is essential for the functional association of cellulose synthase and microtubules in Arabidopsis. Plant Cell 24:163–177
Burian A, Raczynska-Szajgin M, Borowska-Wykret D, Piatek A, Aida M, Kwiatkowska D (2015) The CUP-SHAPED COTYLEDON2 and 3 genes have a post-meristematic effect on Arabidopsis thaliana phyllotaxis. Ann Bot 115:807–820
Burk DH, Ye ZH (2002) Alteration of oriented deposition of cellulose microfibrils by mutation of a katanin-like microtubule-severing protein. Plant Cell 14:2145–2160
Burko Y, Geva Y, Refael-Cohen A, Shleizer-Burko S, Shani E, Berger Y, Halon E, Chuck G, Moshelion M, Ori N (2011) From organelle to organ: ZRIZI MATE-type transporter is an organelle transporter that enhances organ initiation. Plant Cell Physiol 52:518–527
Byrne ME, Groover AT, Fontana JR, Martienssen RA (2003) Phyllotactic pattern and stem cell fate are determined by the Arabidopsis homeobox gene BELLRINGER. Development 130:3941–3950
Callos JD, DiRado M, Xu B, Behringer FJ, Link BM, Medford JI (1994) The forever young gene encodes an oxidoreductase required for proper development of the Arabidopsis vegetative shoot apex. Plant J 6:835–847
Chapman JM, Perry R (1987) A diffusion model of phyllotaxis. Ann Bot 60:377–389
Chaudhury AM, Letham S, Craig S, Dennis ES (1993) amp1—a mutant with high cytokinin levels and altered embryonic pattern, faster vegetative growth, constitutive photomorphogenesis and precocious flowering. Plant J 4:907–916
Chen Q, Atkinson A, Otsuga D, Christensen T, Reynolds L, Drews GN (1999) The Arabidopsis FILAMENTOUS FLOWER gene is required for flower formation. Development 126:2715–2726
Chen MK, Wilson RL, Palme K, Ditengou FA, Shpak ED (2013) ERECTA family genes regulate auxin transport in the shoot apical meristem and forming leaf primordia. Plant Physiol 162:1978–1991
Chu H, Qian Q, Liang W, Yin C, Tan H, Yao X, Yuan Z, Yang J, Huang H, Luo D, Ma H, Zhang D (2006) The FLORAL ORGAN NUMBER4 gene encoding a putative ortholog of Arabidopsis CLAVATA3 regulates apical meristem size in rice. Plant Physiol 142:1039–1052
Chuang C, Running M, Williams RW, Meyerowitz EM (1999) The PERIANTHIA gene encodes a bZIP protein involved in the determination of floral organ number in Arabidopsis thaliana. Genes Dev 13:334–344
Chuck G, Cigan AM, Saeteurn K, Hake S (2007) The heterochronic maize mutant Corngrass1 results from overexpression of a tandem microRNA. Nat Genet 39:544–549
Chuck G, Whipple C, Jackson D, Hake S (2010) The maize SBP-box transcription factor encoded by tasselsheath4 regulates bract development and the establishment of meristem boundaries. Development 137:1243–1250
Chuck GS, Brown PJ, Meeley R, Hake S (2014) Maize SBP-box transcription factors unbranched2 and unbranched3 affect yield traits by regulating the rate of lateral primordia initiation. Proc Natl Acad Sci U S A 111:18775–18780
Church AH (1904) On the relation of phyllotaxis to mechanical laws. Williams and Norgate, London
Clark SE, Running M, Meyerowitz EM (1993) CLAVATA1, a regulator of meristem and flower development in Arabidopsis. Development 119:397–418
Clark SE, Running M, Meyerowitz EM (1995) CLAVATA3 is a specific regulator of shoot and floral meristem development affecting the same processes as CLAVATA1. Development 121:2057–2067
Clark SE, Williams RW, Meyerowitz EM (1997) The CLAVATA1 gene encodes a putative receptor kinase that controls shoot and floral meristem size in Arabidopsis. Cell 89:575–585
Clarke J, Tack D, Findlay K, Van Montagu M, Van Lijsebettens M (1999) The SERRATE locus controls the formation of the early juvenile leaves and phase length in Arabidopsis. Plant J 20:493–501
Couder Y (1998) Initial transitions, order and disorder in phyllotactic patterns: the ontogeny of Helianthus annuus. a case study. Acta Soc Bot Pol 67:129–150
Darwin C (1872) The origin of species. John Murray, London
De Reuille PB, Bohn-Courseau I, Ljung K, Morin H, Carraro N, Godin C, Traas J (2006) Computer simulations reveal properties of the cell-cell signaling network at the shoot apex in Arabidopsis. Proc Natl Acad Sci U S A 103:1627–1632
Deb Y, Marti D, Frenz M, Kuhlemeier C, Reinhardt D (2015) Phyllotaxis involves auxin drainage through leaf primordia. Development 142:1992–2001
Douady S, Couder Y (1992) Phyllotaxis as a physical self-organized growth process. Phys Rev Lett 68:2098–2101
Douady S, Couder Y (1996a) Phyllotaxis as a dynamical self organizing process part I: the spiral modes resulting from time-periodic iterations. J Theor Biol 178:255–274
Douady S, Couder Y (1996b) Phyllotaxis as a dynamical self organizing process part II: the spontaneous formation of a periodicity and the coexistence of spiral and whorled patterns. J Theor Biol 178:275–294
Douady S, Couder Y (1996c) Phyllotaxis as a dynamical self organizing process part III: the simulation of the transient regimes of ontogeny. J Theor Biol 178:295–312
Douady S, Couder Y (1998) The phyllotactic patterns as resulting from self-organization in an iterative process. In: Jean RV, Barabé D (eds) Symmetry in plants. World Scientific, Singapore, pp 539–570
Doust AN (2001) The developmental basis of floral variation in Drimys winteri (Winteraceae). Int J Plant Sci 162:697–717
Erickson RO, Meicenheimer RD (1977) Photoperiod induced change in phyllotaxis in Xanthium. Am J Bot 64:981–988
Etchells JP, Moore L, Jiang W, Prescott H, Capper R, Saunders NJ, Bhatt AM, Dickinson HG (2012) A role for BELLRINGER in cell wall development is supported by loss-of-function phenotypes. BMC Plant Biol 12:212
Evans MMS, Poethig RS (1997) The viviparous8 mutation delays vegetative phase change and accelerates the rate of seedling growth in maize. Plant J 12:769–779
Fal K, Liu M, Duisembekova A, Refahi Y, Haswell ES, Hamant O (2017) Phyllotactic regularity requires the Paf1 complex in Arabidopsis. Development 144:4428–4436
Fambrini M, Bonsignori E, Rapparini F, Cionini G, Michelotti V, Bertini D, Baraldi R, Pugliesi C (2006) stem fasciated, a recessive mutation in sunflower (Helianthus annuus), alters plant morphology and auxin level. Ann Bot 98:715–730
Feraru E, Feraru MI, Kleine-Vehn J, Martiniere A, Mouille G, Vanneste S, Vernhettes S, Runions J, Friml J (2011) PIN polarity maintenance by the cell wall in Arabidopsis. Curr Biol 21:338–343
Fierz V (2015) Aberrant phyllotactic patterns in cones of some conifers: a quantitative study. Acta Soc Bot Pol 84:261–265
Fleming AJ, McQueen-Mason S, Mandel T, Kuhlemeier C (1997) Induction of leaf primordia by the cell wall protein expansin. Science 276:1415–1418
Fletcher JC, Brand U, Running M, Simon R, Meyerowitz EM (1999) Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 283:1911–1914
Friml J, Yang X, Michniemicz M, Weijers D, Quint A, Tietz O, Benjamins R, Ouwerkerk PBF, Ljung K, Sandberg G, Hooykaas PJJ, Palme K, Offringa R (2004) A PINOID-dependent binary switch in apical-basal PIN polar targeting directs auxin efflux. Science 306:862–865
Fujita T (1937) Über die reihe 2, 5, 7, 12 …. in der schraubigen blattstellung und die mathematische betrachtung verschiedener zahlenreihensysteme. Bot Mag Tokyo 51:298–307
Fujita T (1938) Statistiche untersuchung über die zahl der konjugierten parastichen bei den schraubigen organstellungen. Bot Mag Tokyo 52:425–433
Fujita T (1939) Statistische untersuchungen über den divergenzwinkel bei den schraubigen organstellungen. Bot Mag Tokyo 53:194–199
Fujita T (1942) Zur Kenntnis der Organstellungen im Pflanzenreich. Jap J Bot 12:1–55
Fujita H, Toyokura K, Okada K, Kawaguchi M (2011) Reaction-diffusion pattern in shoot apical meristem of plants. PLoS ONE 6:e18243
Furutani M, Kajiwara T, Kato T, Treml BS, Stockum C, Torres-Ruiz RA, Tasaka M (2007) The gene MACCHI-BOU 4/ENHANCER OF PINOID encodes a NPH3-like protein and reveals similarities between organogenesis and phototropism at the molecular level. Development 134:3849–3859
Furutani M, Nakano Y, Tasaka M (2014) MAB4-induced auxin sink generates local auxin gradients in Arabidopsis organ formation. Proc Natl Acad Sci U S A 111:1198–1203
Galvan-Ampudia CS, Cerutti G, Legrand J, Brunoud G, Martin-Arevalillo R, Azais R, Bayle V, Moussu S, Wenzl C, Jaillais Y, Lohmann JU, Godin C, Vernoux T (2020) Temporal integration of auxin information for the regulation of patterning. Elife 9:e55832
Galweiler L, Guan C, Muller A, Wisman E, Mendgen K, Yephremov A, Palme K (1998) Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282:2226–2230
Garrett JJ, Meents MJ, Blackshaw MT, Blackshaw LC, Hou H, Styranko DM, Kohalmi SE, Schultz EA (2012) A novel, semi-dominant allele of MONOPTEROS provides insight into leaf initiation and vein pattern formation. Planta 236:297–312
Giulini A, Wang J, Jackson D (2004) Control of phyllotaxy by the cytokinin-inducible response regulator homologue ABPHYL1. Nature 430:1031–1034
Gola EM (1996) Phyllotaxis diversity in Lycopodium clavatum L. and Lycopodium annotinum L. Acta Soc Bot Pol 65:235–247
Gola EM (1997) Phyllotactic spectra in cacti: Mammillaria species and some genera from Rebutia group. Acta Soc Bot Pol 66:237–257
Gola EM, Banasiak A (2016) Diversity of phyllotaxis in land plants in reference to the shoot apical meristem structure. Acta Soc Bot Pol 85:3529
Goldshmidt A, Alvarez JP, Bowman JL, Eshed Y (2008) Signals derived from YABBY gene activities in organ primordia regulate growth and partitioning of Arabidopsis shoot apical meristems. Plant Cell 20:1217–1230
Golz JF, Roccaro M, Kuzoff R, Hudson A (2004) GRAMINIFOLIA promotes growth and polarity of Antirrhinum leaves. Development 131:3661–3670
Gordon SP, Chickarmane VS, Ohno C, Meyerowitz EM (2009) Multiple feedback loops through cytokinin signaling control stem cell number within the Arabidopsis shoot meristem. Proc Natl Acad Sci U S A 106:16529–16534
Graf P, Dolzblasz A, Wurschum T, Lenhard M, Pfreundt U, Laux T (2010) MGOUN1 encodes an Arabidopsis type IB DNA topoisomerase required in stem cell regulation and to maintain developmentally regulated gene silencing. Plant Cell 22:716–728
Green PB (1992) Pattern formation in shoots: a likely role for minimal energy configurations of the tunica. Int J Plant Sci 153:S59–S75
Green PB (1999) Expression of pattern in plants: combining molecular and calculus-based biophysical paradigms. Am J Bot 86:1059–1076
Green PB, Steele C, Rennich SC (1996) Phyllotactic patterns: a biophysical mechanism for their origin. Ann Bot 77:515–527
Green PB, Steele C, Rennich SC (1998) How plants produce pattern. A review and a proposal that undulating field is the mechanism. In: Jean RV, Barabé D (eds) Symmetry in plants. World Scientific, Singapore, pp 359–392
Gruel J, Landrein B, Tarr P, Schuster C, Refahi Y, Sampathkumar A, Hamant O, Meyerowitz EM, Jonsson H (2016) An epidermis-driven mechanism positions and scales stem cell niches in plants. Sci Adv 2:e1500989
Guedon Y, Refahi Y, Besnard F, Farcot E, Godin C, Vernoux T (2013) Pattern identification and characterization reveal permutations of organs as a key genetically controlled property of post-meristematic phyllotaxis. J Theor Biol 338:94–110
Guenot B, Bayer E, Kierzkowski D, Smith RS, Mandel T, Zadnikova P, Benkova E, Kuhlemeier C (2012) PIN1-independent leaf initiation in Arabidopsis. Plant Physiol 159:1501–1510
Guo J, Chen JG (2008) RACK1 genes regulate plant development with unequal genetic redundancy in Arabidopsis. BMC Plant Biol 8:108
Guyomarc’h S, Vernoux T, Traas J, Zhou DX, Delarue M (2004) MGOUN3, an Arabidopsis gene with tetratricopeptide-repeat-related motifs, regulates meristem cellular organization. J Exp Bot 55:673–684
Hamant O, Heisler MG, Jonsson H, Krupinski P, Uyttewaal M, Bokov P, Corson F, Sahlin P, Boudaoud A, Meyerowitz EM, Couder Y, Traas J (2008) Developmental patterning by mechanical signals in Arabidopsis. Science 322:1650–1655
Hartmann FP, Barbier de Reuille P, Kuhlemeier C (2019) Toward a 3D model of phyllotaxis based on a biochemically plausible auxin-transport mechanism. PLoS Comput Biol 15:e1006896
Hashimura Y, Ueguchi C (2011) The ArabidopsisMERISTEM DISORGANIZATION 1 gene is required for the maintenance of stem cells through the reduction of DNA damage. Plant J 68:657–669
Heisler MG, Hamant O, Krupinski P, Uyttewaal M, Ohno C, Jonsson H, Traas J, Meyerowitz EM (2010) Alignment between PIN1 polarity and microtubule orientation in the shoot apical meristem reveals a tight coupling between morphogenesis and auxin transport. PLoS Biol 8:e1000516
Helliwell CA, Chin-Atkins AN, Wilson IW, Chapple R, Dennis ES, Chaudhury AM (2001) The Arabidopsis AMP1 gene encodes a putative glutamate carboxypeptidase. Plant Cell 13:2115–2125
Hernandez LF, Green PB (1993) Transductions for the expression of structural pattern: analysis in sunflower. Plant Cell 5:1725–1738
Hibara K, Karim MR, Takada S, Taoka K, Furutani M, Aida M, Tasaka M (2006) Arabidopsis CUP-SHAPED COTYLEDON3 regulates postembryonic shoot meristem and organ boundary formation. Plant Cell 18:2946–2957
Hofmeister W (1868) Allgemeine morphologie der gewachse. In: Bary A, Irmisch TH, Sachs J (eds) Handbuch der physiologischen botanik. Engelmann, Leipzig, pp 405–664
Hunter C, Sun H, Poethig RS (2003) The Arabidopsis heterochronic gene ZIPPY is an ARGONAUTE family member. Curr Biol 13:1734–1739
Ikeda K, Nagasawa N, Nagato Y (2005) ABERRANT PANICLE ORGANIZATION 1 temporally regulates meristem identity in rice. Dev Biol 282:349–360
Ikeda K, Ito M, Nagasawa N, Kyozuka J, Nagato Y (2007) Rice ABERRANT PANICLE ORGANIZATION 1, encoding an F-box protein, regulates meristem fate. Plant J 51:1030–1040
Ikeda T, Tanaka W, Toriba T, Suzuki C, Maeno A, Tsuda K, Shiroishi T, Kurata T, Sakamoto T, Murai M, Matsusaka H, Kumamaru T, Hirano HY (2019) BELL1-like homeobox genes regulate inflorescence architecture and meristem maintenance in rice. Plant J 98:465–478
Ikeda-Kawakatsu K, Yasuno N, Oikawa T, Iida S, Nagato Y, Maekawa M, Kyozuka J (2009) Expression level of ABERRANT PANICLE ORGANIZATION1 determines rice inflorescence form through control of cell proliferation in the meristem. Plant Physiol 150:736–747
Ikeda-Kawakatsu K, Maekawa M, Izawa T, Itoh J, Nagato Y (2012) ABERRANT PANICLE ORGANIZATION 2/RFL, the rice ortholog of Arabidopsis LEAFY, suppresses the transition from inflorescence meristem to floral meristem through interaction with APO1. Plant J 69:168–180
Ishida T, Aida M, Takada S, Tasaka M (2000) Involvement of CUP-SHAPED COTYLEDON genes in gynoecium and ovule development in Arabidopsis thaliana. Plant Cell Physiol 41:60–67
Itoh J, Hasegawa A, Kitano H, Nagato Y (1998) A recessive heterochronic mutation, plastochron1, shortens the plastochron and elongates the vegetative phase in rice. Plant Cell 10:1511–1521
Itoh J, Kitano H, Matsuoka M, Nagato Y (2000) SHOOT ORGANIZATION genes regulate shoot apical meristem organization and the pattern of leaf primordium initiation in rice. Plant Cell 12:2161–2174
Itoh J, Hibara K, Kojima M, Sakakibara H, Nagato Y (2012) Rice DECUSSATE controls phyllotaxy by affecting the cytokinin signaling pathway. Plant J 72:869–881
Jackson D, Hake S (1999) Control of phyllotaxy in maize by the abphyl1 gene. Development 126:315–323
Jackson MDB, Duran-Nebreda S, Kierzkowski D, Strauss S, Xu H, Landrein B, Hamant O, Smith RS, Johnston IG, Bassel GW (2019) Global topological order emerges through local mechanical control of cell divisions in the Arabidopsis shoot apical meristem. Cell Syst 8:53–65
Je BI, Gruel J, Lee YK, Bommert P, Arevalo ED, Eveland AL, Wu Q, Goldshmidt A, Meeley R, Bartlett M, Komatsu M, Sakai H, Jonsson H, Jackson D (2016) Signaling from maize organ primordia via FASCIATED EAR3 regulates stem cell proliferation and yield traits. Nat Genet 48:785–791
Jean RV (1994) Phyllotaxis: a systemic study in plant morphogenesis. Cambridge University Press, Cambridge
Jean RV, Barabé (2001) Application of two mathematical models to the Araceae, a family of plants with enigmatic phyllotaxis. Ann Bot 88:173–186
Jonsson H, Heisler MG, Shapiro BE, Meyerowitz EM, Mjolsness E (2006) An auxin-driven polarized transport model for phyllotaxis. Proc Natl Acad Sci U S A 103:1633–1638
Kawakatsu T, Itoh J, Miyoshi K, Kurata N, Alvarez N, Veit B, Nagato Y (2006) PLASTOCHRON2 regulates leaf initiation and maturation in rice. Plant Cell 18:612–625
Kawakatsu T, Taramino G, Itoh J, Allen J, Sato Y, Hong SK, Yule R, Nagasawa N, Kojima M, Kusaba M, Sakakibara H, Sakai H, Nagato Y (2009) PLASTOCHRON3/GOLIATH encodes a glutamate carboxypeptidase required for proper development in rice. Plant J 58:1028–1040
Kaya H, Shibahara K, Taoka K, Iwabuchi M, Stillman B, Araki T (2001) FASCIATA genes for chromatin assembly factor-1 in Arabidopsis maintain the cellular organization of apical meristems. Cell 104:131–142
Kayes JM, Clark SE (1998) CLAVATA2, a regulator of meristem and organ development in Arabidopsis. Development 125:3843–3851
Kelly WJ, Cooke TJ (2003) Geometrical relationships specifying the phyllotactic pattern of aquatic plants. Am J Bot 90:1131–1143
Ki D, Sasayama D, Cho HT (2016) The M3 phosphorylation site is required for trafficking and biological roles of PIN-FORMED1, 2, and 7 in Arabidopsis. Front Plant Sci 7:1479
Kierzkowski D, Lenhard M, Smith R, Kuhlemeier C (2013) Interaction between meristem tissue layers controls phyllotaxis. Dev Cell 26:616–628
Kirchoff BK, Rutishauser R (1990) The phyllotaxy of costus (Costaceae). Bot Gaz 151:88–105
Krogan NT, Marcos D, Weiner AI, Berleth T (2016) The auxin response factor MONOPTEROS controls meristem function and organogenesis in both the shoot and root through the direct regulation of PIN genes. New Phytol 212:42–50
Krusell L, Sato N, Fukuhara I, Koch BE, Grossmann C, Okamoto S, Oka-Kira E, Otsubo Y, Aubert G, Nakagawa T, Sato S, Tabata S, Duc G, Parniske M, Wang TL, Kawaguchi M, Stougaard J (2011) The Clavata2 genes of pea and Lotus japonicus affect autoregulation of nodulation. Plant J 65:861–871
Kurakawa T, Ueda N, Maekawa M, Kobayashi K, Kojima M, Nagato Y, Sakakibara H, Kyozuka J (2007) Direct control of shoot meristem activity by a cytokinin-activating enzyme. Nature 445:652–655
Kwiatkowska D (1995) Ontogenetic changes of phyllotaxis in Anagallis arvensis L. Acta Soc Bot Pol 64:319–325
Kwiatkowska D (1997) Intraspecific variation of phyllotaxis stability in Anagallis arvensis. Acta Soc Bot Pol 66:259–271
Kwiatkowska D (1999) Formation of pseudowhorls in Peperomia verticillata (L.) A. Dietr. shoots exhibiting various phyllotactic patterns. Ann Bot 83:675–685
Kwon CS, Chen C, Wagner D (2005) WUSCHEL is a primary target for transcriptional regulation by SPLAYED in dynamic control of stem cell fate in Arabidopsis. Genes Dev 19:992–1003
Kyozuka J, Konishi S, Nemoto K, Izawa T, Shimamoto K (1998) Down-regulation of RFL, the FLO/LFY homolog of rice, accompanied with panicle branch initiation. Proc Natl Acad Sci U S A 95:1979–1982
Lacroix C, Sattler R (1988) Phyllotaxis theories and tepal-stamen superposition in Basella rubra. Am J Bot 75:906–917
Landrein B, Lathe R, Bringmann M, Vouillot C, Ivakov A, Boudaoud A, Persson S, Hamant O (2013) Impaired cellulose synthase guidance leads to stem torsion and twists phyllotactic patterns in Arabidopsis. Curr Biol 23:895–900
Landrein B, Refahi Y, Besnard F, Hervieux N, Mirabet V, Boudaoud A, Vernoux T, Hamant O (2015) Meristem size contributes to the robustness of phyllotaxis in Arabidopsis. J Exp Bot 66:1317–1324
Larson PR (1977) Phyllotactic transitions in the vascular system of Populus deltoides Bartr. as determined by 14C labeling. Planta 134:241–249
Laubinger S, Sachsenberg T, Zeller G, Busch W, Lohmann JU, Ratsch G, Weigel D (2008) Dual roles of the nuclear cap-binding complex and SERRATE in pre-mRNA splicing and microRNA processing in Arabidopsis thaliana. Proc Natl Acad Sci U S A 105:8795–8800
Laufs P, Dockx J, Kronenberger J, Traas J (1998a) MGOUN1 and MGOUN2: two genes required for primordium initiation at the shoot apical and floral meristems in Arabidopsis thaliana. Development 125:1253–1260
Laufs P, Grandjean O, Jonak C, Kieu K, Traas J (1998b) Cellular parameters of the shoot apical meristem in Arabidopsis. Plant Cell 10:1375–1389
Laux T, Mayer KFX, Berger J, Jurgens G (1996) The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122:87–96
Lee BH, Johnston R, Yang Y, Gallavotti A, Kojima M, Travencolo BA, Costa Lda F, Sakakibara H, Jackson D (2009) Studies of aberrant phyllotaxy1 mutants of maize indicate complex interactions between auxin and cytokinin signaling in the shoot apical meristem. Plant Physiol 150:205–216
Leehy KA, Lee JR, Song X, Renfrew KB, Shippen DE (2013) MERISTEM DISORGANIZATION1 encodes TEN1, an essential telomere protein that modulates telomerase processivity in Arabidopsis. Plant Cell 25:1343–1354
Leibfried A, To JP, Busch W, Stehling S, Kehle A, Demar M, Kieber JJ, Lohmann JU (2005) WUSCHEL controls meristem function by direct regulation of cytokinin-inducible response regulators. Nature 438:1172–1175
Leiboff S, Li X, Hu HC, Todt N, Yang J, Li X, Yu X, Muehlbauer GJ, Timmermans MC, Yu J, Schnable PS, Scanlon MJ (2015) Genetic control of morphometric diversity in the maize shoot apical meristem. Nat Commun 6:8974
Lestiboudois MT (1848) Phyllotaxie anatomique. Ann Sci Nat Bot 3(3):15–105
Leyser O, Furner IJ (1992) Characterisation of three shoot apical meristem mutants of Arabidopsis thaliana. Development 116:397–403
Li S, Lei L, Somerville CR, Gu Y (2012) Cellulose synthase interactive protein 1 (CSI1) links microtubules and cellulose synthase complexes. Proc Natl Acad Sci U S A 109:185–190
Li R, Li J, Li S, Qin G, Novak O, Pencik A, Ljung K, Aoyama T, Liu J, Murphy A, Gu H, Tsuge T, Qu LJ (2014) ADP1 affects plant architecture by regulating local auxin biosynthesis. PLoS Genet 10:e1003954
Li T, Yan A, Bhatia N, Altinok A, Afik E, Durand-Smet P, Tarr PT, Schroeder JI, Heisler MG, Meyerowitz EM (2019) Calcium signals are necessary to establish auxin transporter polarity in a plant stem cell niche. Nat Commun 10:726
Linardić M, Braybrook SA (2017) Towards an understanding of spiral patterning in the Sargassum muticum shoot apex. Sci Rep 7:13887
Lindsay DL, Sawhney VK, Bonham-Smith PC (2006) Cytokinin-induced changes in CLAVATA1 and WUSCHEL expression temporally coincide with altered floral development in Arabidopsis. Plant Sci 170:1111–1117
Liu C, Xu H, Han R, Wang S, Liu G, Chen S, Chen J, Bian X, Jiang J (2019) Overexpression of BpCUC2 influences leaf shape and internode development in Betula pendula. Int J Mol Sci 20:4722
Lobbes D, Rallapalli G, Schmidt DD, Martin C, Clarke J (2006) SERRATE: a new player on the plant microRNA scene. EMBO Rep 7:1052–1058
Lohmann D, Stacey N, Breuninger H, Jikumaru Y, Muller D, Sicard A, Leyser O, Yamaguchi S, Lenhard M (2010) SLOW MOTION is required for within-plant auxin homeostasis and normal timing of lateral organ initiation at the shoot meristem in Arabidopsis. Plant Cell 22:335–348
Luo L, Li W, Miura K, Ashikari M, Kyozuka J (2012) Control of tiller growth of rice by OsSPL14 and strigolactones, which work in two independent pathways. Plant Cell Physiol 53:1793–1801
Mähönen AP, Bishopp A, Higuchi M, Nieminen KM, Kinoshita K, Tormakangas K, Ikeda Y, Oka A, Kakimoto T, Helariutta Y (2006) Cytokinin signaling and its inhibitor AHP6 regulate cell fate during vascular development. Science 311:94–98
Maksymowych R, Erickson RO (1977) Phyllotactic change induced by gibberellic acid in Xanthium shoot apices. Am J Bot 64:33–44
Maksymowych R, Cordero RE, Erickson RO (1976) Long-term developmental changes in Xanthium induced by gibberellic acid. Am J Bot 63:1047–1053
Mandel T, Moreau F, Kutsher Y, Fletcher JC, Carles CC, Williams L (2014) The ERECTA receptor kinase regulates Arabidopsis shoot apical meristem size, phyllotaxy and floral meristem identity. Development 141:830–841
Mandel T, Candela H, Landau U, Asis L, Zelinger E, Carles CC, Williams LE (2016) Differential regulation of meristem size, morphology and organization by the ERECTA, CLAVATA and class III HD-ZIP pathways. Development 143:1612–1622
Marc J, Hackett WP (1991) Gibberellin-induced reorganization of spatial relationships of emerging leaf primordia at the shoot apical meristem in Hedera helix L. Planta 185:171–178
Marc J, Hackett WP (1992) Changes in the pattern of cell arrangement at the surface of the shoot apical meristem in Hedera helix L. following gibberellin treatment. Planta 186:503–510
Martinez CC, Koenig D, Chitwood DH, Sinha NR (2016) A sister of PIN1 gene in tomato (Solanum lycopersicum) defines leaf and flower organ initiation patterns by maintaining epidermal auxin flux. Dev Biol 419:85–98
Matkowski A, Adler K (1999) The diversity and ontogenetic changes of phyllotaxis in wild-types and two leaf form mutants of Antirrhinum majus L. Bot J Linn Soc 131:235–248
Mayer KFX, Schoof H, Haecker A, Lenhard M, Jurgens G, Laux T (1998) Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95:805–815
McConnell JR, Emery J, Eshed Y, Bao N, Bowman JL, Barton MK (2001) Role of PHABULOSA and PHAVOLUTA in determining radial patterning in shoots. Nature 411:709–713
McQueen-Mason S, Cosgrove DJ (1994) Disruption of hydrogen bonding between plant cell wall polymers by proteins that induce wall extension. Proc Natl Acad Sci U S A 91:6574–6578
McQueen-Mason S, Cosgrove DJ (1995) Expansin mode of action on cell walls. Plant Physiol 107:87–100
McQueen-Mason S, Durachko DM, Cosgrove DJ (1992) Two endogenous proteins that induce cell wall extension in plants. Plant Cell 4:1425–1433
Medford JI, Behringer FJ, Callos JD, Feldmann KA (1992) Normal and abnormal development in the Arabidopsis vegetative shoot apex. Plant Cell 4:631–643
Meicenheimer RD (1979) Relationships between shoot growth and changing phyllotaxy of Ranunculus. Am J Bot 66:557–569
Meicenheimer RD (1981) Changes in Epilobium phyllotaxy induced by N-1-naphthylphthalamic acid and α-4-chlorophenoxyisobutyric acid. Am J Bot 68:1139–1154
Meicenheimer RD (1982) Epilobium phyllotaxy during reproductive transition. Am J Bot 69:1108–1118
Meicenheimer RD (1987) Role of stem growth in Linum usitatissimum leaf trace patterns. Am J Bot 74:857–867
Meicenheimer RD (1998) Decussate to spiral transitions in phyllotaxis. In: Jean RV, Barabé D (eds) Symmetry in plants. World Scientific, Singapore, pp 125–144
Meicenheimer RD, Zagórska-Marek B (1989) Consideration of the geometry of the phyllotaxic triangular unit and discontinuous phyllotactic transitions. J Theor Biol 139:359–368
Meinhardt H (1984) Models of pattern formation and their application to plant development. In: Barlow PW, Carr DJ (eds) Positional controls in plant development. Cambridge University Press, Cambridge, pp 1–32
Mimura M, Itoh J (2014) Genetic interaction between rice PLASTOCHRON genes and the gibberellin pathway in leaf development. Rice 7:25
Mimura M, Nagato Y, Itoh J (2012) Rice PLASTOCHRON genes regulate leaf maturation downstream of the gibberellin signal transduction pathway. Planta 235:1081–1089
Mingo-Castel AM, Gomez-Campo C, Tortosa ME, Pelacho AM (1984) Hormonal effects on phyllotaxis of Euphorbia lathyris L. Bot Mag Tokyo 97:171–178
Mirabet V, Besnard F, Vernoux T, Boudaoud A (2012) Noise and robustness in phyllotaxis. PLoS Comput Biol 8:e1002389
Mitchison GJ (1977) Phyllotaxis and the Fibonacci series. Science 196:270–275
Miyoshi K, Ahn B, Kawakatsu T, Ito Y, Itoh J, Nagato Y, Kurata N (2004) PLASTOCHRON1, a timekeeper of leaf initiation in rice, encodes cytochrome P450. Proc Natl Acad Sci U S A 101:875–880
Moss BL, Mao H, Guseman JM, Hinds TR, Hellmuth A, Kovenock M, Noorassa A, Lanctot A, Villalobos LI, Zheng N, Nemhauser JL (2015) Rate motifs tune auxin/indole-3-acetic acid degradation dynamics. Plant Physiol 169:803–813
Muller R, Bleckmann A, Simon R (2008) The receptor kinase CORYNE of Arabidopsis transmits the stem cell-limiting signal CLAVATA3 independently of CLAVATA1. Plant Cell 20:934–946
Nagasaki H, Itoh J, Hayashi K, Hibara K, Satoh-Nagasawa N, Nosaka M, Mukouhata M, Ashikari M, Kitano H, Matsuoka M, Nagato Y, Sato Y (2007) The small interfering RNA production pathway is required for shoot meristem initiation in rice. Proc Natl Acad Sci U S A 104:14867–14871
Nagasawa N, Miyoshi M, Kitano H, Satoh H, Nagato Y (1996) Mutations associated with floral organ number in rice. Planta 198:627–633
Nakayama N, Smith RS, Mandel T, Robinson S, Kimura S, Boudaoud A, Kuhlemeier C (2012) Mechanical regulation of auxin-mediated growth. Curr Biol 22:1468–1476
Navarro C, Efremova N, Golz JF, Rubiera R, Kuckenberg M, Castillo R, Tietz O, Saedler H, Schwarz-Sommer Z (2004) Molecular and genetic interactions between STYLOSA and GRAMINIFOLIA in the control of Antirrhinum vegetative and reproductive development. Development 131:3649–3659
O’Connor DL, Runions A, Sluis A, Bragg J, Vogel JP, Prusinkiewicz P, Hake S (2014) A division in PIN-mediated auxin patterning during organ initiation in grasses. PLoS Comput Biol 10:e1003447
O’Connor DL, Elton S, Ticchiarelli F, Hsia MM, Vogel JP, Leyser O (2017) Cross-species functional diversity within the PIN auxin efflux protein family. Elife 6:e31804
Okabe T (2016) The riddle of phyllotaxis: exquisite control of divergence angle. Acta Soc Bot Pol 85:3527
Okada K, Ueda J, Komaki MK, Bell CJ, Shimura Y (1991) Requirement of the auxin polar transport system in early stages of Arabídopsis floral bud formation. Plant Cell 3:677–684
Pautler M, Eveland AL, LaRue T, Yang F, Weeks R, Lunde C, Je BI, Meeley R, Komatsu M, Vollbrecht E, Sakai H, Jackson D (2015) FASCIATED EAR4 encodes a bZIP transcription factor that regulates shoot meristem size in maize. Plant Cell 27:104–120
Peaucelle A, Couder Y (2016) Fibonacci spirals in a brown alga [Sargassum muticum (Yendo) Fensholt] and in a land plant [Arabidopsis thaliana (L.) Heynh.]: a case of morphogenetic convergence. Acta Soc Bot Pol 85:3526
Peaucelle A, Morin H, Traas J, Laufs P (2007) Plants expressing a miR164-resistant CUC2 gene reveal the importance of post-meristematic maintenance of phyllotaxy in Arabidopsis. Development 134:1045–1050
Peaucelle A, Louvet R, Johansen JN, Hofte H, Laufs P, Pelloux J, Mouille G (2008) Arabidopsis phyllotaxis is controlled by the methyl-esterification status of cell-wall pectins. Curr Biol 18:1943–1948
Peaucelle A, Braybrook SA, Le Guillou L, Bron E, Kuhlemeier C, Hofte H (2011a) Pectin-induced changes in cell wall mechanics underlie organ initiation in Arabidopsis. Curr Biol 21:1720–1726
Peaucelle A, Louvet R, Johansen JN, Salsac F, Morin H, Fournet F, Belcram K, Gillet F, Hofte H, Laufs P, Mouille G, Pelloux J (2011b) The transcription factor BELLRINGER modulates phyllotaxis by regulating the expression of a pectin methylesterase in Arabidopsis. Development 138:4733–4741
Peng X, Chai W, Tan Y, Dong Q, Jiang H, Cheng B, Zhao Y (2017) Transcriptome analysis reveals critical genes and key pathways involved in early phyllotaxy development in maize. Genes Genom 39:15–26
Peragine A, Yoshikawa M, Wu G, Albrecht HL, Poethig RS (2004) SGS3 and SGS2/SDE1/RDR6 are required for juvenile development and the production of trans-acting siRNAs in Arabidopsis. Genes Dev 18:2368–2379
Pien S, Wyrzykowska J, McQueen-Mason S, Smart C, Fleming AJ (2001) Local expression of expansin induces the entire process of leaf development and modifies leaf shape. Proc Natl Acad Sci U S A 98:11812–11817
Pinon V, Prasad K, Grigg SP, Sanchez-Perez GF, Scheres B (2013) Local auxin biosynthesis regulation by PLETHORA transcription factors controls phyllotaxis in Arabidopsis. Proc Natl Acad Sci U S A 110:1107–1112
Prasad K, Grigg SP, Barkoulas M, Yadav RK, Sanchez-Perez GF, Pinon V, Blilou I, Hofhuis H, Dhonukshe P, Galinha C, Mähönen AP, Muller WH, Raman S, Verkleij AJ, Snel B, Reddy GV, Tsiantis M, Scheres B (2011) Arabidopsis PLETHORA transcription factors control phyllotaxis. Curr Biol 21:1123–1128
Prigge MJ, Wagner D (2001) The Arabidopsis SERRATE gene encodes a zinc-finger protein required for normal shoot dvelopment. Plant Cell 13:1263–1279
Prigge MJ, Otsuga D, Alonso JM, Ecker JR, Drews GN, Clark SE (2005) Class III homeodomain-leucine zipper gene family members have overlapping, antagonistic, and distinct roles in Arabidopsis development. Plant Cell 17:61–76
Prusinkiewicz P, Lindenmayer A (1990) The algorithmic beauty of plants. Springer-Verlag, New York
Przemeck GKH, Mattsson J, Hardtke CS, Sung ZR, Berleth T (1996) Studies on the role of the Arabidopsis gene MONOPTEROS in vascular development and plant cell axialization. Planta 200:229–237
Ranftl QL, Bastakis E, Klermund C, Schwechheimer C (2016) LLM-domain containing B-GATA factors control different aspects of cytokinin-regulated development in Arabidopsis thaliana. Plant Physiol 170:2295–2311
Reinhardt D (2005) Phyllotaxis—a new chapter in an old tale about beauty and magic numbers. Curr Opin Plant Biol 8:487–493
Reinhardt D, Wittwer F, Mandel T, Kuhlemeier C (1998) Localized upregulation of a new expansin gene predicts the site of leaf formation in the tomato meristem. Plant Cell 10:1427–1437
Reinhardt D, Mandel T, Kuhlemeier C (2000) Auxin regulates the initiation and radial position of plant lateral organs. Plant Cell 12:507–518
Reinhardt D, Pesce E, Stieger P, Mandel T, Baltensberger K, Bennett M, Traas J, Friml J, Kuhlemeier C (2003) Regulation of phyllotaxis by polar auxin transport. Nature 426:255–260
Rhoades MW, Reinhart BJ, Lim LP, Burge CB, Bartel B, Bartel DP (2002) Prediction of plant microRNA targets. Cell 110:513–520
Richards FJ (1948) The geometry of phyllotaxis and its origin. Symp Soc Exp Biol 2:217–245
Richards FJ (1951) Phyllotaxis: its quantitative expression and relation to growth in the apex. Philos Trans R Soc Lond B 235:509–564
Rogler CE, Hackett WP (1975) Phase change in Hedera helix: induction of the mature to juvenile phase change by gibberellin A3. Physiol Plant 34:141–147
Ronse De Craene LP (2010) Floral diagrams: and aid to understanding flower morphology and evolution. Cambridge University Press, Cambridge
Running M, Meyerowitz EM (1996) Mutations in the PERIANTHIA gene of Arabidopsis specifically alter floral organ number and initiation pattern. Development 122:1261–1269
Running M, Fletcher JC, Meyerowitz EM (1998) The WIGGUM gene is required for proper regulation of floral meristem size in Arabidopsis. Development 125:2545–2553
Rutishauser R (1998) Plastochrone ratio and leaf arc as parameters of a quantitative phyllotaxis analysis in vascular plants. In: Jean RV, Barabé D (eds) Symmetry in plants. World Scientific, Singapore, pp 171–212
Rutishauser R (1999) Polymerous leaf whorls in vascular plants: developmental morphology and fuzziness of organ identities. Int J Plant Sci 160:S81–S103
Rutishauser R (2016) Acacia (wattle) and Cananga (ylang-ylang): from spiral to whorled and irregular (chaotic) phyllotactic patterns—a pictorial report. Acta Soc Bot Pol 85:3531
Rutishauser R (2019) Ever since Darwin: why plants are important for evo-devo research. In: Fusco G (ed) Perspectives on evolutionary and developmental biology: essays for Alessandro Minelli. Padova University Press, Padova, pp 41–55
Ryu MY, Cho SK, Kim WT (2009) RNAi suppression of RPN12a decreases the expression of type-A ARRs, negative regulators of cytokinin signaling pathway, in Arabidopsis. Mol Cells 28:375–382
Sassi M, Ali O, Boudon F, Cloarec G, Abad U, Cellier C, Chen X, Gilles B, Milani P, Friml J, Vernoux T, Godin C, Hamant O, Traas J (2014) An auxin-mediated shift toward growth isotropy promotes organ formation at the shoot meristem in Arabidopsis. Curr Biol 24:2335–2342
Satoh N, Hong SK, Nishimura A, Matsuoka M, Kitano H, Nagato Y (1999) Initiation of shoot apical meristem in rice: characterization of four SHOOTLESS genes. Development 126:3629–3636
Satoh N, Itoh J, Nagato Y (2003) The SHOOTLESS2 and SHOOTLESS1 genes are involved in both initiation and maintenance of the shoot apical meristem through regulating the number of indeterminate cells. Genetics 164:335–346
Sawa S, Ito T, Shimura Y, Okada K (1999) FILAMENTOUS FLOWER controls the formation and development of Arabidopsis inflorescences and floral meristems. Plant Cell 11:69–86
Schimper CF (1830) Beschreibung des symphytum zeyheri und seiner zwei deutschen verwandten der S. bulborum Schimp. und S. tuberosum Jacqu. Mag Pharm 29:1–92
Schoof H, Lenhard M, Haecker A, Mayer KFX, Jurgens G, Laux T (2000) The stem cell population of Arabidopsis shoot meristems is maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100:635–644
Schoute JC (1913) Beitrage zur blattstellunglehre. I. die theorie. Rec Trav Bot Néerl 10:153–339
Schoute JC (1922) On whorled phyllotaxis. I Growth whorls. Rec Trav Bot Néerl 19:184–206
Schoute JC (1925) On whorled phyllotaxis. II. Late binding whorls of Peperomia. Rec Trav Bot Néerl 22:128–172
Schwab R, Palatnik JF, Riester M, Schommer C, Schmid M, Weigel D (2005) Specific effects of microRNAs on the plant transcriptome. Dev Cell 8:517–527
Schwabe WW (1971) Chemical modification of phyllotaxis. Symp Soc Exp Biol 25:301–322
Schwabe WW, Clewer AG (1984) Phyllotaxis—a simple computer model based on the theory of a polarly-translocated inhibitor. J Theor Biol 109:595–619
Schwarz S, Grande AV, Bujdoso N, Saedler H, Huijser P (2008) The microRNA regulated SBP-box genes SPL9 and SPL15 control shoot maturation in Arabidopsis. Plant Mol Biol 67:183–195
Shi B, Guo X, Wang Y, Xiong Y, Wang J, Hayashi KI, Lei J, Zhang L, Jiao Y (2018) Feedback from lateral organs controls shoot apical meristem growth by modulating auxin transport. Dev Cell 44:204–216
Shipman PD, Newell AC (2005) Polygonal planforms and phyllotaxis on plants. J Theor Biol 236:154–197
Sieber P, Wellmer F, Gheyselinck J, Riechmann JL, Meyerowitz EM (2007) Redundancy and specialization among plant microRNAs: role of the MIR164 family in developmental robustness. Development 134:1051–1060
Simonini S, Bencivenga S, Trick M, Ostergaard L (2017) Auxin-induced modulation of ETTIN activity orchestrates gene expression in Arabidopsis. Plant Cell 29:1864–1882
Smith R, Guyomarc’h S, Mandel T, Reinhardt D, Kuhlemeier C, Prusinkiewicz P (2006) A plausible model of phyllotaxis. Proc Natl Acad Sci U S A 103:1301–1306
Snow R (1952) On the shoot apex and phyllotaxis of Costus. New Phytol 51:359–363
Snow M, Snow R (1931) Experiments on phyllotaxis. I. The effect of isolating a primordium. Philos Trans R Soc Lond B 221:1–43
Snow M, Snow R (1933) Experiments on phyllotaxis. II. – the effect of displacing a primordium. Philos Trans R Soc Lond B 222:353–400
Snow M, Snow R (1935) Experiments on phyllotaxis. III. Diaganol splits through decussate apices. Philos Trans R Soc Lond B 225:63–94
Snow M, Snow R (1937) Auxin and leaf formation. New Phytol 36:1–18
Snow M, Snow R (1947) On the determination of leaves. New Phytol 46:5–19
Soma K (1958) Morphogenesis in the shoot apex of Euphorbia lathyris L. J Fac Sci Univ Tokyo III 7:199–256
Song X, Leehy K, Warrington R, Lamb JC, Surovtseva YV, Shippen DE (2008) STN1 protects chromosome ends in Arabidopsis thaliana. Proc Natl Acad Sci U S A 105:19815–19820
Steeves TA, Sussex IM (1989) Patterns in plant development. Cambridge University Press, Cambridge
Stevenson DW (1976) Observations on phyllotaxis, stelar morphology, the shoot apex and gemmae of Lycopodium lucidulum Michaux (Lycopodiaceae). Bot J Linn Soc 72:81–100
Stieger P, Reinhardt D, Kuhlemeier C (2002) The auxin influx carrier is essential for correct leaf positioning. Plant J 32:509–517
Stoma S, Lucas M, Chopard J, Schaedel M, Traas J, Godin C (2008) Flux-based transport enhancement as a plausible unifying mechanism for auxin transport in meristem development. PLoS Comput Biol 4:e1000207
Strauss S, Lempe J, Prusinkiewicz P, Tsiantis M, Smith RS (2020) Phyllotaxis: is the golden angle optimal for light capture? New Phytol 225:499–510
Surovtseva YV, Churikov D, Boltz KA, Song X, Lamb JC, Warrington R, Leehy K, Heacock M, Price CM, Shippen DE (2009) Conserved telomere maintenance component 1 interacts with STN1 and maintains chromosome ends in higher eukaryotes. Mol Cell 36:207–218
Suzaki T, Sato M, Ashikari M, Miyoshi M, Nagato Y, Hirano HY (2004) The gene FLORAL ORGAN NUMBER1 regulates floral meristem size in rice and encodes a leucine-rich repeat receptor kinase orthologous to Arabidopsis CLAVATA1. Development 131:5649–5657
Suzaki T, Toriba T, Fujimoto M, Tsutsumi N, Kitano H, Hirano HY (2006) Conservation and diversification of meristem maintenance mechanism in Oryza sativa: function of the FLORAL ORGAN NUMBER2 gene. Plant Cell Physiol 47:1591–1602
Suzuki T, Inagaki S, Nakajima S, Akashi T, Ohto MA, Kobayashi M, Seki M, Shinozaki K, Kato T, Tabata S, Nakamura K, Morikami A (2004) A novel Arabidopsis gene TONSOKU is required for proper cell arrangement in root and shoot apical meristems. Plant J 38:673–684
Suzuki T, Nakajima S, Inagaki S, Hirano-Nakakita M, Matsuoka K, Demura T, Fukuda H, Morikami A, Nakamura K (2005) TONSOKU is expressed in S phase of the cell cycle and its defect delays cell cycle progression in Arabidopsis. Plant Cell Physiol 46:736–742
Suzuki M, Latshaw S, Sato Y, Settles AM, Koch KE, Hannah LC, Kojima M, Sakakibara H, McCarty DR (2008) The maize Viviparous8 locus, encoding a putative ALTERED MERISTEM PROGRAM1-like peptidase, regulates abscisic acid accumulation and coordinates embryo and endosperm development. Plant Physiol 146:1193–1206
Suzuki M, Sato Y, Wu S, Kang BH, McCarty DR (2015) Conserved functions of the MATE transporter BIG EMBRYO1 in regulation of lateral organ size and initiation rate. Plant Cell 27:2288–2300
Swinton J, Ochu E, Consortium MSITsS (2016) Novel Fibonacci and non-Fibonacci structure in the sunflower: results of a citizen science experiment. R Soc Open Sci 3:160091
Szczesny T, Routier-Kierzkowska AL, Kwiatkowska D (2009) Influence of clavata3-2 mutation on early flower development in Arabidopsis thaliana: quantitative analysis of changing geometry. J Exp Bot 60:679–695
Szymanowska-Pulka J (1994) Phyllotactic patterns in capitula of Carlina acaulis L. Acta Soc Bot Pol 63:229–245
Taguchi-Shiobara F, Yuan Z, Hake S, Jackson D (2001) The fasciated ear2 gene encodes a leucine-rich repeat receptor-like protein that regulates shoot meristem proliferation in maize. Genes Dev 15:2755–2766
Takahashi T, Matsuhara S, Abe M, Komeda Y (2002) Disruption of a DNA topoisomerase I gene affects morphogenesis in Arabidopsis. Plant Cell 14:2085–2093
Takeda S, Tadele Z, Hofmann I, Probst AV, Angelis KJ, Kaya H, Araki T, Mengiste T, Mittelsten Scheid O, Shibahara K, Scheel D, Paszkowski J (2004) BRU1, a novel link between responses to DNA damage and epigenetic gene silencing in Arabidopsis. Genes Dev 18:782–793
Tamura Y, Kitano H, Satoh H, Nagato Y (1992) A gene profoundly affecting shoot organization in the early phase of rice development. Plant Sci 82:91–99
Tang Y, Knap HT (1998) Fasciation mutation enhances meristematic activity and alters pattern formation in soybean. Int J Plant Sci 159:249–260
Thornley JHM (1975) Phyllotaxis I a mechanistic model. Ann Bot 39:491–507
To JP, Haberer G, Ferreira FJ, Deruere J, Mason MG, Schaller GE, Alonso JM, Ecker JR, Kieber JJ (2004) Type-A Arabidopsis response regulators are partially redundant negative regulators of cytokinin signaling. Plant Cell 16:658–671
Tokunaga H, Kojima M, Kuroha T, Ishida T, Sugimoto K, Kiba T, Sakakibara H (2012) Arabidopsis lonely guy (LOG) multiple mutants reveal a central role of the LOG-dependent pathway in cytokinin activation. Plant J 69:355–365
Torii KU, Mitsukawa N, Oosumi T, Matsuura Y, Yokoyama R, Whittier RF, Komeda Y (1996) The ArabidopsisERECTA gene encodes a putative receptor protein kinase with extracellular leucine-rich repeats. Plant Cell 8:735–746
Turing AM (1952) The chemical basis of morphogenesis. Philos Trans R Soc Lond B 237:37–72
Uchida N, Shimada M, Tasaka M (2013) ERECTA-family receptor kinases regulate stem cell homeostasis via buffering its cytokinin responsiveness in the shoot apical meristem. Plant Cell Physiol 54:343–351
Uyttewaal M, Burian A, Alim K, Landrein B, Borowska-Wykret D, Dedieu A, Peaucelle A, Ludynia M, Traas J, Boudaoud A, Kwiatkowska D, Hamant O (2012) Mechanical stress acts via katanin to amplify differences in growth rate between adjacent cells in Arabidopsis. Cell 149:439–451
Valladares F, Brites D (2004) Leaf phyllotaxis: does it really affect light capture? Plant Ecol 174:11–17
Van Iterson G (1907) Mathematische und mikroskopisch-anatomische studien über blattstellungen nebst betraschtungen über den schalenbau der miliolinen. Gustav Fischer, Jena
Veen AH, Lindenmayer A (1977) Diffusion mechanism for phyllotaxis. Plant Physiol 60:127–139
Veit B, Briggs SP, Schmidt RJ, Yanovsky MF, Hake S (1998) Regulation of leaf initiation by the terminal ear1 gene of maize. Nature 393:166–168
Vernoux T, Brunoud G, Farcot E, Morin V, Van den Daele H, Legrand J, Oliva M, Das P, Larrieu A, Wells D, Guedon Y, Armitage L, Picard F, Guyomarc’h S, Cellier C, Parry G, Koumproglou R, Doonan JH, Estelle M, Godin C, Kepinski S, Bennett M, De Veylder L, Traas J (2011) The auxin signalling network translates dynamic input into robust patterning at the shoot apex. Mol Syst Biol 7:508
Waites R, Hudson A (1995) phantastica: a gene required for dorsoventrality of leaves in Antirrhinum majus. Development 121:2143–2154
Waites R, Selvadurai HRN, Oliver IR, Hudson A (1998) The PHANTASTICA gene encodes a MYB transcription factor involved in growth and dorsoventrality of lateral organs in Antirrhinum. Cell 93:779–789
Wang JW, Schwab R, Czech B, Mica E, Weigel D (2008) Dual effects of miR156-targeted SPL genes and CYP78A5/KLUH on plastochron length and organ size in Arabidopsis thaliana. Plant Cell 20:1231–1243
Wardlaw CW (1949) Experiments on organogenesis in ferns. Growth (suppl) 13:93–131
Wardlaw CW (1957) Experimental and analytical studies of pteridophytes: XXXV. The effects of direct applications of various substances to the shoot apex of Dryopteris austriaca (D. aristata). Ann Bot 21:85–120
Wardlaw CW, Mitra GC (1958) Responses of a fern apex to gibberellic acid, kinetin and α-naphthaleneacetic acid. Nature 181:400–401
Werner T, Motyka V, Laucou V, Smets R, Van Onckelen H, Schmulling T (2003) Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15:2532–2550
Wiesner J (1875) Bemerkungen über rationale und irrationale divergenzen. Flora 58(113–115):139–143
Williams RF (1975) The shoot apex and leaf growth: a study in quantitative biology. Cambridge University Press, Cambridge
Williams L, Grigg SP, Xie M, Christensen S, Fletcher JC (2005) Regulation of Arabidopsis shoot apical meristem and lateral organ formation by microRNA miR166g and its AtHD-ZIP target genes. Development 132:3657–3668
Wu G, Poethig RS (2006) Temporal regulation of shoot development in Arabidopsis thaliana by miR156 and its target SPL3. Development 133:3539–3547
Xie Z, Allen E, Wilken A, Carrington JC (2005) DICER-LIKE 4 functions in trans-acting small interfering RNA biogenesis and vegetative phase change in Arabidopsis thaliana. Proc Natl Acad Sci U S A 102:12984–12989
Xiong GS, Hu XM, Jiao YQ, Yu YC, Chu CC, Li JY, Qian Q, Wang YH (2006) LEAFY HEAD2, which encodes a putative RNA-binding protein, regulates shoot development of rice. Cell Res 16:267–276
Xu C, Liberatore KL, MacAlister CA, Huang Z, Chu YH, Jiang K, Brooks C, Ogawa-Ohnishi M, Xiong G, Pauly M, Van Eck J, Matsubayashi Y, Van Der Knaap E, Lippman ZB (2015) A cascade of arabinosyltransferases controls shoot meristem size in tomato. Nat Genet 47:784–792
Yadav RK, Perales M, Gruel J, Girke T, Jonsson H, Reddy GV (2011) WUSCHEL protein movement mediates stem cell homeostasis in the Arabidopsis shoot apex. Genes Dev 25:2025–2030
Yalovsky S, Kulukian A, Rodriguez-Concepcion M, Young CA, Gruissem W (2000) Functional requirement of plant farnesyltransferase during development in Arabidopsis. Plant Cell 12:1267–1278
Yang L, Liu Z, Lu F, Dong A, Huang H (2006) SERRATE is a novel nuclear regulator in primary microRNA processing in Arabidopsis. Plant J 47:841–850
Yang F, Bui HT, Pautler M, Llaca V, Johnston R, Lee BH, Kolbe A, Sakai H, Jackson D (2015) A maize glutaredoxin gene, Abphyl2, regulates shoot meristem size and phyllotaxy. Plant Cell 27:121–131
Yin X, Lacroix C, Barabé D (2011) Phyllotactic transitions in seedlings: the case of Thuja occidentalis. Botany 89:387–396
Yin X, Meicenheimer RD (2017a) Anisotomous dichotomy results from an unequal bifurcation of the original shoot apical meristem in Diphasiastrum digitatum (Lycopodiaceae). Am J Bot 104:782–786
Yin X, Meicenheimer RD (2017b) The ontogeny, phyllotactic diversity, and discontinuous transitions of Diphasiastrum digitatum (Lycopodiaceae). Am J Bot 104:8–23
Yonekura T, Iwamoto A, Fujita H, Sugiyama M (2019) Mathematical model studies of the comprehensive generation of major and minor phyllotactic patterns in plants with a predominant focus on orixate phyllotaxis. PLoS Comput Biol 15:e1007044
Yoshida S, Mandel T, Kuhlemeier C (2011) Stem cell activation by light guides plant organogenesis. Genes Dev 25:1439–1450
Yoshida A, Ohmori Y, Kitano H, Taguchi-Shiobara F, Hirano HY (2012) ABERRANT SPIKELET AND PANICLE1, encoding a TOPLESS-related transcriptional co-repressor, is involved in the regulation of meristem fate in rice. Plant J 70:327–339
Yoshikawa M, Peragine A, Park MY, Poethig RS (2005) A pathway for the biogenesis of trans-acting siRNAs in Arabidopsis. Genes Dev 19:2164–2175
Yoshikawa T, Ito M, Sumikura T, Nakayama A, Nishimura T, Kitano H, Yamaguchi I, Koshiba T, Hibara K, Nagato Y, Itoh J (2014) The rice FISH BONE gene encodes a tryptophan aminotransferase, which affects pleiotropic auxin-related processes. Plant J 78:927–936
Young DA (1978) On the diffusion theory of phyllotaxis. J Theor Biol 71:421–432
Zagórska-Marek B (1985) Phyllotactic patterns and transitions in Abies balsamea. Can J Bot 63:1844–1854
Zagórska-Marek B (1987) Phyllotaxis triangular unit; phyllotactic transitions as the consequences of the apical wedge disclinations in a crystal-like pattern of the units. Acta Soc Bot Pol 56:229–255
Zagórska-Marek B (1994) Phyllotaxic diversity in Magnolia flowers. Acta Soc Bot Pol 63:117–137
Zhao F, Chen W, Sechet J, Martin M, Bovio S, Lionnet C, Long Y, Battu V, Mouille G, Moneger F, Traas J (2019) Xyloglucans and microtubules synergistically maintain meristem geometry and phyllotaxis. Plant Physiol 181:1191–1206
Ziegelhoffer EC, Medrano LJ, Meyerowitz EM (2000) Cloning of the Arabidopsis WIGGUM gene identifies a role for farnesylation in meristem development. Proc Natl Acad Sci U S A 97:7633–7638
Acknowledgements
I thank Prof. Denis Barabé, Prof. Jun-Ichi Itoh, Prof. Christian Lacroix, Prof. Munetaka Sugiyama, Dr. Teva Vernoux, and two anonymous reviewers for their valuable comments and suggestions. I also thank Prof. Christian Lacroix, Prof. Kiyotaka Okada, Prof. Beata Zagórska-Marek, and Dr. Teng Zhang for providing figures and Prof. Hirokazu Tsukaya for plant identification.
Funding
This work is supported by Japan Society for the Promotion of Science Postdoctoral Fellowship for Research in Japan (Grant No. P19085).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Xiaofeng Yin is a co-organizer of the JPR International Symposium “Beyond Fibonacci patterns and the golden angle: phyllotactic variations and their cellular origin” held during the 83th Annual Meeting of Botanical Society of Japan.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.








Rights and permissions
About this article
Cite this article
Yin, X. Phyllotaxis: from classical knowledge to molecular genetics. J Plant Res 134, 373–401 (2021). https://doi.org/10.1007/s10265-020-01247-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-020-01247-3