Abstract
To investigate the cell cycle and cellular mechanisms of leukocyte elevation by laser acupuncture in rats with cyclophosphamide (CTX)-induced leukopenia. Sixty-six rats were randomized into six groups: normal, model control group, sham treatment group, 10.6 μm laser treatment group, 650 nm laser treatment group, and 10.6 μm–650 nm compound laser treatment group. Eleven rats were used in the normal group and 55 were models that were injected with cyclophosphamide to induce leukopenia. For the three laser treatment groups, 10.6-μm and 650-nm lasers, and 10.6-μm–650-nm compound lasers were used to irradiate the DU14 (Dazhui) and bilateral ST36 (Zusanli) for 5 min each. The sham laser group received the same operation as the laser group but without irradiation. The normal group and model group were not treated. Differences in the number of nucleated cells in the femoral bone marrow, and cell cycle and cellular apoptosis of peripheral leukocytes in rats in various groups were compared. Compared with the model group and the sham laser group, the number of nucleated cells in the femoral bone marrow in the 10.6-μm laser, 650-nm laser, and 10.6-μm–650-nm compound laser group was significantly increased after treatment (P = 0.001, 0.002, 0.034, respectively) and did not show any significant difference with the normal group (P = 0.964, P = 0.838, P = 0.287, respectively). The number of cells in G2 phase in the 10.6 μm laser group was similar to that of the normal group (P = 0.973). The number of cells in G2 phase in the model, sham, 650-nm laser group, and 10.6-μm–650-nm compound laser group were significantly lower than in the normal group and 10.6-μm laser group (P = 0.016, P = 0.023, P = 0.044, P = 0.039, respectively). In the model group and the sham treatment group, the apoptosis rates of peripheral leukocytes were increased compared with the normal group (P = 0.001), while the proportion of cells in the G2 phase was significantly lower than in the normal group (P = 0.016), and the proportion of cells in S phase was higher than in the normal group (P = 0.014). The incidence of apoptosis in peripheral blood cells in the three laser treatment groups did not show any statistically significant difference when compared with the normal group (P > 0.05). Treatment with the 10.6-μm, 650-nm, and 10.6-μm–650-nm compound lasers increased the number of nucleated cells in the bone marrow, decreased the unfavorable effects of cyclophosphamide on the cell cycle, induced the cell cycle towards proliferation, decreased apoptosis, improved the intramedullary hematopoietic system, and increased peripheral leukocyte count.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There were 14.1 million new cancer cases, 8.2 million cancer deaths, and 32.6 million people living with cancer (within 5 years of diagnosis) in 2012 worldwide. Fifty-seven percent (8 million) of new cancer cases, 65% (5.3 million) of the cancer deaths, and 48% (15.6 million) of the 5-year prevalent cancer cases occurred in less developed regions [1]. Surgery, radiotherapy, and chemotherapy are the major methods of tumor therapy [2, 3]; however, these methods lack specificity and selectivity, and severely affect the treatment regimen for malignant tumors and patients’ quality of life [4,5,6,7,8,9]. Among the side effects of chemotherapy, such as gastrointestinal reactions, bone marrow suppression, and immunosuppression, leukopenia due to the toxicity of anti-cancer drugs accounts for a significant proportion of secondary leukopenia [10]. The major secondary cause of leukopenia is bone marrow suppression caused by anti-tumor drugs [11]. Modern research has found that acupuncture has positive effects, such as ameliorating leukopenia after chemotherapy, promoting the division of murine bone marrow granulocytes after chemotherapy, increasing peripheral leukocyte count, inducing granulocyte-macrophage colony-stimulating factor [12, 13], alleviating pain in cancer patients [14], ameliorating chemotherapy side effects, and improving immunity in patients [15]. In addition, traditional acupuncture has stable effects and is low cost, providing a new avenue for clinical treatment of leukopenia [16]. However, acupuncture has shortcomings such as wound pain and dizziness, and can cause infection, while moxibustion can cause pollution [17]. Therefore, clinical applications and promotion require further improvements.
Research on the use of lasers on acupoints has a history of more than 20 years. The wavelength of lasers that are commonly used in clinical practice is mainly concentrated in the range of 600–1500 nm. Lasers in this waveband have very good penetration and can reach a depth of 10–25 mm in biological tissue to produce effects akin to acupuncture and are termed as “light needles” [18]. The human body is a natural biological infrared radiation source, and its radiation peak is around 10 μm [19, 20]. Studies have found that the infrared radiation spectrum of indirect moxibustion with monkshood cake, ginger, or garlic used for insulation is astonishingly identical to the spontaneous radiation spectrum of the human body [21]. This demonstrates that indirect moxibustion and infrared resonance absorption of acupoints may play an important role in the therapeutic effects of moxibustion, particularly insulated moxibustion (indirect moxibustion). The wavelength of carbon dioxide lasers is 10.6 μm, which is very close to the infrared radiation of the human body. Therefore, the radiation of carbon dioxide lasers is absorbed by skin tissues to produce a certain level of heat stimulation with similar therapeutic efficacy to moxibustion [22].
Laser acupuncture is the combination of China’s traditional acupuncture and modern laser technology. This technique does not cause trauma to the skin, does not produce pain or smoke, has adjustable treatment parameters, and is an improvement over traditional acupuncture [23]. Carbon dioxide lasers can produce resonance effects on human tissues, which is the biophysical foundation for their therapeutic effects [24]. In addition, low-dose lasers can produce heating effects that will have longer-lasting effects on the human body [25]. A 650-nm semi-conductor laser can penetrate deep below the subcutaneous tissues and stimulate multiple nerve endings on the acupoints to produce similar effects as acupuncture [26]. Previous experiments have found that 10.6-μm carbon dioxide lasers and 650-nm semi-conductor lasers can effectively increase peripheral leukocytes in a rat model of cyclophosphamide-induced leukopenia [27].
Cyclophosphamide is a non-specific drug that acts on the cell cycle by cross-linking to DNA and inhibiting DNA synthesis. Due to the high number of cancer cells that are in the mitotic phase, cyclophosphamide can interfere with abnormal proliferation of cells in a timely manner and over a wide range, thereby killing cancer cells [28]. However, chemotherapy damages bone marrow hematopoietic cells that are proliferating and result in increased apoptosis of bone marrow cells, stem cell depletion, and decreased proliferation, thereby causing leukocyte synthesis to decrease [29]. It is known that laser acupuncture can promote an increase in peripheral leukocyte count, but not whether laser acupuncture can improve the bone marrow hematopoiesis environment and decrease the inhibitory effects of cyclophosphamide on bone marrow cells. In this study, we examined the effects of laser acupuncture on peripheral blood cell apoptosis, changes in cell cycle, and on bone marrow nucleated cells in a rat leukopenia model to investigate the effector mechanisms of laser acupuncture.
Materials and methods
Experimental animals
Sixty-six healthy adult male Sprague-Dawley rats were obtained from the experimental animal center of Shanghai University of Traditional Chinese Medicine. These rats weighed 200 ± 20 g, and leukocyte counts were within normal range. The temperature of the animal room was (24 ± 1) °C, humidity was 50–70%, and animals were kept in clean housing conditions. Animals were purchased from Shanghai SIPPR-Bk Lab Animal Co., Ltd. (production approval number SCXK (Hu) 2009-0018). The approval number for use of experimental facilities at the Shanghai University of Traditional Chinese Medicine was SCXK (Hu) 2009-0069.
Main experimental equipment
The 10.6-μm CO2 laser treatment device (SX10-C1) was produced by Shanghai Wonderful Opto-Electrics Tech Co, Ltd., and has a laser wavelength of 10.6 μm. The 650 semi-conductor laser treatment device (LFJ-650A) was produced by Shanghai Wonderful Opto-Electrics Tech Co, Ltd. and has a laser wavelength of 650 nm.
The output power of the continuous 10.6-μm CO2 laser was 80 mW with the size of irradiation spot as 2 mm in diameter on the skin, and thus the power density as 2.5 W/cm2, and the dose is 750 J/cm2. The output power of continuous 650 nm laser was 36 mW, with the size of irradiation spot as 1 mm in diameter on the skin, and thus, the power density as 4.6 W/cm2, and the dose is 1380 J/cm2.
Experimental groups
The 66 rats were randomized into six groups according to weight numbers and random numbers generated by SAS 18.0. Models were not created for the normal group, while identical models were created for the remaining groups (Fig. 1).
-
1.
Normal group: Normal housing in a laboratory environment with the same batch of experimental animals.
-
2.
Model control group: Intraperitoneal injection of cyclophosphamide (CTX) was carried out with an initial dose of 80 mg/kg. On days 6 and 10 after the initial dose, intraperitoneal injections of 40 mg/kg were given according to weight. After injection, the rats were housed normally in laboratory conditions [30]. When the rats in the laser treatment groups were receiving laser irradiation, the rats in model control group were immobilized with a rat fixator without anesthetized in the same way as those in the laser treatment groups.
-
3.
The three laser treatment groups: 1 cm in diameter of hair was shaved around the ST36 (Zusanli) on both lower limbs and the DU14 (Dazhui) at the back, and the acupoints were labeled. Then, the rats were immobilized with a rat fixator without anesthetic. In the 10.6-μm laser group, the 10.6-μm laser (70 mW in output power, spot size of 2 mm in diameter on the skin) was switched on and the irradiation probe was aimed at the acupoints. In the 650-nm laser group, a 650-nm semi-conductor laser (36 mW in output power, spot size of 1 mm in diameter on the skin) was used to irradiate at the points. In the 10.6-μm–650-nm compound laser group, both lasers were used to irradiate at the points simultaneously. Each acupoint was irradiated for 5 min, and each rat was treated for a total of 15 min/day on alternate days for 15 days (seven treatments).
-
4.
Sham laser treatment group: The procedures were identical with the three laser treatment groups, except that the laser output was switched off, with only the indicator lights on.
Model generation and treatment. An initial dose of 80 mg/kg was administered by intraperitoneal injection. On days 6 and 10 after the initial dose, intraperitoneal injections of 40 mg/kg were given according to weight. Treatment was carried out on alternate days for 15 days for a total of seven treatments
Acupoint selection and localization
The DU14 (Dazhui) and ST36 (Zusanli) were selected. The localization of the acupoints was based on the criteria from the state-regulated educational material, “Experimental Acupuncture,” from the Eleventh Five-Year Plan [31]. The ST36 (Zusanli) is located at 5 mm below the head of the fibula behind the knee. The DU14 (Dazhui) is located on the midline of the back, between the spinous process of the seventh cervical vertebra and the first thoracic vertebra.
Sample collection, testing, and method of experimental markers
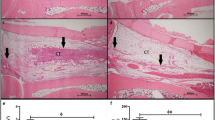
Use of a high-magnification microscope to count number of nucleated cells in bone marrow
Rat femurs were extracted, and holes were drilled in the front and back of the femur. Ten milliliters of 3% glacial acetic acid was used to repeatedly flush the bone marrow, and a size 4 needle was used to disperse the bone marrow cells. The total number of nucleated cells at the four large squares in the four corners was enumerated at low magnification. The number of nucleated cells = number of cells under the microscope × 2.5 × 10,000. Enumeration was carried out four times for each rat, and the average number of nucleated cells in the left and right femoral bone marrow was calculated.
Flow cytometry analysis of cell cycle and apoptosis in peripheral blood cells
On day 15 of the experiment, 10 mL of blood was obtained from the carotid artery, and erythrocyte lysis buffer was added. Flow cytometry was used to analyze apoptosis and the cell cycle of peripheral blood cells. Flow cytometry software was used for fitting and analysis of the cell cycle.
Statistical analysis
SPSS 18.0 statistical software was used for statistical analysis. Quantitative data is shown as means ± standard error (mean ± SE), and intergroup comparisons were carried out using one-way ANOVA. Intra-group comparisons were carried out using repeated measures ANOVA for pairwise comparisons. A difference of P < 0.05 was deemed to be significant.
Experimental results
Enumeration of femoral bone marrow nucleated cells
There were no significant differences in the number of nucleated cells in the bone marrow when rats in the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group were compared with the normal group (P = 0.964, P = 0.838, P = 0.287, respectively). The number of bone marrow nucleated cells in the model group and the sham treatment group was significantly lower than in the normal group (P = 0.001, 0.010, respectively).
The number of bone marrow nucleated cells in the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group was significantly higher than in the model group (P = 0.001, 0.002, 0.034, respectively).
The number of bone marrow nucleated cells in the three laser treatment groups was significantly higher than in the sham treatment group (P > 0.05). There was no statistical difference in the number of bone marrow nucleated cells between the model group and the sham treatment group (P = 0.937) (Table 1, Figs. 2 and 3).
Apoptosis and cell cycle of peripheral leukocytes
Cellular apoptosis
The apoptosis rates in the model group and the sham treatment group were significantly higher than in the normal group (P = 0.001). There were no significant differences between the 10.6-μm laser group, 650-nm laser group, and the normal group in apoptosis rates (P > 0.05). The relative apoptosis rates of the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group did not show any significant differences (P = 0.988, P = 0.731, P = 0.999, respectively) (Table 2, Fig. 4).
Comparison of proportions of cells in cell cycle stages and apoptosis of blood cells after treatment. The apoptosis rates in the model group and the sham treatment group were significantly higher than in the normal group (P = 0.001). There were no significant differences between the 10.6-μm laser group, 650-nm laser group, and the normal group in apoptosis rates (P > 0.05). The relative apoptosis rates of the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group did not show any significant differences (P = 0.988, P = 0.731, P = 0.999, respectively). The proportion of cells in G1 phase for all groups were generally similar, with no statistically significant differences (P = 0.998, P = 0.998, P = 0.998, P = 0.999, respectively). The number of cells in S phase for the model group and sham treatment group was significantly higher than in the normal group (P = 0.014 and 0.020, respectively). There were no significant differences in the proportion of cells in S phase between the 10.6-μm laser group, 650-nm laser group, 10.6-μm–650-nm compound laser group, and the normal group (P = 0.988, 0.998, 0.999, respectively). The proportion of cells in S phase in the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group were significantly higher than in the model group and the sham treatment group (P = 0.013, P = 0.017, P = 0.025, respectively). The number of cells in G2 phase in the 10.6-μm laser group was similar to that of the normal group (P = 0.973). While the cell number in G2 phase in the other two laser groups (i.e., 650-nm laser group and 10.6-μm–650-nm compound laser group) was significantly lower than that in normal group (P = 0.044, P = 0.039, respectively), and similar with that in the model group and sham treatment group (P = 0.283, P = 0.207, respectively)
Cell cycle
The proportion of cells in G1 phase for all groups were generally similar, with no statistically significant differences (P = 0.998, P = 0.998, P = 0.998, P = 0.999, respectively).
The number of cells in S phase for the model group and sham treatment group was significantly higher than in the normal group (P = 0.014, 0.020, respectively). There were no significant differences in the proportion of cells in S phase between the 10.6-μm laser group, 650-nm laser group, 10.6-μm–650-nm compound laser group, and the normal group (P = 0.988, 0.998, 0.999, respectively). The proportion of cells in S phase in the 10.6-μm laser group, 650-nm laser group, and 10.6-μm–650-nm compound laser group were significantly higher than in the model group and the sham treatment group (P = 0.013, P = 0.017, P = 0.025, respectively).
The number of cells in G2 phase in the 10.6-μm laser group was similar to that of the normal group (P = 0.973), while the cell number in G2 phase in the other two laser groups (i.e., 650-nm laser group and 10.6-μm–650-nm compound laser group) was significantly lower than that in normal group (P = 0.044, P = 0.039, respectively), and similar with that in the model group and sham treatment group (P = 0.283, P = 0.207, respectively).
Discussion
The major side effect of cyclophosphamide treatment is bone marrow suppression, which results in significant damage to actively proliferating hematopoietic cells and direct damage to the bone marrow microenvironment and myeloid progenitor/stem cells. This causes leukopenia and reduced immune function, thereby causing severe damage to the body [32, 33]. In this study, we found that irradiation with a 10.6-μm carbon dioxide laser and 650-nm semi-conductor laser alone or in combination increased the number of nucleated cells in the femoral bone marrow of cyclophosphamide-treated model rats. That is, after seven treatments in 15 days, the reservoir in the bone marrow produced large amounts of undifferentiated leukocytes. Laser acupuncture stimulated bone marrow hematopoietic cell numbers that were destroyed by cyclophosphamide and preserved the proliferation ability of bone marrow nucleated cells. In contrast, the model group and the sham treatment group were still affected by the cytotoxicity of cyclophosphamide, and the number of bone marrow nucleated cells was decreased and did not recover to normal values. In a previous study, after a 10.6-μm carbon dioxide laser was used for eight treatments, it was found that on day 17, there were no significant differences in peripheral leukocytes when compared with the normal group. Our study found that after seven treatments, there were no significant differences in nucleated cells between the laser groups and the normal group. The combined results of the two studies suggest that lasers can improve the bone marrow hematopoietic system, promote division in intramedullary nucleated cells, accelerate division, and protect cells that have undergone division and maturation to enter into peripheral circulation. This is one of the mechanisms by which laser acupuncture increases leukocyte count.
Apoptosis refers to the process where cells undergo automatic programmed cell death under certain physiological or pathological conditions as regulated by genes. Cancer is a disease in which there are abnormalities in the regulation of cell growth and is intimately associated with abnormal cellular apoptosis. Chemotherapy drugs can disrupt the cell cycle and apoptosis in normal cells while interfering with the abnormal growth of cancer cells. After the generation of the cyclophosphamide model, DNA in rat blood cells loses its normal ability to regulate apoptosis. Our experimental results showed that the apoptosis rates in the model group and the sham treatment group were significantly higher than in the normal group. This is due to the initiation of cellular apoptosis in peripheral blood cells, resulting in a decrease in peripheral leukocyte count. Laser treatment can improve the ability of apoptosis regulation in cells that have experienced DNA disruption, resulting in apoptosis rates in rats with leukopenia being the same as in the normal group, thus maintaining peripheral blood cells.
Cyclophosphamide (an alkylating agent) is a highly active compound and can form covalent bonds with various nucleophilic groups to cause DNA breakage and damage [34]. Cyclophosphamide is a non-specific drug that targets the cell cycle and is effective at various stages, including the G0 phase (non-proliferating quiescent phase). Therefore, it can kill a wide range of cancer cells and damage normal cells at the same time. The cell cycle results showed that the number of cells in the S phase of the cyclophosphamide model group and the sham treatment group was significantly increased, but cells in G2 phase were significantly decreased. This demonstrated that cyclophosphamide caused S phase arrest, resulting in cells unable to successfully carry out cell division and replication and enter G2 phase. The main reason for this is that nucleic acid synthesis is affected, resulting in incomplete DNA replication and cells unable to progress to the next phase. In addition, cyclophosphamide may also inhibit cell division, resulting in cells that have completed DNA replication but are unable to divide, causing the proportion of cells in G2 phase to decrease. The G1 phase of the cell cycle (gap 1 phase, before DNA synthesis) is a phase where new daughter cells from the previous mitotic division synthesize RNA and proteins that are associated with the unique functions of specific cells. G1 is longer than the other phases of the cell cycle. The results of this study found that whether cyclophosphamide was used or whether laser treatment was carried out did not greatly affect G1 phase. Therefore, the effects of cyclophosphamide as an alkylating agent on immune cells do not occur in G1 phase, i.e., cyclophosphamide does not affect the ability to synthesize cellular materials. During S phase (synthesis phase, the DNA replication phase), the cells synthesize DNA, and this period is relatively short. The DNA content in cells is increased by 2-fold for subsequent equal distribution into two daughter cells. The results of this study showed that the number of cells in S phase after laser treatment was comparable to that in the normal group. Parental cells can therefore divide rapidly in a short period to produce daughter cells and increase the number of peripheral leukocytes. The G2 (gap 2) phase refers to the phase after DNA replication and before mitosis starts, and is usually very short. The results of G2 phase in three laser groups are higher than model group, while it only has statistically significant differences in 10.6-μm laser group. The number of cells in G2 phase was increased with 10.6-μm laser acupuncture, which was higher than in the model group and identical to the normal group. Namely that mild stimuli excite biologic activity, stronger ones support it, even stronger ones retard it and very strong ones arrest it completely. The power of combined laser may be too strong to rats. What is more, there was some “negative interference” between the photochemical effect induced by the red light, and the photophysical effect from the CO2 energy. Thus, the 10.6-μm laser moxibustion is more suitable for the treatment of this symptom. There are evidences that the moxibustion method with white cell disease has a definite effect [12, 35]. This demonstrates that the 10.6-μm lasers affect the cell cycle and improve the ability of cells to synthesize RNA and proteins. As G2 phase is also where DNA damage is repaired before chromosome separation [36], the increase in number of cells in G2 provides more opportunity for DNA damage repair. However, further research is required to identify the pathways that are affected. Our experimental results also show that lasers can specifically affect cell division in leukopenia, promote cell division and proliferation, affect DNA damage caused by cyclophosphamide, promote DNA synthesis, and thereby increase leukocyte counts. This shows that laser acupuncture can promote S phase cells to enter into G2 phase and promote cell proliferation, achieving the aim of increasing peripheral leukocyte counts.
In our current research, we only investigate the changes due to laser stimulation in the target tissue (bone marrow), but not at the local area of stimulation. We therefore do not know the changes at the local area (the skin layer), and how the stimulation transmits to the target organs or tissues. We speculate that the effect of 650-nm red laser in our research may partly share a similar mechanism with that of its wound healing and pain attenuation effect [37] at the local area. The mechanism is a series of photochemical reactions of the light absorption in the end terminal enzyme of the respiratory chain of the cellular mitochondria, cytochrome oxidase, leading to a photochemical cascade the end result of which is adenosine triphosphate (ATP) production, accompanied by nitrous oxide (NO) and small but beneficial amounts of reactive oxygen species (ROS) [38]. The CO2 laser, on the other hand, emits a wavelength more than two orders of magnitude longer, and at that physically long wavelength compared with visible red light, cell membranes are opaque, just as glass is opaque to 10.6 μm, so the LLLT reaction cannot be primarily photochemical, but rather photophysical in nature. The 10.6-μm CO2 laser is primarily absorbed by the tissue water, and its wavelength is absorbed principally in the first 20 μm of tissue [39]; therefore, the effect of the low-density 10.6-μm laser may be mainly due to the heat conduction into the deeper tissues. However, since all infrared lasers have thermal effects, the future research is warranted to investigate the effects of different infrared lasers (such as 830-nm diode laser, 2.95 μm erbium: YAG laser, etc.) and whether the effect of 10.6-μm laser is superior than others, due to its similarity to the biophotonic radiation from human point in wavelength.
In conclusion, we found that treatment with a 10.6-μm laser, 650-nm laser, and 10.6-μm–650-nm compound laser increased the number of nucleated cells in the bone marrow, decreased the unfavorable effects of cyclophosphamide on the cell cycle, advanced the cell cycle towards proliferation, decreased apoptosis, affected the intramedullary hematopoietic system, and affected peripheral leukocyte counts. This study did not include a positive drug as a control group, and we did not carry out in-depth study on spinal cord-related mechanisms. These will be further examined in future studies.
References
World Health Organization (2012) Section of cancer information [DB/OL]. GLOBO CAN 2012. http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx
Burish TG, Lyles JN (1981) Effectiveness of relaxation training in reducing adverse reactions to cancer chemotherapy. J Behav Med 4(1):65
Hunter J, Chaoul A, Fisch M (2015) A relaxation strategy for reducing chemotherapy side-effects. J Psychosom Res 78(6):603
Zhang AY, Gary F, Zhu H et al (2015) Attitudes toward cancer and implications for mental health outcome in African-American cancer patients. J Cancer Educ 30(1):138–144
Yoshii H, Kikuchi F, Kang L (2016) Mental health care for cancer patients, their families and nurses in China: a review. Echocardiography 26(26):625–629
Wang Jin, (2008) Basic necessities of life and control of blood disease [M]. Chongqing press page7
Han Y-F, Gong Z, Huang L-Q, Xia X (2010) Clinical study on acupuncture for leukopenia induced by chemotherapy. Zhongguo Zhen Jiu 30(10):802–805
Lu W, Matulonis UA, Doherty-Gilman A et al (2009) Acupuncture for chemotherapy-induced neutropenia in patients with gynecologic malignancies: a pilot randomized, sham-controlled clinical trial. J Altern Complement Med 15(7):745–753
Ross JS, Schenkein DP, Pietrusko R et al (2004) Hortobagyi. Targeted therapies for cancer. Am J Clin Pathol 12(2):598–609
Devlin EJ, Linley A (2017) Cancer treatment side effects: a meta-analysis of the relationship between response expectancies and experience. J Pain Symptom Manag 54(2):245–258
(2005) Pemetrexed: new drug. Pleuralmesothelionma: a first encouraging trial. Prescrire Int 14(80):212–214
Ji HL, Jang E, Jung MH et al (2016) Clinical effectiveness of acupuncture in the treatment of chemotherapy-induced leukopenia: a systematic review. Eur J Integr Med 8(5):802–808
Pais I, Correia N, Pimentel I et al (2014) Effects of acupuncture on leucopenia, neutropenia, NK, and B cells in cancer patients: a randomized pilot study. Evid Based Complement Alternat Med:217–397
Fan AY, Miller DW, Bolash B, Bauer M, Pang J (2017) Acupuncture’s role in solving the opioid epidemic: evidence, cost-effectiveness, and care availability for acupuncture as a primary, non-pharmacologic method for pain relief and management–white paper 2017. J Integr Med 15(6):411–425
Posadzki P, Moon TW, Choi TY, Park TY, Lee MS, Ernst E (2013) Acupuncture for cancer-related fatigue: a systematic review of randomized clinical trials. Support Care Cancer 21:2067–2073
Huang QF, Cui XJ, Li S (2006) Review of leucopenia treated by acupuncture and moxibustion from 1949 to 2004. J Acupunct Tuina Sci 4:129–131
Han L (2013) Toxicological study of moxa smoke[D]. Beijing University of Traditional Chinese Medicine
Whittaker P (2004) Laser acupuncture: past, present, and future. Lasers Med Sci 19(2):69–80
Jianzi W, Shen X, Guanghong D et al (2007) Investigation of pathway and mechanism of heat stimulating action of sandwiched moxibustion. Chin Acupunct Moxibustion 27(5):391–393
Xiao XZ, Chu XF (2002) Effects of low-intensity laser treatment on neuro-endocrine-immune network and clinical progress. Anat Res 24(1):46–48 [In Chinese]
Xueyong S, Guanghong D, Junhao C et al (2003) Comparison of infrared radiation spectrum of traditional moxibustion, substitute moxibustion and acupoints of human body. J Infrared Millin Waves 22(2):123–126
Zhao L, Shen XY, Gao JP et al (2006) Effect of different LLLT on pituitrin-induced bradycardiain the rabbit. Lasers Med Sci 21(6):61–66
Shen X, Zhao L, Ding G et al (2009) Effect of combined laser acupuncture on knee osteoarthritis: a pilot study. Lasers Med Sci 24(2):129–136
Shen X, Ding G, Deng H et al (2008) Infrared radiation spectrum of acupuncture point on patients with coronary heart disease. Am J Chin Med 36(2):211–218
Xue-yong S, Guang-hong D, Jian-zi W et al (2006) An infrared radiation study of the biophysical characteristics of traditional moxibustion. Complement Ther Med 14(3):213–219
Makihara E, Makhara M, Masumi S et al (2005) Evaluation of facial thermo2 graphic changes before and after low-level laser irradiation. Phto Hed Laser Surg 23(2):191
Zhao J, Cheng K, Deng H (2014) The effect of different laser irradiation on cyclophosphamide-induced leucopenia in rats. Evid Based Complement Alternat Med Article ID 756406
Arnold H, Bourseaux F, Brock N (1958) Chemotherapeutic action of a cyclic nitrogen mustard phosphamideester (B 518-ASTA) in experimental tumours of the rat. Nature 181(4613):931
Galluzzi L, Buque A, Kepp O (2015) Immunological effects of conventional chemotherapy and targeted anticancer agents. Cancer Cell 28(14):690–714
Jacolot C, Judel M, Chaumais C et al (2012) Animal model methodology: immunocompetent or leucopenic rats, which is the best? Results from a model of experimental pneumonia due to derepressed cephalosporinase-producing Enterobacter cloacae. Chemotherapy 58:129–133
Li ZR (2007) Experimental Acupuncture M. China Press of Traditional Chinese Medicine, Beijing, p 242
Arnold H, Bourseaux F, Brock N (1958) Chemotherapeutic action of a cyclic nitrogen mustard phosphamideester (B 518-ASTA) in experimental tumours of the rat. Nature 181:931
Levine LA, Richie JP (1989) Urological complications of cyclophosphamide. J Urol 141(5):1063–1069
Mills BA, Roberts RW (1979) Cyclophosphamide-induced cardiomyopathy: a report of two cases and review of the English literature. Cancer 43(6):2223–2226
Shen XY, Fei L, Wu YC et al (2005) The effect of specific wavelength of infrared moxibustion on leukopenia in tumor patients undergoing radiotherapy and chemotherapy. Shanghai J Acupunct Moxibustion 24(4):1–3
Pollard TD, Earnshaw WC, Lippincott-Schwartz J, Johnson G (2006) Cell biology (Third Edition) Section X: cell cycle. ISBN: 978–0–12-164730-8. p 291–299
Ahmed OM, Mohamed T, Moustafa H et al (2018) Quercetin and low level therapy promote wound healing process in diabetic rats via structural reorganization and modulatory effects on inflammation and oxidative stress. Biomed Pharmacother 101:58–73
Lievens PC (1991) The effect of the combined He-Ne and IR laser treatment on the regeneration of the lymphatic system during the process of wound healing. Lasers Med Sci 6:189–191
Sliney DH, Trokel SL (1993) Medical laser and their safe use. Springer, New York, 93(6):89–90
Acknowledgements
We thank our peer reviewers, ** and ** (Dear editor, please provide us the name of the two peer reviewers), for their insightful comments to the manuscript.
Contract grant sponsor
This study was supported by the 973 Program of China (2015CB554505), Projects of the NNSF of China (81320108028, 81202648), and the Budget research project of Shanghai University of TCM (2016YSN19). The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experiments were carried out in accordance with the experimental animal center of Shanghai University of Traditional Chinese Medicine (Animal Experimental Ethical Number 2013013). Animals were purchased from Shanghai SIPPR-Bk Lab Animal Co., Ltd. (production approval number SCXK (Hu) 2009-0018). The approval number for use of experimental facilities at the Shanghai University of Traditional Chinese Medicine was SCXK (Hu) 2009-0069.
Conflict of interest
The authors declared that they have no conflicts of interest to this work.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Liu, L., Zhao, L., Cheng, K. et al. Examination of the cellular mechanisms of leukocyte elevation by 10.6 μm and 650 nm laser acupuncture-moxibustion. Lasers Med Sci 34, 263–271 (2019). https://doi.org/10.1007/s10103-018-2581-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-018-2581-y