Abstract
Incineration is one of the key technologies in disposal of municipal waste, which produces municipal solid waste incineration (MSWI) residues with high valuable metal contents. The recycling strategy for the MSWI residues is typically focused on the recovery of scrap metals yielding processed municipal solid waste incineration residues (PIR) as the main byproduct. However, the PIR still contains valuable metals, particularly gold, which cannot be extracted by conventional methods. Here, we evaluated the feasibility of using the 0.5–2.0 mm grain size fraction of PIR containing 28.82 ± 1.62 mg/kg of gold as raw material for a two-stage extraction process. In the first stage the alkalic fine-grained PIR was acidified with a solution of 20% (v/v) of HCl-containing flue gas cleaning liquid that is obtained by the municipal waste incineration plant itself as a waste product. In the second stage we leached the acidified fine-grained PIR by thiourea with Fe3+ as an oxidant. Application of the thiourea-Fe3+ leaching system resulted in recovery of 16.4 ± 1.56 mg/kg of gold from the fine-grained PIR within 6 h of incubation. Due to high gold market prices, upscaling of the suggested technology can represent a suitable strategy for gold recovery from PIR and other MSWI residues.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Municipal solid waste incineration (MSWI) is one of the most common technologies for municipal solid waste (MSW) treatment [1,2,3,4]. In 2015–2016, there were 512 MSWI plants operating in Europe. These plants burned a total of 80–93 million tons of MSW per year (ca. 27% of the total annual amount of MSW) [2, 5, 6]. This amount of waste was converted to approximately 20 million tons of MSWI residues through incineration, which is about 20–25% of the weight of the input MSW [3, 6]. Reduction of weight leads to a concentration of scrap metals in the incinerated material [7]. Therefore, MSWI residues are commonly used as a secondary source of ferrous (Fe) and non-ferrous (NFe) metals [3, 8]. The amount of scrap in the MSWI residues reaches 7–15% for the Fe fraction and 1–5% for the NFe fraction [6, 9, 10]. The main amount of scrap is extracted with magnets and eddy current separators [3]. These techniques are efficient for the recovery of metals from MSWI residues with particle sizes larger than 2 mm [11]. However, due to disposal of electronic products, the fine-grained fractions of MSWI might be enriched with valuable elements [12,13,14,15,16]. Nevertheless, the finer particles are often separated from MSWI residues and directly mixed with material left after recycling, forming processed municipal solid waste incineration residues (PIR). One of the metals lost in the fine PIR fraction is gold (Au), which stems from fine electronics, such as soldering of electronic circuit boards [17]. The content of Au in MSWI varies in the range from 0.1 to 21.7 g/ton of MSWI residues [12, 13, 16,17,18,19,20,21]. This range corresponds to the Au content in the ore of the world's largest Au mines (1.1 g per ton of material on average) [22,23,24]. Therefore, the MSWI residues can be considered as prospective Au source. However, potentially recoverable separate pieces of Au (e.g., jewelry, coins, etc.) or composite materials (e.g., Au plated stainless steel wristbands of watches) are mainly present in the coarse-grained fraction [21]. The recovery of Au from fine-grained fraction can be performed using traditional pyrometallurgical and hydrometallurgical processes. However, these processes have several limitations. Pyrometallurgical processes (e.g., smelting) require high financial investments for energy and generate hazardous emissions [25]. The density separation cannot be easily adapted for the treatment of MSWI residues due to the presence of reactive substances such as calcium oxide (CaO), metal chlorides, and sulfates. These substances enable hardening and cementation and therefore facilitate mineral incrustation on metal and melt particles that are detrimental to the success of separation [3, 11, 26]. The use of cyanide leaching near populated areas is of widespread concern due to a series of accidents at various gold mines around the world [27].
Steadily rising Au market prices result in a strong motivation for considering the fine-grained fraction of MSWI as a potential source for Au recovery [28, 29]. Although Au is extremely valuable and critical for many industries (e.g., jewelry, electronics, computers, medical, and aerospace), its outstanding chemical resistance becomes a disadvantage in hydrometallurgical processes [30]. The organic compound thiourea (CS(NH2)2) is a promising Au complexing agent [30,31,32]. Thiourea leaching of Au is typically performed at low pH (1–3) and redox potential (Eh) 400–450 mV (vs SHE) [33]. The process requires an oxidizing agent to form the reactive compound from the thiourea [34, 35]. Ferric ion in sulfuric acid solution can serve as one of the most effective oxidizing agents for Au leaching [36]. Specifically, in acidic solutions, the oxidation of thiourea by Fe3+ leads to the formation of formamidine disulfide (Eq. 1). Subsequently, Au is oxidized by formamidine and forms a cationic Au thiourea complex (Eq. 2) [34, 36].
Thus, formamidine acts as an oxidant and as a complexing agent. Application of thiourea for Au extraction from several materials such as domestic ore, chalcopyrite ore, crushed electronic waste, and ceramic wastes allows for the extraction of up to 90% of Au at optimal conditions (low pH, presence of an oxidant) [34, 37,38,39]. However, unlike most typically used Au-containing materials, MSWI residues are often characterized by an alkaline pH of 11–13. The increase of pH occurs as a result of hydration of MSWI residues via lime (Ca(OH)2) formation [40,41,42]. Therefore, application of the thiourea-Fe3+ system for Au leaching requires acidification of MSWI residues [34].
The goal of this study was to test the capability of Au extraction from PIR. Our previous work demonstrated that the 0.5–2.0 mm grain size fraction reached 30.2% of the total MSWI residues weight and contained 28.82 ± 1.62 mg of Au per kg. Furthermore, this fraction was characterized by less buffering capacity compared to the finer fraction (< 0.5 mm) and therefore required less acid consumption for neutralization [16]. Thus, we sampled PIR from a regional processing plant, extracted the fine-grained fraction (0.5–2.0 mm grain size) and acidified it with flue gas cleaning liquid produced by the MSWI plant. The produced material was then used for extraction of Au by the thiourea-Fe3+ system.
Materials and methods
Source of PIR and industrial acid
The PIR was provided by a recycling plant that processes regional MSWI residues (bottom ash) from the southwestern areas of Germany (States of Hessen and Baden-Württemberg) (Fig. 1a). The plant processes this waste by ageing, sieving, crushing, magnetic recycling and eddy current separation to recover Fe and NFe metal scrap. After recycling, the remaining PIR is either disposed in landfills or used further as construction material. The PIR used in our study had been stored in piles for approximately three months before and approximately three months after the recycling process (from late spring until late autumn) at an open site, exposed to air and precipitation. The PIR consisted of quartz (SiO2), hematite (Fe2O3), calcite (CaCO3), magnetite (Fe3O4), anhydrite (CaSO4), akermanite (Ca2Mg(Si2O7)) and gehlenite (Ca2Al2SiO7) as it was shown in a previous study [16]. Approximately 100 kg of bulk samples were taken from different depths of the pile (1–2 m) and kept in five sealed plastic buckets in the laboratory at room temperature. Before Au leaching, one kg of PIR from each of the five buckets was mixed in a plastic container. The mixed PIR was air-dried for three days, and sieved into fractions of < 0.5 mm, 0.5–2.0 mm, and > 2 mm with an analytical vibration sieving instrument (Vibratory Sieve Shaker Analysette 3, Fritsch GmbH). The fine-grained fraction (0.5–2.0 mm) was stored at room temperature before acidification and leaching.
The pH value of the PIR was determined using a benchtop pH-meter (inoLab pH 7110, WTW GmbH) equipped with a Mettler-Toledo electrode (InLab Easy DIN). A suspension of 10 g of the PIR in 25 mL of Milli-Q H2O was shaken manually several times and the pH was directly determined in the settling suspension after one hour, 24 h and one week.
Flue gas cleaning liquid (industrial acid; IA) was provided by the waste-to-energy incineration plant (Fig. 1a). It was produced by purification of flue gas from chlorine in wet scrubbers. The temporal variation in concentrations of the key anions (Cl−, SO42−, Br−, I−, NO3− and F−) in the IA was monitored by the waste-to-energy incineration plant using ion chromatography (Table 1).
Impact of industrial acid on pH in suspensions of fine-grained PIR
To select the appropriate dilution for the addition of IA to the PIR, we prepared solutions of 5, 10, 20, 25 and 30% (v/v) of IA in Milli-Q H2O for acidification of the fine-grained size fraction of PIR (0.5–2.0 mm). Duplicate IA solutions (100 mL for each of five dilutions) were used for acidification of 30 g of PIR (solid:liquid ratio (w:w) ca. 1:3) for 12 days in 200 mL bottles (oxically; non-shaken).
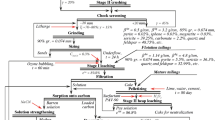
Two-stage Au-leaching process
Stage 1. Acidification of fine-grained PIR
One kg of fine-grained PIR was acidified with 3 L of IA solution (solid:liquid ratio (w:w) 1:3) diluted in Milli-Q H2O (20% v/v) (Fig. 1b). The treatment was done in duplicates in standing mode (non-shaken) in two plastic containers at room temperature. During incubation, we followed pH and sampled (in triplicates) the overlying IA solution for Au quantification hourly. To quantify the Au content, 3 mL of the solution was transferred into 15 mL centrifuge tubes and centrifuged for 10 min (7000 g). The supernatant was filtered with 0.2 µm syringe filters (Puradisc FP 30 CA, Whatman) and diluted in 10% Aqua Regia solution to maintain Au in the dissolved state. All samples were kept at 4 °C before Au quantification with inductively coupled plasma—optical emission spectrometry (ICP-OES). After 24 h of incubation, the IA solution was decanted completely. The remaining acidified PIR was centrifuged for 15 min (7000 g) to remove excess liquid. The PIR was dried at room temperature for 4 days and stored for further processing.
Stage 2. Mobilization of Au from fine-grained PIR with thiourea
After air-drying, 100 g of the fine-grained acid-treated PIR was transferred into eight glass beakers with magnetic stir bars (Fig. 1b). The acid-treated PIR was extracted with one of the following leaching solutions (Milli-Q H2O): i) 300 mL of 5% sulfuric acid (SA; v/v) containing 20 g/L thiourea and 10 g/L Fe2(SO4)3 × 7H2O; ii) 300 mL of 20% IA (v/v); iii) 300 mL of 5% SA (v/v); or iv) 300 mL of a mixture of 20% IA and 5% SA (v/v). The incubation of all setups was performed in duplicates each for six hours at room temperature under stirring (300 rpm). Samples for pH and Eh monitoring as well as leachate samples for Au quantification were taken hourly following the same protocol as described above. Eh was determined using a pH-meter (Mettler-Toledo; SG2) with Pt and Ag/AgCl electrodes.
The pregnant leaching solutions were removed from each of the eight beakers, the residual PIR was collected, centrifuged for 15 min (7000 g) to remove excess liquid, dried at room temperature for four days, and used for Au quantification following the protocol described below. All solid-phase samples were stored in a desiccator with silica gel at room temperature for further digestion and subsequent Au quantification.
Elemental analysis
Triplicates of air-dried samples of the fine-grained PIR as well as samples of leaching residuals were milled to a fine powder with a planetary mill (Fritsch Pulverisette, Fritsch GmbH). To minimize the risk of metal contamination, the milling buckets were cleaned with pure sand in a 10 min run (at grade 5–6), followed by an additional 10 min run with sample material to prime the milling buckets. The samples were then milled for 25 min. After milling, the powdered samples were dried at 105 °C for 48 h and stored in 50 mL polypropylene centrifuge tubes (Orange Scientific or SLG Süd-Laborbedarf Gauting) at room temperature in a desiccator dryer with silica gel.
Closed microwave digestion of powdered samples was performed in a microwave system (Multiwave Go Microwave Digestion System, Anton Paar). To this end, 0.25 g of samples were weighed in modified polytetrafluoroethylene (PTFE-TFM) digestion vessel and extracted with 0.5 mL of Milli-Q H2O, 3.6 mL of 37% HCl, and 1.2 mL of 65% HNO3. All samples were extracted in triplicates. The following temperature gradient parameters were applied in the microwave digestion process: step 1 (10 min, 180 °C), step 2 (20 min 200 °C), step 3 (30 min, 180 °C), ventilation (> 30 min). After cooling, the samples were subsequently filtered (filter paper, 619 G ¼, Macherey–Nagel) into 100 mL measuring flasks. Digestion vessels and the residues were rinsed several times with Milli-Q H2O to a final volume of 100 mL. Aqua regia digests were stored in polyethylene bottles at 4 °C until further analysis. Before analysis samples were filtered with 0.2 µm syringe filters (Puradisc FP 30 CA, Whatman). ICP-OES (SPECTROBLUE TI, Ametek) was used for quantification of Au in PIR digests as well as in diluted leachate samples (Online Resource 1).
Materials and solutions
All plasticware and glassware that were used for analytical work were pre-cleaned with 1 M HCl overnight and rinsed with Milli-Q H2O. The following reagents were used for leaching experiments and for subsequent analysis: 25% H2SO4 (for analysis; Merck); thiourea (99%, extra pure; Acros Organics); Fe2(SO4)3 7H2O (puris p.a. Sigma-Aldrich); 65% HNO3 (for analysis; Merck); 37% HCl (puris p.a.; Merck); Milli-Q H2O (18.2 MΩ·cm; Millipore); noble metals multi-element standard (VHG labs).
Results and discussion
Gold content in the fine-grained PIR size fraction
The PIR’s composition was heterogeneous even after recycling, as it still contained large pieces of scrap and NFe metals. The fine-grained size fraction (0.5–2.0 mm) of PIR was separated and weighed. It constituted 30.2% of the total PIR weight. In our previous study we demonstrated that the fine-grained fraction contained 28.82 ± 1.62 mg of Au per kg of PIR compared to an Au content of ca. 21.69 mg per kg in the raw PIR material [16]. Therefore, the 0.5–2.0 mm size fraction was selected to investigate Au extraction from PIR.
Acidification of fine-grained PIR size fraction with industrial acid
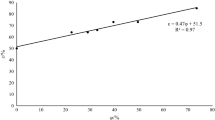
Storage of PIR in open-air waste piles contributes to weathering and capturing atmospheric CO2 which lowers the pH of the material via calcite (CaCO3) and ettringite (Ca6Al12(SO4)3(OH)12 × 26H2O) formation [41, 43,44,45]. However, the six-month storage period of the PIR did not lead to an appreciable pH decrease. The pH of the fine-grained fraction remained high (10.5 ± 0.5), indicating the presence of hydrated lime. Since the leaching efficiency of the thiourea-Fe3+ solution is determined by the formation of anionic complexes that dissolve Au, a strong ability of the leaching solution to decrease the pH value of the PIR is essential [38]. This can be achieved by treatment of PIR with acidic solutions, for example with industrial acid. Typically, the IA is disposed after neutralization and reduction of the heavy metal load [46]. However, due to the high HCl content (8–13% v/v; Table 1) it can be used instead for acidification of the fine-grained PIR. Therefore, we tested the capability of IA solutions (5, 10, 20, 25 and 30%; v/v) to decrease the pH of PIR suspension. Low pH values allow Fe3+ to oxidize thiourea accompanied by the formation of formamidine disulfide (Eq. 1), which in turn oxidizes Au forming a cationic Au thiourea complex [34, 36]. Triplicate IA solutions were mixed with fine-grained PIR and incubated oxically for 12 days. The pH of all PIR suspensions increased sharply during the first day of incubation (Fig. 2). However, the pH of the PIR suspensions containing 10, 20, 25 and 30% of IA (v/v) remained low and sufficient for oxidation thiourea by Fe3+. After 12 days of incubation, the pH values of the 5 and 10% solutions increased above 7 and 5, respectively, while the pH values of the 20, 25 and 30% solutions stabilized at a range of 2.9 to 3.8. These results demonstrated that treatment with 20–30% IA was sufficient to maintain pH values below 4 as required for effective Au extraction using Fe3+ as an oxidant [34, 36].
Based on these results we further acidified 1 kg of fine-grained PIR material with 3 L of 20% IA solution (Fig. 1b). Temporal pH monitoring of PIR suspension demonstrated that after six hours of incubation, the pH continued to increase but only slightly (Fig. 3). At this time, a sufficient portion of salts could be dissolved thereby reducing the alkaline capacity of PIR. The effect of PIR acidification on Au mobilization was also determined (Fig. 3). Quantification of dissolved Au demonstrated that 1.74 ± 0.4 mg of Au per 1 kg of fine-grained PIR were mobilized. This amount of Au was equivalent to 5.9 ± 0.5% of the total Au content in the fine-grained PIR (28.82 mg/kg) [16]. The mobilization of Au by 20% IA solution was supposedly caused by the peculiarities of IA composition (Table 1). Specifically, the HCl and HNO3 could partially digest and dissolve the bulk PIR material, while lixivalents (e.g., bromide and chloride) could potentially have complexed and mobilized the Au [30, 31, 47]. Chloride and bromide are typically applied together for Au leaching. Thus, several studies demonstrated earlier that Au can be complexed by chloride (120–210 g/L) and bromide (23.7–102.7 g/L) at atmospheric pressure [48, 49]. The 20% IA solution contained lower concentrations of both chloride (16–23 g/L) and bromide (0.2–0.56 g/L). However, they could still partially mobilize Au, while concentrations of iodide was not sufficient for complexation of Au [50, 51].
Leaching of Au from acid-treated fine-grained PIR using the thiourea-Fe3+ extraction system
At the second step of Au recovery, 300 mg of fine-grained acid-treated PIR was resuspended in 100 mL of thiourea in 5% sulfuric acid (SA) with Fe3+ as an oxidant. Leaching was accompanied by mixing of the PIR suspension to improve extraction efficiency. Furthermore, to reveal the contribution of Au complexation by thiourea, we additionally incubated the fine-grained fraction of the PIR in 20% IA solution without the thiourea- Fe3+ system. Additionally, to monitor the impact of low pH value on Au recovery, the fine-grained fraction of the PIR was incubated in 5% SA solution, and in a solution of 20% IA and 5% SA (Fig. 1b). The pH and Eh values during incubation were clearly dependent on the concentration of acids in the leaching solution. Thus, the incubation of fine-grained PIR in the solution containing 20% IA and 5% SA for six hours led to a minor change of the pH and Eh values from 0.01 to 0.27 and from + 406 to + 387 mV, respectively (Fig. 4a, b). Incubation of fine-grained PIR material in 20% IA resulted in more noticeable changes of pH and Eh. During six hours of incubation, the Eh decreased from + 395 to + 256 mV while the pH increased from 0.12 to 2.51. The application of thiourea in 5% SA maintained the pH and Eh values comparatively stable during incubation, i.e., the Eh value decreased slightly from + 393 to + 344 mV while the pH increased from 0.24 to 0.99. Similar values were observed for incubation in 5% SA, where the pH value increased from 0.20 to 0.87 while the Eh value decreased from + 398 to + 369 mV. In general, these results correlated well with the total acid content where the values showed the largest changes with decreasing acid strength, i.e., IA (20%) + SA (5%) < thiourea + SA (5%) = SA (5%) < IA (20%), and thus the highest acid content maintained the lowest pH and highest Eh values.
Ferric iron oxidizes thiourea and leads to the formation of formamidine disulfide (Eq. 1), which in turn oxidizes Au and forms a cationic Au thiourea complex (Eq. 2) within a Eh range from + 350 mV to 450 mV (vs SHE). However, treatment of acidified fine-grained PIR with IA (20%) + SA (5%), thiourea + SA (5%) and SA (5%) maintained an Eh value from + 340 mV to + 390 mV (from + 539 mV to + 589 mV; vs SHE). Higher (more positive) redox potentials can lead to undesirable oxidative degradation of thiourea via formamidine disulfide (NH2(NH)CSSC(NH)NH2) formation, which can decompose into thiourea (CS(NH2)2), cyanamide (NH2CN) and elemental sulfur (S°), which covers the Au particles and prevents its mobilization [33]. Nevertheless, the application of thiourea in 5% SA led to a mobilization of 15.1 ± 0.7 mg of Au per kg of fine-grained PIR during the first two hours of leaching (5.02 ± 0.22 mg/L of Au in the leaching solution). After six hours of leaching, the Au concentration reached 5.46 ± 0.52 mg/L, corresponding to 16.4 ± 1.56 mg Au leached per kg of fine-grained PIR material (56.8 ± 5.4% of the total Au content; Fig. 3c). Under controlled leaching conditions, the solution of 20% IA contributed to the release of 5.3 ± 0.2 mg/kg of Au (18.4 ± 0.5% from the total Au content; Fig. 4c), while the application of a mixture of 20% IA and 5% SA led to the release of 9.1 ± 0.1 mg/kg of Au (31.5 ± 0.2% of the total Au content). These results were supposedly achieved due to digestion/dissolution of the PIR bulk material by the HCl acid and complexation of the released Au by the lixiviants (chloride and bromide) that were present in the IA (Table 1) [30, 31, 47]. Finally, the incubation of fine-grained PIR in 5% SA resulted in a release of 1.1 ± 0.9 mg/kg of Au (4.0 ± 3.1% of the total Au content) supposedly due to dissolution of chlorides, nitrates and other salts from the PIR bulk phase followed by Au complexation. The concentration of chloride in municipal waste incineration residues can reach 1–3 wt% [52, 53] and most of the chloride in the MSWI residues is present in the fine-grained fraction [54]. The total Au contents consisting of mobilized Au and Au left in the PIR in all treatments were equivalent to 30–32 mg/kg (Fig. 1c). These concentrations were comparable to the concentration of Au determined in the original untreated fine-grained PIR (28.82 ± 1.62 mg/kg) [16]. Therefore, the application of thiourea-Fe3+ system as well as other leaching agents did not lead to a complete Au extraction from PIR and more than 40% of the Au was still left in the remaining PIR material.
Potential techniques of Au recovery from thiourea-Fe3+ leaching solution
Our results have shown that the efficiency of Au mobilization by the thiourea-Fe3+ system from acid-treated fine-grained PIR was the highest among the tested treatments. The next step after the mobilization of Au is its recovery from the pregnant leaching solution. However, the techniques of Au recovery from dissolved state are not well-established so far. Conventional methods such as the Merill-Crowe process wherein Au is recovered from solution by cementation with zinc powder or methods that involve the use of activated carbon followed by electrolysis were developed for the Au cyanidation approach [39, 55,56,57,58]. Several studies demonstrated that Au can be recovered from thiourea leaching solution by adsorption on activated carbon [59,60,61] and organic materials (e.g., persimmon tannin based sorbents, rice husk or chitin) [61,62,63,64] or via ion exchange [65, 66]. The adsorption capacities of activated carbon, rice husk and chitin are relatively high and allow to recover ca. 36 mg/g, 28 mg/g and 58 mg/g of Au, respectively, from the thiourea leaching solution [61, 64]. These methods might be used for recovery of Au from leachate produced by treatment of PIR, since the application of IA for acidification of PIR can also reduce the amount of soluble metals (particularly Fe and Cu) thereby reducing the competing adsorption.
The selective recovery of Au from thiourea leaching solution might be achieved by electrowinning [67]. However, the efficient electrowinning requires leachate with high Au concentration [67, 68]. This requirement, therefore, does not allow considering electrowinning for Au extraction from PIR leachate. The precipitation of Au from thiourea solution by H2 reduction is another method that has been successfully applied. However, this process is carried out at high pressure and temperature and requires the application of a catalyst [69]. Nevertheless, the precipitation can be also considered as a potential method for recovery of Au from PIR leachate. For example, the precipitation of Au by sodium borohydride in acidic solution of thiourea may facilitate the selective reduction of Au and even allows to recycle the thiourea [70]. Finally, cementation can be used for recovery of Au. Several metal species have been shown to be useful cementing agents for Au from thiourea solutions including Cu, Zn, Ni and Al [71,72,73]. However, the Fe3+ used as oxidizing agent for leaching of Au from PIR can significantly affect the cementation reaction kinetics via increasing of redox potential of the solution. Nevertheless, the high redox potential can be lowered by supplementation of the cementation reaction with sodium citrate which can form Fe3+-citrate complexes [73].
In summary, the recovery of Au from the thiourea complex might be performed by different techniques (e.g., adsorption, precipitation, cementation, and electrowinning). However, low Au and high Fe3+ contents of the PIR leachate may decrease the efficiency of the extraction process. Therefore, the acidification of PIR with IA can help to decrease concentration of metals that can be mobilized during thiourea-Fe3+ leaching of Au. In turn, the lower concentration of dissolved metals (primarily Fe and Cu) can potentially increase the efficiency of Au extraction by adsorption, via reducing the competing adsorption, or by cementation via reducing the redox potential of the solution. In addition, the application of sodium citrate may help to further neutralize the effect of residual Fe3+ on Au recovery from the leaching solution.
Use of secondary materials for Au leaching
Approximately 2551 tons of Au (63.44% of global Au demand) were used for production of jewelry and electronics in 2021 [74]. Increasing production of Au-containing goods will inevitably lead to increased amounts of Au in the various types of waste (e.g., sweeping jewelry, electronic wastes, slags, etc.) [12, 13, 25, 75,76,77,78,79,80,81,82]. Therefore, over the past decades there have been several attempts to recover Au from secondary materials through leaching techniques using aqua regia, cyanide, ionic liquids, thiosulfate, or thiourea as lixiviants [27, 30, 68]. Among these commonly used lixiviants, thiourea is relatively effective in Au mobilization whilst having the least negative impacts on the environment [68, 83, 84]. Indeed, the application of different thiourea-based leaching systems is usually accompanied by a high yield of mobilized Au. Thus more than 80% of Au can be mobilized by thiourea leaching from such wastes as activated carbon (2.9 µg/g of Au) [85], wastes of amalgamation and cyaniding processes (1.7–8.5 µg/g of Au) [86], and Cu smelting slag (0.44–0.46 µg/g of Au) [87]. Furthermore, thiourea-based leaching systems are often used for extraction of Au from electronic wastes [14, 68, 76, 83, 84, 88, 89]. For example, more than 90% of Au can be leached from dust generated during processing of electronic waste (141 µg/g of Au) or printed circuit boards (PCBs) (43 µg/g of Au) [88, 89].
The high efficiency of Au recovery by thiourea-based leaching often requires the pretreatment of secondary materials. In particularly, the size reduction [62, 90], the removal of impurities (plastics, metal peace, etc.) [91, 92] or pre-leaching of other metals included in the composition of the Au-containing secondary material [93] help to increase the efficiency of the following Au-leaching process. Pre-leaching of Fe and Cu is especially important for Au mobilization by thiourea since the dissolution of these metals increases the redox potential of acidic leachate thereby oxidizing thiourea and forming formamidine disulfide [90, 94,95,96]. Different inorganic acids HCl, HNO3 and H2SO4 with or without additional oxidants (e.g., O2, H2O2), organic acids (e.g., citric acid, oxalic acid and acetic acid), and other lixiviants (e.g., EDTA, sodium acetate and sodium hydroxide) can be used for the recovery of Fe and Cu from Au-containing secondary materials [68, 97, 98]. These methods have consistently demonstrated a high efficiency of Fe and Cu leaching and recovery. For example, fly ash from waste incineration plants (L/S ratio—20) that was treated with 5–7 M HCl extracted ca. 80% Fe and 86% of Cu [97]. The ammonia (5 M)/ammonium sulphate (2 M) leaching system supplemented by oxidants (O2 or H2O2) can extract up to 91% of Cu from PCBs (L/S ratio—20) [99]. EDTA (4 M / 1 M of total heavy metals content) can facilitate the extraction of Cu up to 88% from electrostatic precipitator ash from an incineration plant (L/S ratio—10) [100], while 0.02 M ammonium citrate promoted the mobilization of Cu and Fe from MSWI bottom ash (L/S ratio—10) [101]. Copper can also be volatilized from the MSW fly ash at 700–900 °C in the presence of poly(vinyl chloride). This method enables the recovery of ca. 50% of Cu [4]. In addition, the efficiency of Fe and Cu recovery can be improved with the application of microwave radiation [87]. Bioleaching can be also considered as a promising method for pretreatment of secondary materials. Thus, Acidithiobacillus ferrooxidans in association with A. thiooxidans [102] or alone [103] can be used for leching of Cu from PCBs. In both cases, the efficiency of Cu recovery exceeds 90%.
When considering the existing applications of thiourea-based leaching as well as the rising price of Au (ca. 57,454 USD per 1 kg in January 2020) [104], Au recovery from fine-grained PIR using the suggested two-step thiourea-Fe3+ extraction procedure could represent a new high-profit niche for the MSWI residue recycling industry. The high content of Au in the PIR (28.82 ± 1.62 mg/kg) and the high extraction efficiency (more than 50%) allows us to consider the fine-grained PIR fraction as a valuable urban ore comparable in Au content with other types of secondary materials [27, 105, 106]. Upscaling of laboratory technology is a complex process that can lead to losses in Au-leaching efficiency. However, if the efficiency of Au extraction remains at the level of 50% thiourea-Fe3+, extraction procedures will mobilize up to 14 g of Au per 1 ton of PIR. This amount of Au is equivalent to 804.5 USD according to the current price [104]. Further research is needed to assess the operating costs of this technology.
Conclusions
The sufficient part of MSW (ca. 27% in Europe) is currently burned in incineration plants [2, 5, 6]. The recovery of valuable metals (e.g., Au) from MSWI residues (e.g., PIR) by conventional methods including ageing, sieving, crushing, magnetic separation, density separation and eddy current separation have several limitations due to the relatively low abundance of precious metals and the high structural and compositional complexity of the incineration waste matrix [25]. Therefore, there is a strong need to develop and apply low-cost and eco-friendly methods to recover precious metals. This study presents a two-stage leaching process for Au from fine-grained PIR material. Although this fine-grained fraction was characterized as a highly alkaline material, it was efficiently acidified by a pretreatment with 20% IA, resulting in the extraction of up to 6% of the total Au content. The second extraction step, using a thiourea-Fe3+ leaching system, led to the recovery of a large fraction of Au from PIR material (in total 56.8 ± 5.41% of total Au content) during only six hours of incubation. Due to the high Au price and the rising need for Au in electronics, we suggest that upscaling of the thiourea-Fe3+ leaching approach can be applied for treatment of the fine-grained fraction of PIR and Au extraction. Furthermore, this approach has the potential to be not only cost-effective but also environmentally friendly in comparison with cyanide leaching, which is highly toxic. Therefore, we can assume that recovery of Au with thiourea-Fe3+ system has the potential to become an integral part of sustainable waste management in the future.
Change history
03 October 2022
Missing Open Access funding information has been added in the Funding Note.
References
Malinauskaite J, Jouhara H, Czajczyńska D, Stanchev P, Katsou E, Rostkowski P, Thorne RJ, Colón J, Ponsá S, Al-Mansour F, Anguilano L, Krzyżyńska R, López IC, Vlasopoulos A, Spencer N (2017) Municipal solid waste management and waste-to-energy in the context of a circular economy and energy recycling in Europe. Energy 141:2013–2044. https://doi.org/10.1016/j.energy.2017.11.128
Scarlat N, Fahl F, Dallemand J-F (2019) Status and opportunities for energy recovery from municipal solid waste in Europe. Waste Biomass Valor 10(9):2425–2444. https://doi.org/10.1007/s12649-018-0297-7
Šyc M, Simon FG, Hykš J, Braga R, Biganzoli L, Costa G, Funari V, Grosso M (2020) Metal recovery from incineration bottom ash: state-of-the-art and recent developments. J Hazard Mater 393:122433. https://doi.org/10.1016/j.jhazmat.2020.122433
Kurashima K, Kumagai S, Kameda T, Saito Y, Yoshioka T (2020) Heavy metal removal from municipal solid waste fly ash through chloride volatilization using poly(vinyl chloride) as chlorinating agent. J Mater Cycles Waste Manag 22(4):1270–1283. https://doi.org/10.1007/s10163-020-01021-6
Lamers F (2015) Treatment of bottom ashes of waste-to-energy installations – state of the art. In: Thomé-Kozmiensky KJTS (ed) Waste management. TK-Verlag, Neuruppin, pp 271–290
Blasenbauer D, Huber F, Lederer J, Quina MJ, Blanc-Biscarat D, Bogush A, Bontempi E, Blondeau J, Chimenos JM, Dahlbo H, Fagerqvist J, Giro-Paloma J, Hjelmar O, Hyks J, Keaney J, Lupsea-Toader M, O’Caollai CJ, Orupõld K, Pająk T, Simon F-G, Svecova L, Šyc M, Ulvang R, Vaajasaari K, Van Caneghem J, van Zomeren A, Vasarevičius S, Wégner K, Fellner J (2020) Legal situation and current practice of waste incineration bottom ash utilisation in Europe. Waste Manag 102:868–883. https://doi.org/10.1016/j.wasman.2019.11.031
Allegrini E, Vadenbo C, Boldrin A, Astrup TF (2015) Life cycle assessment of resource recovery from municipal solid waste incineration bottom ash. J Environ Manag 151:132–143. https://doi.org/10.1016/j.jenvman.2014.11.032
Van Caneghem J, De Coster E, Vandenbergh K, De Broyer S, Lambrix N, Weemaels L (2019) Closing the household metal packaging cycle through recovery of scrap from waste-to-energy bottom ash: the case study of Flanders. Resour Conserv Recycl 144:115–122. https://doi.org/10.1016/j.resconrec.2019.01.028
Sabbas T, Polettini A, Pomi R, Astrup T, Hjelmar O, Mostbauer P, Cappai G, Magel G, Salhofer S, Speiser C, Heuss-Assbichler S, Klein R, Lechner P (2003) Management of municipal solid waste incineration residues. Waste Manage 23(1):61–88. https://doi.org/10.1016/S0956-053X(02)00161-7
Grosso M, Biganzoli L, Rigamonti L (2011) A quantitative estimate of potential aluminium recovery from incineration bottom ashes. Resour Conserv Recycl 55(12):1178–1184. https://doi.org/10.1016/j.resconrec.2011.08.001
Holm O, Simon FG (2017) Innovative treatment trains of bottom ash (BA) from municipal solid waste incineration (MSWI) in Germany. Waste Manag 59:229–236. https://doi.org/10.1016/j.wasman.2016.09.004
Morf LS, Gloor R, Haag O, Haupt M, Skutan S, Lorenzo FD, Böni D (2013) Precious metals and rare earth elements in municipal solid waste – sources and fate in a Swiss incineration plant. Waste Manag 33(3):634–644. https://doi.org/10.1016/j.wasman.2012.09.010
Allegrini E, Maresca A, Olsson ME, Holtze MS, Boldrin A, Astrup TF (2014) Quantification of the resource recovery potential of municipal solid waste incineration bottom ashes. Waste Manag 34(9):1627–1636. https://doi.org/10.1016/j.wasman.2014.05.003
Khaliq A, Rhamdhani MA, Brooks G, Masood S (2014) Metal extraction processes for electronic waste and existing industrial routes: a review and Australian perspective. Resources 3(1):152–179. https://doi.org/10.3390/resources3010152
Funari V, Bokhari SNH, Meisel T, Vigliotti L, Braga R (2014) The REE potential in “urban” ore deposits: and evaluation and prospecting tools from Italian municipal solid waste incinerators (ERES). Proceedings of the 1st International Conference on European Rare Earth Resources. Milos (Greece). 468–475.
Abramov S, He J, Wimmer D, Lemloh M-L, Muehe EM, Gann B, Roehm E, Kirchhof R, Babechuk MG, Schoenberg R, Thorwarth H, Helle T, Kappler A (2018) Heavy metal mobility and valuable contents of processed municipal solid waste incineration residues from Southwestern Germany. Waste Manag 79:735–743. https://doi.org/10.1016/j.wasman.2018.08.010
Muchova L, Bakker E, Rem P (2009) Precious metals in municipal solid waste incineration bottom ash. Water Air Soil Pollut Focus 9(1):107–116. https://doi.org/10.1007/s11267-008-9191-9
Buchholz BA, Landsberger S (1995) Leaching dynamics studies of municipal solid waste incinerator ash. J Air Waste Manag Assoc 45(8):579–590. https://doi.org/10.1080/10473289.1995.10467388
Kida A, Noma Y, Imada T (1996) Chemical speciation and leaching properties of elements in municipal incinerator ashes. Waste Manag 16(5):527–536. https://doi.org/10.1016/S0956-053X(96)00094-3
Fujimori E, Minamoto K, Haraguchi H (2005) Comparative study on the distributions of precious metals (Ru, Rh, Pd, Ir, Pt, and Au) in industrial waste incineration ashes as determined by tellurium coprecipitation and ICP-MS. Bull Chem Soc Jpn 78(11):1963–1969. https://doi.org/10.1246/bcsj.78.1963
Bunge R (2015) Recovery of metals from waste incinerator bottom ash. Switzerland, Institute fur Umwelt und Verfahrenstechnik UMTEC.
Domínguez A, Valero A (2013) Global gold mining: is technological learning overcoming the declining in ore grades? J Environ Account Manag 1:85–101. https://doi.org/10.5890/JEAM.2012.01.007
Prichard HM, Wedin F, Sampson J, Jackson MT, Fisher PC (2016) Precious metals in urban waste. Water Environ J 30(1–2):151–156. https://doi.org/10.1111/wej.12166
Bilston DW, La Brooy SR, Woodcock JT (1984) Gold and silver leaching from an oxidized gold ore with thiourea under controlled conditions. Symposium on Extractive Metallurgy. Melbourne, 51–60
He J, Kappler A (2017) Recovery of precious metals from waste streams. Microb Biotechnol 10(5):1194–1198. https://doi.org/10.1111/1751-7915.12759
Simon F-G, Holm O (2017) Exergetische Bewertung von Rohstoffen am Beispiel von Kupfer. Chem Ing Tec 89(1–2):108–116. https://doi.org/10.1002/cite.201600089
Cui J, Zhang L (2008) Metallurgical recovery of metals from electronic waste: a review. J Hazard Mater 158(2–3):228–256. https://doi.org/10.1016/j.jhazmat.2008.02.001
Shafiee S, Topal E (2010) An overview of global gold market and gold price forecasting. Resour Policy 35(3):178–189. https://doi.org/10.1016/j.resourpol.2010.05.004
Reig M, Vecino X, López J, Gibert O, Valderrama C, Cortina JL (2019) Metal recovery by ion-exchange resins from municipal incinerated bottom ash. 3rd IWA Resource Recovery Conference: IWA RR2019. Venice (Italy). 462–463.
Gökelma M, Birich A, Stopic S, Friedrich B (2016) A review on alternative gold recovery re-agents to cyanide. J Mater Sci Chem Eng 4:8–17. https://doi.org/10.4236/msce.2016.48002
Birich A, Stopic S, Friedrich B (2019) Kinetic investigation and dissolution behavior of cyanide alternative gold leaching reagents. Sci Rep 9(1):7191. https://doi.org/10.1038/s41598-019-43383-4
Chang S, Halim S (2019) Recovery of precious metals from discarded mobile phones by thiourea leaching. Mater Sci Forum 962:112–116. https://doi.org/10.4028/www.scientific.net/MSF.962.112
Aylmore MG (2016) Chapter 27 - alternative lixiviants to cyanide for leaching gold ores. In: Adams MD (ed) Gold Ore Processing (Second Edition). Elsevier, pp 447–484
Li J, Miller JD (2006) A review of gold leaching in acid thiourea solutions. Miner Process Extr Metall Rev 27(3):177–214. https://doi.org/10.1080/08827500500339315
Zhang J, Shen S, Cheng Y, Lan H, Hu X, Wang F (2014) Dual lixiviant leaching process for extraction and recovery of gold from ores at room temperature. Hydrometallurgy 144–145:114–123. https://doi.org/10.1016/j.hydromet.2014.02.001
Syed S (2012) Recovery of gold from secondary sources—a review. Hydrometallurgy 115–116:30–51. https://doi.org/10.1016/j.hydromet.2011.12.012
Ubaldini S, Fornari P, Massidda R, Abbruzzese C (1998) An innovative thiourea gold leaching process. Hydrometallurgy 48(1):113–124. https://doi.org/10.1016/S0304-386X(97)00076-5
Ficeriová J, Baláz P, Dutková E, Gock E (2008) Leaching of gold and silver from crushed Au-Ag wastes. Open Chem Eng J 2(6):9. https://doi.org/10.2174/1874123100802010006
Rao MD, Singh KK, Morrison CA, Love JB (2020) Challenges and opportunities in the recovery of gold from electronic waste. RSC Adv 10(8):4300–4309. https://doi.org/10.1039/C9RA07607G
Chandler AJ, Eighmy T, Hjelmar O, Kosson D, Sawell S, Vehlow J, Van der Sloot H, Hartlén J (1997) Municipal solid waste incinerator residues. Elsevier, Amsterdam, p 974
Meima JA, Comans RNJ (1997) Geochemical modeling of weathering reactions in municipal solid waste incinerator bottom ash. Environ Sci Technol 31(5):1269–1276. https://doi.org/10.1021/es9603158
Rendek E, Ducom G, Germain P (2007) Influence of waste input and combustion technology on MSWI bottom ash quality. Waste Manag 27(10):1403–1407. https://doi.org/10.1016/j.wasman.2007.03.016
Forteza R, Far M, Seguı́ C, Cerdá V (2004) Characterization of bottom ash in municipal solid waste incinerators for its use in road base. Waste Manag 24(9):899–909. https://doi.org/10.1016/j.wasman.2004.07.004
Rendek E, Ducom G, Germain P (2006) Influence of organic matter on municipal solid waste incinerator bottom ash carbonation. Chemosphere 64(7):1212–1218. https://doi.org/10.1016/j.chemosphere.2005.11.053
Alam Q, Schollbach K, Rijnders M, van Hoek C, van der Laan S, Brouwers HJH (2019) The immobilization of potentially toxic elements due to incineration and weathering of bottom ash fines. J Hazard Mater 379:120798. https://doi.org/10.1016/j.jhazmat.2019.120798
Dziedzic K, Jurczyk M, Mikus M (2016) Flue gas cleaning in municipal waste-to-energy plants-part II. Infrastruct Ecol Rural Areas 4:1309–1321. https://doi.org/10.14597/infraeco.2016.4.2.096
Usher A, McPhail D, Brugger J (2009) A spectrophotometric study of aqueous Au(III) halide–hydroxide complexes at 25–80°C. Geochim Cosmochim Acta 73:3359–3380. https://doi.org/10.1016/j.gca.2009.01.036
Ahtiainen R, Lundström M (2019) Cyanide-free gold leaching in exceptionally mild chloride solutions. J Clean Prod 234:9–17. https://doi.org/10.1016/j.jclepro.2019.06.197
Sousa R, Futuro A, Fiúza A, Vila MC, Dinis ML (2018) Bromine leaching as an alternative method for gold dissolution. Miner Eng 118:16–23. https://doi.org/10.1016/j.mineng.2017.12.019
Baghalha M (2012) The leaching kinetics of an oxide gold ore with iodide/iodine solutions. Hydrometallurgy 113–114:42–50. https://doi.org/10.1016/j.hydromet.2011.11.013
Lucheva B, Iliev P, Kolev D (2017) Recovery of gold from electronic waste by iodine-iodide leaching. J Chem Technol Metall 52(2):326–332
Yang S, Saffarzadeh A, Shimaoka T, Kawano T (2013) Distribution of chloride in municipal solid waste incineration bottom ash. 3rd International Conference on Sustainable Construction Materials and Technologies - SCMT3. Kyoto, Japan
Ito R, Fujita T, Sadaki J, Matsumoto Y, Ahn J-W (2006) Removal of chloride in bottom ash from the industrial and municipal solid waste incinerators. Int J Soc Mater Eng Resour 13:70–74. https://doi.org/10.5188/ijsmer.13.70
Joseph AM, Snellings R, Van den Heede P, Matthys S, De Belie N (2018) The use of municipal solid waste incineration ash in various building materials: a Belgian point of view. Materials 11:141. https://doi.org/10.3390/ma11010141
Soleimani M, Kaghazchi T (2008) Gold recovery from loaded activated carbon using different solvents. J Chin Inst Chem Eng 39(1):9–11. https://doi.org/10.1016/j.jcice.2007.11.004
Tahli L, Wahyudi T (2017) Desorption of gold and silver from activated carbon. Indones Min J 20(1):39–47. https://doi.org/10.30556/imj.Vol20.No1.2017.184
Mpinga CN, Bradshaw SM, Akdogan G, Snyders CA, Eksteen JJ (2014) Evaluation of the Merrill-Crowe process for the simultaneous removal of platinum, palladium and gold from cyanide leach solutions. Hydrometallurgy 142:36–46. https://doi.org/10.1016/j.hydromet.2013.11.004
Naumov KD, Lobanov VG, Zelyakh YD (2017) Gold electrowinning from cyanide solutions using three-dimensional cathodes. Metallurgist 61(3):249–253. https://doi.org/10.1007/s11015-017-0485-x
Zhang H, Ritchie IM, La Brooy SR (2004) The adsorption of gold thiourea complex onto activated carbon. Hydrometallurgy 72(3):291–301. https://doi.org/10.1016/S0304-386X(03)00182-8
Masiya TT, Gudyanga FP (2009) Investigation of granular activated carbon from peach stones for gold adsorption in acidic thiourea. J South Afr Inst Min Metall 465–474
Nakbanpote W, Thiravetyan P, Kalambaheti C (2002) Comparison of gold adsorption by Chlorella vulgaris, rice husk and activated carbon. Miner Eng 15(7):549–552. https://doi.org/10.1016/S0892-6875(02)00064-X
Gurung M, Adhikari BB, Kawakita H, Ohto K, Inoue K, Alam S (2013) Recovery of gold and silver from spent mobile phones by means of acidothiourea leaching followed by adsorption using biosorbent prepared from persimmon tannin. Hydrometallurgy 133:84–93. https://doi.org/10.1016/j.hydromet.2012.12.003
Nakbanpote W, Thiravetyan P, Kalambaheti C (2000) Preconcentration of gold by rice husk ash. Miner Eng 13(4):391–400. https://doi.org/10.1016/S0892-6875(00)00021-2
Côrtes LN, Tanabe EH, Bertuol DA, Dotto GL (2015) Biosorption of gold from computer microprocessor leachate solutions using chitin. Waste Manag 45:272–279. https://doi.org/10.1016/j.wasman.2015.07.016
Stofkova M, Stofko M (2002) Ion exchange resin use for Au and Ag separation from diluted solutions of thiourea. Metalurgija 41(1):33–36
Conradie PJ, Johns MW, Fowles RJ (1995) Elution and electrowinning of gold from gold-selective strong-base resins. Hydrometallurgy 37(3):349–366. https://doi.org/10.1016/0304-386X(94)00032-X
Juarez CM, Dutra AJB (2000) Gold electrowinning from thiourea solutions. Miner Eng 13(10):1083–1096. https://doi.org/10.1016/S0892-6875(00)00092-3
Ray DA, Baniasadi M, Graves JE, Greenwood A, Farnaud S (2022) Thiourea leaching: an update on a sustainable approach for gold recovery from e-waste. J Sustain Metall. https://doi.org/10.1007/s40831-022-00499-8
Kachaniwsky G, Newman C (1987) Proceedings of the metallurgical society of the Canadian institute of mining and metallurgy. Elsevier Science & Technology Books, Winnipeg, Canada, Pergamon, pp 5–6
Awadalla FT, Ritcey GM (1991) Recovery of gold from thiourea, thiocyanate, or thiosulfate solutions by reduction-precipitation with a stabilized form of sodium borohydride. Sep Sci Technol 26(9):1207–1228. https://doi.org/10.1080/01496399108050525
Lee HY, Kim SG, Oh JK (1997) Cementation behavior of gold and silver onto Zn, Al, and Fe powders from acid thiourea solutions. Can Metall Q 36(3):149–155. https://doi.org/10.1016/S0008-4433(97)00004-9
Raudsepp R (1987) Cementation of gold onto atomized aluminium from acid thiourea solutions. In: Strathdee GL, Klein MO, Melis LA (eds) Crystallization and precipitation. Elsevier, Pergamon, pp 297–306
Wang Z, Li Y, Ye C (2011) The effect of tri-sodium citrate on the cementation of gold from ferric/thiourea solutions. Hydrometallurgy 110(1):128–132. https://doi.org/10.1016/j.hydromet.2011.08.011
Gold demand trends full year (2021) From https://www.gold.org/goldhub/research/gold-demand-trends/gold-demand-trends-full-year-2021. Retrieved 7 May 2022
Burat F, Demirağ A, Şafak MC (2020) Recovery of noble metals from floor sweeping jewelry waste by flotation-cyanide leaching. J Mater Cycles Waste Manag 22(3):907–915. https://doi.org/10.1007/s10163-020-00982-y
Abdelbasir SM, El-Sheltawy CT, Abdo DM (2018) Green processes for electronic waste recycling: a review. J Sustain Metall 4(2):295–311. https://doi.org/10.1007/s40831-018-0175-3
Tickner J, Rajarao R, Lovric B, Ganly B, Sahajwalla V (2016) Measurement of gold and other metals in electronic and automotive waste using gamma activation analysis. J Sustain Metall 2(4):296–303. https://doi.org/10.1007/s40831-016-0051-y
Dinç Nİ, Tosun AU, Baştürkcü E, Özer M, Burat F (2022) Recovery of valuable metals from WPCB fines by centrifugal gravity separation and froth flotation. J Mater Cycles Waste Manag 24(1):224–236. https://doi.org/10.1007/s10163-021-01310-8
Arshadi M, Yaghmaei S, Esmaeili A (2020) Evaluating the optimal digestion method and value distribution of precious metals from different waste printed circuit boards. J Mater Cycles Waste Manag 22(5):1690–1698. https://doi.org/10.1007/s10163-020-01043-0
Hosoda E (2007) International aspects of recycling of electrical and electronic equipment: material circulation in the East Asian region. J Mater Cycles Waste Manag 9(2):140–150. https://doi.org/10.1007/s10163-007-0179-8
Rudnik E, Handzlik PM, P, (2016) Ammoniacal leaching and recovery of copper from alloyed low-grade e-waste. J Mater Cycles Waste Manag 18(2):318–328. https://doi.org/10.1007/s10163-014-0335-x
Tunali M, Tunali MM, Yenigun O (2021) Characterization of different types of electronic waste: heavy metal, precious metal and rare earth element content by comparing different digestıon methods. J Mater Cycles Waste Manag 23(1):149–157. https://doi.org/10.1007/s10163-020-01108-0
Ponghiran W, Charoensaeng A, Khaodhiar S (2021) The environmental impact assessment of gold extraction processes for discarded computer RAM: a comparative study of two leaching chemicals. J Mater Cycles Waste Manag 23(4):1412–1422. https://doi.org/10.1007/s10163-021-01221-8
Barrueto Y, Hernández P, Jiménez Y, Morales J (2021) Leaching of metals from printed circuit boards using ionic liquids. J Mater Cycles Waste Manag 23(5):2028–2036. https://doi.org/10.1007/s10163-021-01275-8
Adams CR, Porter CP, Robshaw TJ, Bezzina JP, Shields VR, Hides A, Bruce R, Ogden MD (2020) An alternative to cyanide leaching of waste activated carbon ash for gold and silver recovery via synergistic dual-lixiviant treatment. J Ind Eng Chem 92:120–130. https://doi.org/10.1016/j.jiec.2020.08.031
Boboev IR, Kurbonov SK, Sel’nitsyn RS (2019) Use of thiourea leaching during gold-containing dump treatment. Metallurgist 63(6):82–87. https://doi.org/10.1007/s11015-019-00869-w
Shoshay Z, Sapinov RV, Sadenova MA, Varbanov PS (2021) Intensification of the process of extracting non-ferrous metals from kazakhstani technogenic raw materials. Chem Eng Trans 88:1069–1074. https://doi.org/10.3303/CET2188178
Lee H, Molstad E, Mishra B (2018) Recovery of gold and silver from secondary sources of electronic waste processing by thiourea leaching. JOM 70(8):1616–1621. https://doi.org/10.1007/s11837-018-2965-2
Jing-ying L, Xiu-li X, Wen-quan L (2012) Thiourea leaching gold and silver from the printed circuit boards of waste mobile phones. Waste Manag 32(6):1209–1212. https://doi.org/10.1016/j.wasman.2012.01.026
Birloaga I, De Michelis I, Ferella F, Buzatu M, Vegliò F (2013) Study on the influence of various factors in the hydrometallurgical processing of waste printed circuit boards for copper and gold recovery. Waste Manag 33(4):935–941. https://doi.org/10.1016/j.wasman.2013.01.003
Tripathi A, Kumar M, Sau D, Agrawal A, Chakravarty S, Mankhand T (2012) Leaching of gold from the waste mobile phone printed circuit boards (PCBs) with ammonium thiosulphate. Int J Metall Eng 1:17–21. https://doi.org/10.5923/j.ijmee.20120102.02
Stratiotou Efstratiadis V, Michailidis N (2022) Sustainable recovery, recycle of critical metals and rare earth elements from waste electric and electronic equipment (circuits, solar, wind) and their reusability in additive manufacturing applications: a review. Metals. https://doi.org/10.3390/met12050794
Birloaga I, Vegliò F (2016) Study of multi-step hydrometallurgical methods to extract the valuable content of gold, silver and copper from waste printed circuit boards. J Environ Chem Eng 4(1):20–29. https://doi.org/10.1016/j.jece.2015.11.021
Calla-Choque D, Nava-Alonso F, Fuentes-Aceituno JC (2016) Acid decomposition and thiourea leaching of silver from hazardous jarosite residues: effect of some cations on the stability of the thiourea system. J Hazard Mater 317:440–448. https://doi.org/10.1016/j.jhazmat.2016.05.085
Birloaga I, Coman V, Kopacek B, Vegliò F (2014) An advanced study on the hydrometallurgical processing of waste computer printed circuit boards to extract their valuable content of metals. Waste Manag 34(12):2581–2586. https://doi.org/10.1016/j.wasman.2014.08.028
Torres R, Lapidus GT (2016) Copper leaching from electronic waste for the improvement of gold recycling. Waste Manag 57:131–139. https://doi.org/10.1016/j.wasman.2016.03.010
Elomaa H, Seisko S, Lehtola J, Lundström M (2019) A study on selective leaching of heavy metals vs iron from fly ash. J Mater Cycles Waste Manag 21(4):1004–1013. https://doi.org/10.1007/s10163-019-00858-w
Meer I, Nazir R (2018) Removal techniques for heavy metals from fly ash. J Mater Cycles Waste Manag 20(2):703–722. https://doi.org/10.1007/s10163-017-0651-z
Pinho SC, Ribeiro C, Ferraz CA, Almeida MF (2021) Copper, zinc, and nickel recovery from printed circuit boards using an ammonia–ammonium sulphate system. J Mater Cycles Waste Manag 23(4):1456–1465. https://doi.org/10.1007/s10163-021-01226-3
Yin K, Li P, Chan WP, Dou X, Wang J-Y (2018) Characteristics of heavy metals leaching from MSWI fly ashes in sequential scrubbing processes. J Mater Cycles Waste Manag 20(1):604–613. https://doi.org/10.1007/s10163-017-0631-3
Takahashi F, Etoh J, Shimaoka T (2010) Metal mobilization from municipal solid waste incineration bottom ash through metal complexation with organic and inorganic ligands. J Mater Cycles Waste Manag 12(1):1–9. https://doi.org/10.1007/s10163-009-0266-0
Arslan V (2021) Bacterial leaching of copper, zinc, nickel and aluminum from discarded printed circuit boards using acidophilic bacteria. J Mater Cycles Waste Manag 23(5):2005–2015. https://doi.org/10.1007/s10163-021-01274-9
Liang G, Li P, Liu W, Wang B (2016) Enhanced bioleaching efficiency of copper from waste printed circuit boards (PCBs) by dissolved oxygen-shifted strategy in Acidithiobacillus ferrooxidans. J Mater Cycles Waste Manag 18(4):742–751. https://doi.org/10.1007/s10163-015-0375-x
Goldprice (2020) From https://goldprice.org/gold-price-history.html. Retrieved 20 Mar 2020
Shen H, Forssberg E (2003) An overview of recovery of metals from slags. Waste Manag 23(10):933–949. https://doi.org/10.1016/S0956-053X(02)00164-2
Boduen A, Petrov G, Ttoitskaya M, Fokina S (2014) Technogenic deposits in Russia: precious metals stocks and prospects of their recovery. J Mater Sci Res 3(1):21–24. https://doi.org/10.5539/jmsr.v3n1p21
Acknowledgements
We acknowledge Ellen Röhm, Benjamin Gann, Rainer Kirchhof and Muammar Mansor for technical support and discussions. This study was supported by The Central Innovation Program for SMEs (grant number: KF2041705RH4; Germany), the International Academic Mobility Network with Russia (IAMONET-RU) under the Erasmus Mundus Action 2 Partnerships—Programme of the European Union, and by infrastructural support by the German Research Foundation (DFG) under Germany’s Excellence Strategy, cluster of Excellence EXC2124, project ID 390838134.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abramov, S.M., He, J., Wimmer, D. et al. Thiourea leaching of gold from processed municipal solid waste incineration residues. J Mater Cycles Waste Manag 24, 2243–2254 (2022). https://doi.org/10.1007/s10163-022-01476-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-022-01476-9