Abstract
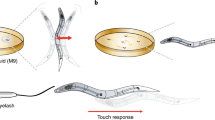
Nematodes such as Caenorhabditis elegans offer a very effective and tractable system to probe the underlying mechanisms of diverse sensory behaviors. Numerous platforms exist for quantifying nematode behavior and often require separate dependencies or software. Here I describe a novel and simple tool called NemaCount that provides a versatile solution for the quantification of nematode chemotaxis behavior. The ease of installation and user-friendly interface makes NemaCount a practical tool for measuring diverse behaviors and image features of nematodes such as C. elegans. The main advantage of NemaCount is that it operates from within a modern browser such as Google Chrome or Apple Safari. Any features that change in total number, size, shape, or angular distance between control and experimental preparations are suited to NemaCount for image analysis, while commonly used chemotaxis assays can be quantified, and statistically analyzed using a suite of functions from within NemaCount. NemaCount also offers image filtering options that allow the user to improve object detection and measurements. NemaCount was validated by examining nematode chemotaxis behavior; angular distances of locomotory tracks in C. elegans; and body lengths of Heterorhabditis bacteriophora nematodes. Apart from a modern browser, no additional software is required to operate NemaCount, making NemaCount a cheap, simple option for the analysis of nematode images and chemotaxis behavior.





Similar content being viewed by others
References
Avery L, Bargmann CI, Horvitz HR (1993) The Caenorhabditis elegans unc-31 gene affects multiple nervous system-controlled functions. Genetics 134:455–464
Bargmann CI, Hartwieg E, Horvitz HR (1993) Odorant-selective genes and neurons mediate olfaction in C. elegans. Cell 74:515–527
Beets I, Janssen T, Meelkop E, Temmerman L, Suetens N, Rademakers S, Jansen G, Schoofs L (2012) Vasopressin/oxytocin-related signaling regulates gustatory associative learning in C. elegans. Science 338:543–545. doi:10.1126/science.1226860
Bogoch II, Andrews JR, Speich B, Utzinger J, Ame SM, Ali SM, Keiser J (2013) Mobile phone microscopy for the diagnosis of soil-transmitted helminth infections: a proof-of-concept study. Am J Trop Med Hyg 88:626–629. doi:10.4269/ajtmh.12-0742
Brenner S (1974) The genetics of Caenorhabditis elegans. Genetics 77:71–94
Cronin CJ, Mendel JE, Mukhtar S, Kim YM, Stirbl RC, Bruck J, Sternberg PW (2005) An automated system for measuring parameters of nematode sinusoidal movement. BMC Genet 6:5
Goodman MB, Klein M, Lasse S, Luo L, Mori I, Samuel A, Sengupta P, Wang D (2014) Thermotaxis navigation behavior. WormBook. doi:10.1895/wormbook.1.168.1
Hart A (2006) Behavior. In: The C. elegans Research Community (ed) WormBook. doi:10.1895/wormbook.1.87.1
Husson SJ, Costa WS, Schmitt C, Gottschalk A (2012) Keeping track of worm trackers. WormBook. doi:10.1895/wormbook.1.156.1
Ito H, Inada H, Mori I (2006) Quantitative analysis of thermotaxis in the nematode Caenorhabditis elegans. J Neurosci Methods 154:45–52
Komatsu H, Mori I, Rhee JS, Akaike N, Ohshima Y (1996) Mutations in a cyclic nucleotide-gated channel lead to abnormal thermosensation and chemosensation in C. elegans. Neuron 17:707–718
Kwon N, Pyo J, Lee SJ, Je JH (2013) 3-D worm tracker for freely moving C. elegans. PLoS ONE 8:e57484. doi:10.1371/journal.pone.0057484
L’Etoile ND, Bargmann CI (2000) Olfaction and odor discrimination are mediated by the C. elegans guanylyl cyclase ODR-1. Neuron 25:575–586
Murphy CT, McCarroll SA, Bargmann CI, Fraser A, Kamath RS, Ahringer J, Li H, Kenyon C (2003) Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424:277–283. doi:10.1038/nature01789
O’Halloran DM, Burnell AM (2003) An investigation of chemotaxis in the insect parasitic nematode Heterorhabditis bacteriophora. Parasitology 127:375–385
O’Leary SA, Stack CM, Chubb MA, Burnell AM (1998) The effect of day of emergence from the insect cadaver on the behavior and environmental tolerances of infective juveniles of the entomopathogenic nematode Heterorhabditis megidis (strain UK211). J Parasitol 84:665–672
Pirnstill CW, Cote GL (2015) Malaria diagnosis using a mobile phone polarized microscope. Sci Rep 5:13368. doi:10.1038/srep13368
Raizen DM, Zimmerman JE, Maycock MH, Ta UD, You YJ, Sundaram MV, Pack AI (2008) Lethargus is a Caenorhabditis elegans sleep-like state. Nature 451:569–572. doi:10.1038/nature06535
Ramot D, Johnson BE, Berry TL Jr, Carnell L, Goodman MB (2008) The Parallel Worm Tracker: a platform for measuring average speed and drug-induced paralysis in nematodes. PLoS ONE 3:e2208. doi:10.1371/journal.pone.0002208
Schafer WR, Kenyon CJ (1995) A calcium-channel homologue required for adaptation to dopamine and serotonin in Caenorhabditis elegans. Nature 375:73–78. doi:10.1038/375073a0
Skandarajah A, Reber CD, Switz NA, Fletcher DA (2014) Quantitative imaging with a mobile phone microscope. PLoS ONE 9:e96906. doi:10.1371/journal.pone.0096906
Swierczek NA, Giles AC, Rankin CH, Kerr RA (2011) High-throughput behavioral analysis in C. elegans. Nat Methods 8:592–598. doi:10.1038/nmeth.1625
Switz NA, D’Ambrosio MV, Fletcher DA (2014) Low-cost mobile phone microscopy with a reversed mobile phone camera lens. PLoS ONE 9:e95330. doi:10.1371/journal.pone.0095330
Tramm N, Oppenheimer N, Nagy S, Efrati E, Biron D (2014) Why do sleeping nematodes adopt a hockey-stick-like posture? PLoS ONE 9:e101162. doi:10.1371/journal.pone.0101162
Troemel ER, Kimmel BE, Bargmann CI (1997) Reprogramming chemotaxis responses: sensory neurons define olfactory preferences in C. elegans. Cell 91:161–169
Vidal-Gadea A, Ward K, Beron C, Ghorashian N, Gokce S, Russell J, Truong N, Parikh A, Gadea O, Ben-Yakar A, Pierce-Shimomura J (2015) Magnetosensitive neurons mediate geomagnetic orientation in Caenorhabditis elegans. Elife. doi:10.7554/eLife.07493.10.7554/eLife.07493
Wang SJ, Wang ZW (2013) Track-a-worm, an open-source system for quantitative assessment of C. elegans locomotory and bending behavior. PLoS ONE 8:e69653. doi:10.1371/journal.pone.0069653
Wang D, O’Halloran D, Goodman MB (2013) GCY-8, PDE-2, and NCS-1 are critical elements of the cGMP-dependent thermotransduction cascade in the AFD neurons responsible for C. elegans thermotaxis. J Gen Physiol 142:437–449. doi:10.1085/jgp.201310959
Ward S (1973) Chemotaxis by the nematode Caenorhabditis elegans: identification of attractants and analysis of the response by use of mutants. Proc Natl Acad Sci USA 70:817–821
White GF (1927) A method for obtaining infective nematode larvae from cultures. Science 66:302–303
Wicks SR, de Vries CJ, van Luenen HG, Plasterk RH (2000) CHE-3, a cytosolic dynein heavy chain, is required for sensory cilia structure and function in Caenorhabditis elegans. Dev Biol 221:295–307. doi:10.1006/dbio.2000.9686
Woodring JL, Kaya HK (1998) Steinernematid and heterorhabditid nematodes: a handbook of biology and techniques. Southern Cooperative Series Bulletin Arkansas Agricultural Experiment Station, Fayetteville
Yang JS, Nam HJ, Seo M, Han SK, Choi Y, Nam HG, Lee SJ, Kim S (2011) OASIS: online application for the survival analysis of lifespan assays performed in aging research. PLoS ONE 6:e23525. doi:10.1371/journal.pone.0023525
Yuan J, Shaham S, Ledoux S, Ellis HM, Horvitz HR (1993) The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1 beta-converting enzyme. Cell 75:641–652
Acknowledgments
I thank members of the O’Halloran laboratory for critical reading of the manuscript.
Funding
The George Washington University (GWU) Columbian College of Arts and Sciences, GWU Office of the Vice-President for Research, and the GWU Department of Biological Sciences. Some strains were provided by the Caenorhabditis Genetics Center (CGC), which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440).
Author contributions
D.M.O’H conceived the idea for NemaCount, wrote the code, and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
O’Halloran, D.M. NemaCount: quantification of nematode chemotaxis behavior in a browser. Invert Neurosci 16, 5 (2016). https://doi.org/10.1007/s10158-016-0188-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10158-016-0188-1