Abstract
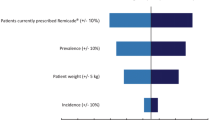
The first biosimilar monoclonal antibody (infliximab, CT-P13) was registered by the European Medicines Agency in 2013 for the treatment of several inflammatory conditions including rheumatoid arthritis (RA). Biosimilar infliximab is first being marketed in the Central and Eastern European countries. This paper presents the estimated budget impact of the introduction of biosimilar infliximab in RA over a 3-year time period in six selected countries, namely Bulgaria, the Czech Republic, Hungary, Poland, Romania and Slovakia. A prevalence-based model was constructed for budget impact analysis. Two scenarios were compared to the reference scenario (RSc) where no biosimilar infliximab is available: biosimilar scenario 1 (BSc1), where interchanging the originator infliximab with biosimilar infliximab is disallowed, and only patients who start new biological therapy are allowed to use biosimilar infliximab; as well as biosimilar scenario 2 (BSc2), where interchanging the originator infliximab with biosimilar infliximab is allowed, and 80 % of patients treated with originator infliximab are interchanged to biosimilar infliximab. Compared to the RSc, the net savings are estimated to be €15.3 or €20.8 M in BSc1 and BSc2, respectively, over the 3 years. If budget savings were spent on reimbursement of additional biosimilar infliximab treatment, approximately 1,200 or 1,800 more patients could be treated in the six countries within 3 years in the two biosimilar scenarios, respectively. The actual saving is most sensitive to the assumption of the acquisition cost of the biosimilar drug and to the initial number of patients treated with biological therapy. The study focused on one indication (RA) and demonstrated that the introduction of biosimilar infliximab can lead to substantial budget savings in health care budgets. Further savings are expected for other indications where biosimilar medicines are implemented.

Similar content being viewed by others
References
Sokka, T., Kautiainen, H., Pincus, T., Verstappen, S.M., Aggarwal, A., Alten, R., Andersone, D., Badsha, H., Baecklund, E., Belmonte, M., Craig-Muller, J., da Mota, L.M., Dimic, A., Fathi, N.A., Ferraccioli, G., Fukuda, W., Geher, P., Gogus, F., Hajjaj-Hassouni, N., Hamoud, H., Haugeberg, G., Henrohn, D., Horslev-Petersen, K., Ionescu, R., Karateew, D., Kuuse, R., Laurindo, I.M., Lazovskis, J., Luukkainen, R., Mofti, A., Murphy, E., Nakajima, A., Oyoo, O., Pandya, S.C., Pohl, C., Predeteanu, D., Rexhepi, M., Rexhepi, S., Sharma, B., Shono, E., Sibilia, J., Sierakowski, S., Skopouli, F.N., Stropuviene, S., Toloza, S., Valter, I., Woolf, A., Yamanaka, H.: Work disability remains a major problem in rheumatoid arthritis in the 2000s: data from 32 countries in the QUEST-RA study. Arthritis Res. Ther. 12(2), R42 (2010). doi:10.1186/ar2951
Laires, P.A., Exposto, F., Mesquita, R., Martins, A.P., Cunha-Miranda, L., Fonseca, J.E.: Patients’ access to biologics in rheumatoid arthritis: a comparison between Portugal and other European countries. Eur. J. Health Econ. 14(6), 875–885 (2013). doi:10.1007/s10198-012-0432-5
Mauskopf, J.A., Sullivan, S.D., Annemans, L., Caro, J., Mullins, C.D., Nuijten, M., Orlewska, E., Watkins, J., Trueman, P.: Principles of good practice for budget impact analysis: report of the ISPOR Task Force on good research practices—budget impact analysis. Value Health 10(5), 336–347 (2007)
Orlewska, E., Gulacsi, L.: Budget-impact analyses: a critical review of published studies. PharmacoEconomics 27(10), 807–827 (2009)
van de Vooren, K., Duranti, S., Curto, A., Garattini, L.: A critical systematic review of budget impact analyses on drugs in the EU countries. Appl. Health Econ. Health Policy 12(1), 33–40 (2014). doi:10.1007/s40258-013-0064-7
Launois, R., Payet, S., Saidenberg-Kermanac’h, N., Francesconi, C., Franca, L.R., Boissier, M.C.: Budget impact model of rituximab after failure of one or more TNFalpha inhibitor therapies in the treatment of rheumatoid arthritis. Joint Bone Spine 75(6), 688–695 (2008). doi:10.1016/j.jbspin.2008.04.012
Sorensen, J., Andersen, L.S.: The case of tumour necrosis factor-alpha inhibitors in the treatment of rheumatoid arthritis: a budget impact analysis. PharmacoEconomics 23(3), 289–298 (2005)
Tóthfalusi, L., Endrényi, L., Chow, S.-C.: Statistical and regulatory considerations in assessments of interchangeability of biological drug products. Eur. J. Health Econ. (2014). doi:10.1007/s10198-014-0589-1
Péntek, M., Poór, G., Wiland, P., Martina, O., Brzosko, M., Codrenau, C., Brodszky, N., Gulácsi, L.: Biological therapy in inflammatory rheumatic diseases: issues in Central and Eastern European countries. Eur. J. Health Econ. (2014). doi:10.1007/s10198-014-0592-6
Laki, J., Székelyné Mónok, G.: Biological treatments—analysis by the NHIFA. http://www.oep.hu/pls/portal/url/ITEM/C97D04DF65C8B4CEE040A8C0CB324B94. (2012)
Koncz, T., Pentek, M., Brodszky, V., Érsek, K., Orlevska, E., Gulacsi, L.: Adherence to biologic DMARD therapies in rheumatoid arthritis. Expert Opin. Biol. Ther. 9, 1367–1378 (2010)
Laki, J., Monok, G., Palosi, M., Gajdacsi, J.Z.: Economical aspect of biological therapy in inflammatory conditions in Hungary. Expert Opin. Biol. Ther. 13(3), 327–337 (2013). doi:10.1517/14712598.2013.735654
Acknowledgments
The study was supported by an unrestricted grant from EGIS Pharmaceuticals and Center for Public Affairs Studies Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brodszky, V., Baji, P., Balogh, O. et al. Budget impact analysis of biosimilar infliximab (CT-P13) for the treatment of rheumatoid arthritis in six Central and Eastern European countries. Eur J Health Econ 15 (Suppl 1), 65–71 (2014). https://doi.org/10.1007/s10198-014-0595-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10198-014-0595-3