Abstract
The implementation of sustainable development goals in the cosmetics industry is focused primarily on the phase of product design and the formulation of fully functional cosmetics based on raw materials of natural origin. The aim of the present study was to evaluate the feasibility of using a surfactin-rich digestate extract—a raw material of biotechnological origin—as an ingredient in cleansing cosmetics. A composition was designed, and shower gels based on the digestate extract were formulated. In the next stage of the study, the prototypical products were subjected to a quality assessment with an emphasis on the properties determining their functionality and safety of use by consumers. The shower gels formulated with raw materials of natural origin with the addition of the surfactin-rich extract show acceptable functional properties including viscosity, foaming ability and fat emulsification properties. Importantly, the use of the digestate extract was found to significantly improve the safety in use of the evaluated cosmetic prototypes. This finding is corroborated by a significant decrease in the zein value, which is a measure of the irritant effect of cosmetic formulations. A slight decrease in the ability to emulsify fats found for the tested prototypes confirmed their appropriate functionality and mild effect on the skin. Through the use of the surfactin-rich digestate extract, the cosmetics acquired a natural color derived from the extract. Colorimetric analysis shows that the color changes in the samples should be noticeable even by inexperienced observers.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The last decade has seen a major impact of the concept of sustainability throughout the cosmetics industry (Bom et al. 2019; Kolling et al. 2022). Consumers, and hence also cosmetics manufacturers, are increasingly interested in cosmetic products based on raw materials of natural origin which, by their definition, incorporate the principles of sustainable development at all stages of their life cycle (Amberg and Fogarassy 2019; Kapoor et al. 2019). In particular, widespread concerns about the safety of cosmetics and their impact on the environment have contributed to an increasing scientific interest in this area of study. Of all the stages in the life cycle of a cosmetic product, the development of product formulation and technology is the most crucial one. Designing formulations based on alternative ingredients recognized as more sustainable can be a significant challenge because of their instability, potential lack of the desirable functional parameters, issues related to the safety in use (i.e., effect of cosmetics on skin) (Seweryn 2018; Antignac et al. 2011; Nohynek et al. 2010) and esthetic considerations that are deemed very important by consumers. It is also important to note that the assessment of cosmetics formulated in line with the goal of sustainable development is a multifaceted process that takes into account environmental, social and economic factors as well as the quality and performance of the final product (Bom et al. 2019).
The growing demand for natural cosmetics has led to a recent major increase in the number of scientific studies investigating dedicated raw materials as well as formulations and manufacturing technologies of natural cosmetics (Savic et al. 2021; Goyal and Jerold 2021; Dini and Laneri 2021; Singh et al. 2022; Majchrzak et al. 2022; Fonseca-Santos et al. 2015). However, only some of the available studies evaluate complete cosmetic formulations based on raw materials of natural origin. Information related to this topic is still scattered and incomplete. Most reports address the application of plant extracts (Savic et al. 2021; Nizioł-Łukaszewska et al. 2017; Sikora et al. 2015), surfactants of natural origin (Klimaszewska et al. 2022; Seweryn and Bujak 2018; Nizioł‐Łukaszewska and Bujak 2018) and raw materials of biotechnological origin (Rincón-Fontán et al. 2018; Bezerra et al. 2018; Ferreira et al. 2017; Vecino et al. 2017), including surfactin.
High hopes are also pinned on the application of raw materials of biotechnological origin in cosmetics. Biotechnological bioprocesses offer an opportunity to obtain a range of compounds with interesting properties for the cosmetics industry, such as vitamins (Mani 2020) and biopolymers (Velamakanni et al. 2022; Furtado et al. 2022; Luft et al. 2020; Domżał-Kędzia et al. 2019). Also, there seems to be a great potential in the application of microorganisms in the production of surfactants. In fact, biosurfactants represent a viable alternative to the surfactants of plant or petrochemical origin that are widely used in cosmetics. They are fully natural and have a range of properties that make them potentially desirable cosmetic ingredients (Ferreira et al. 2017; Rincón-Fontán et al. 2018; Bezerra et al. 2018; Vecino et al. 2017; Jahan et al. 2020; Akbari et al. 2018; Lee et al. 2018; Lourith et al. 2009). The properties of surfactin seem to be particularly beneficial in this application. The compound is highly effective in reducing interfacial tension and has a low critical micelle concentration (Jahan et al. 2020; Ganesan and Rangarajan 2021; Bjerk et al. 2021). Research findings show that surfactin has a limited potential for skin irritation and toxicity compared to synthetic surfactants (Fei et al. 2020). Surfactin shows a number of characteristics that are suitable for use in cosmetics. Nonetheless, the range of applications of this compound is still fairly narrow because of high costs of production. Consequently, biosurfactants including surfactin are used in industrial practice only to a limited extent (Singh et al. 2022).
The use of ingredients derived from fermentation processes is known to bring a number of functional benefits in such formulations. Raw materials of this type have been scientifically validated to improve the bioavailability of ingredients that play a major role in skin care applications. Furthermore, the fermentation process itself, through an appropriate selection of processing conditions, offers an opportunity to obtain a specific range of compounds with desired functional properties. It has been shown that such raw materials may contain amino acids and peptides, vitamins, enzymes, fatty acids, minerals, antioxidants, etc. (Majchrzak et al. 2022; Mathur et al. 2020; Domżał-Kędzia et al. 2019; Sirilun et al. 2016). In conventional cosmetics, these compounds must be incorporated into the formulation at the production stage as separate raw materials. In contrast, digestate extracts are mixtures of components that may contribute to a major extent to the process of formulating cosmetic products. The compositions of digestate extracts are largely determined by the selection of microorganisms. Some raw materials obtained by fermentation may have a preservative effect on cosmetic systems (Majchrzak et al. 2022; Lee et al. 2020; Herman et al. 2019), while others display antioxidant or anti-aging effects (Majchrzak et al. 2022; Ziemlewska et al. 2021; Sivamaruthi et al. 2018). The use of digestate extracts as a completely natural source of ingredients that are known to nourish the skin and prevent skin irritation, as well as surfactants (biosurfactants), offering the advantage of not being obtained through chemical synthesis, may be a perfect solution for meeting sustainability goals in the cosmetics industry. It is important to note that the extract in question is a mixture of various ingredients in water. Consequently, the production of the extract contributes to a significant reduction in the use of water, which is a waste product in the process of obtaining the raw material. Producing a digestate rich in biosurfactants (surfactin) is associated with considerably less water waste compared to the conventional process of obtaining dry surfactin as a commercially available cosmetic raw material. In the current approach to the production process of this biosurfactant, all the water required for manufacturing purposes is waste. In the prototypes of cleansing cosmetics developed for the purpose of the present study, the digestate extract replaces water in the formulation, which also contributes to reducing the final water consumption.
The study was carried out to explore the possibilities for using a surfactin-rich digestate extract in cleansing cosmetics. It was decided that in compliance with the sustainability policy the cosmetic prototypes would be formulated with raw materials of natural origin. The first stage involved a biotechnological process that was aimed to produce a digestate extract with a high content of surfactin with the use of bacteria of the species Bacillus subtilis. Tests were carried out on the raw material to confirm the presence of surfactin. The extract was then added to the shower gel prototypes that were formulated entirely with ingredients of natural origin. The composition of the formulations was chosen in such a manner that preservatives conventionally used in cosmetics would not be necessary. The analysis included formulations containing different concentrations of the digestate extract (4, 40; 80%). The raw material replaced water in the product composition. The reference sample used in the study was a cosmetic without the digestate extract. The cosmetic suitability of the surfactin-rich digestate extract was evaluated by analyzing the results of basic physicochemical and functional tests of the formulated product prototypes. The formulations were assessed against a range of criteria including viscosity, color, fat emulsification ability, skin irritation potential and skin drying effect. The aim was to determine whether the surfactin-rich digestate extract may serve as a valuable ingredient of cleansing cosmetics based on natural ingredients and to evaluate the potential benefits of the digestate extract in terms of the application properties of the finished product and its safety of use by consumers.
Materials and methods
Materials
Raw materials for cosmetics
The prototypes shower gels were made using certified, vegetable-based raw materials which are approved for the production of natural products according to COSMOS standards: disodium cocoyl glutamate (trade name: Plantapon ACG 50; supplier: BASF, Ludwigshafen, Germany), xanthan gum (trade name: Cosphaderm X34; supplier: Cosphatec GmbH, Hamburg, Germany), propanediol (trade name: Cosphaderm Propanediol natural, supplier: Cosphatec GmbH, Hamburg, Germany), lactic acid (trade name: lactic acid 88%, supplier: WarChem, Warsaw, Poland), Bacillus subtilis ferment extract, propanediol, surfactin (supplier: InventionBio, Bydgoszcz, Poland) and distilled water.
Fermentation process for surfactin production with Bacillus subtilis natto KB1
Bacillus Subtilis fermentation extract was obtained according to (Lewińska 2021) with some modifications. The preculture was carried out at 37 °C with shaking the medium at 180 rpm. The initial optical density was set to OD600 nm = 0.1. The cultures were grown at 37 °C with continuous shaking at 200 rpm for 72 h. The cultures were then centrifuged (14,000 × g) at 4 °C for 30 min (Sigma 6K15, 12,500 rotor, DJB Labcare Ltd., Newport Pagnell, UK) to remove the bacteria. The precipitation step was omitted and the liquid broth was subjected to ultrafiltration and then reverse osmosis (RO) in order to concentrate the surfactin (Mini UF-NF-RO bench-scale crossflow skid, Convergence). Finally obtained enriched surfactin sample was mixed with propanediol.
Methods
Bacillus fermented extract analysis: confirmation of the surfactin
The mass spectrometry (MS) detection of surfactin sample was performed using a triple quadrupole mass spectrometer (4500 QTRAP, AB Sciex, Concord, ON, Canada), equipped with an electrospray ionization source (ESI) working in positive and negative-scan modes. The surfactin sample was diluted with methanol and directly injected by insert pump with a 10 μL min−1 flow rate. MS spectrum was obtained using full scan mode of the total ions with a mass scan of m/z 50–2000 Da. The identification of selected compounds was conducted by molecular mass and fragment of anion/cation entries of each compound and confirmed by MS2 fragmentation. Chromatografic separation of identified surfactin homologues was performed by ultrahigh-performance liquid chromatography, UHPLC (Sciex Exion, AB Sciex, Concord, ON, Canada) coupled to mass spectrometer, working in the multiple reaction monitoring (MRM) scan mode. 100 × 4.6 mm chromatographic column Kinetex 3.5 µm XB-C18 100 Å with iso-butyl side chains and with TMS endcapping stationary phase with similar composition guard column (Phenomenex, Torrance, CA, USA) was used and maintained at 30 °C. A binary solvent system comprising 0.1% (v/v) aqueous formic acid as solvent A and methanol as solvent B was used under gradient mode during 7.2 min of the run time. The elution conditions applied were as follows: 0.0–0.2 min 80% B, 0.2–6.2 min 85% B, 6.2–6.7 min 85% B and 6.7–7.2 min 80% B. The flow rate of the mobile phase was 0.6 mL/min and injection volume 10 μL. The eluent was monitored by electrospray ionization mass spectrometer (ESI–MS) under positive ion mode. The MS parameters were set as follows: capillary temperature of 650 °C, curtain gas at 35 psi, nebulizer gas at 50 psi and drying gas at 50 psi. 4500 V voltage was applied for positive ionization mode source. Nitrogen was used as curtain and collision gas. Data analysis was processed with Analyst 1.7.2 software.
Bacillus fermented extract analysis: surfactin’s quantitative analysis
Surfactin’s quantitative analysis was performed with the use of UHPLC-UV system produced by Waters (AcquityArc). Liquid samples after centrifugation were diluted to proper level with methanol, filtered through 0.22 µm nylon syringe filter and injected (10 µL). Detection was done with UV lamp set to 210 nm, and the column used for separation was Cortex C18 (4.6 × 50 mm, 2.7 µm). Mobile phases were water (A) and acetonitrile (B) (both with 0.1% addition of formic acid). The method was 10 min long with increasing B gradient form initial conditions (50% B). Quantitative analysis was done with external standard which was obtained from InventioBio SA biorefinery.
Determination of irritant potential: zein value
Irritant potential of the prototypes cosmetics was measured using zein test. The study was carried out using the automatic mineralization system Digestor 8AR and the automatic nitrogen analyzer Kjeltec 8400 (Producer FOSS, Denmark). In the zein test procedure, 2 g of protein was solubilized in 40 g solution of shower gel (10% wt.). The amount of solubilized protein was determined by Kjeldahl analysis, and the result of the zein value procedure was expressed as mg of solubilized protein (calculated as nitrogen) in 100 mL of sample. The final result was the arithmetic mean of three independent measurements. The testing methodology was described by Seweryn and Bujak (2018) and Wasilewski et al. (2016).
Evaluation of ability to emulsify fatty soils
The ability to emulsify fatty soils was evaluated in tests conforming to the PN-C-77003 standard. The test consisted in determining the maximum weight of rapeseed oil which can be emulsified by 1 L of a washing bath containing 1 wt % of the evaluated formulation. The final result (mean value of three independent measurements) obtained in the test determining the ability of the evaluated formulation to emulsify fatty soils was expressed in grams of oil per liter of the evaluated formulation at the concentration of 1 wt %. The test methodology was described by Seweryn and Bujak(2018).
Evaluation of the foaming properties
Foaming properties were tested according to PN-74 C-04801 standard. Foam height was measured using cylinder of the Ross–Miles apparatus. 10% aqueous solutions of tested preparations were investigated. An aliquot of 50 mL of the solution was poured into the cylinder and 200 mL of the solution into a dropper. The dropper was placed 1 m above the liquid level in the cylinder and the dropper tap was turned on. The height of the foam formed in the cylinder was measured after 1 and 10 min. The measurement over time was used to calculate the foam stability (Wasilewski et al. 2022b).
Viscosity measurements
The viscosity of the model shower gel formulations was measured at 20 °C in triplicate, using a Brookfield rheometer DV2TRV with a small sample adapter and cylindrical spindle SC4 (Brookfield, WI, USA). An aliquot of 8 mL of the sample was used in each test. Different shear rates and shear stresses were applied to the sample, and the resulting rheogram was constructed to determine the rheological behavior (Wasilewski et al. 2022b).
Determination of the color parameters
The cosmetics shower gel prototypes, with and without surfactins extracts, were subjected to reflectance color measurements using a Konica Minolta CM-3600 (Konica Minolta, Sensing Singapore Pte Ltd., Tokyo, Japan) with color data software CM-S100w, SpectraMagic NX, version 1.07 (Konica Minolta Sensing Europe B.V., Nieuwegein, the Netherlands) and light source D65, which represented a daylight phase. The CIE LAB system (defined by the International Commission on Illumination in 1978) organized the perception of the three-dimensional color space represented in rectangular coordinates: L*, a* and b*. The L* axis is associated with the lightness of the color: A value of 100 corresponds to white and a value of 0 to black. The axes a* and b* are associated with the changes of red–green and yellow–blue, respectively (+ a* correlates with red, − a* with green, + b* corresponds to yellow, − b* to blue). The chromaticity coordinates (a*, b*) are represented in polar coordinates by the chroma (C*), the Euclidian distance from the lightness axis and the hue angle (h*), expressed in degrees, starting from the positive a* axis (red) and turning in an anticlockwise direction.
The differences (\(\Delta\)E) between the color of the cosmetics with fermented extract and base cosmetic (without extracts) were calculated by the formula:
where \(\Delta\)L*, \(\Delta\)a* and \(\Delta\)b* are the mathematical differences between the cosmetics with extracts L*, a* and b* and the base cosmetic L*, a* and b* values.
Stability study
Stability was assessed under laboratory storage conditions. Samples were stored for 1 month under constant air temperature of 23 °C (air-conditioned room) and conditions of variable sunlight. In addition, the stability of the cosmetics was assessed with a mechanical loading test. After 24 h of detergent preparation, the samples were subjected to centrifugal force using a Rotofix 32 A (producer Zentrifugen) centrifuge. Samples were centrifuged at 2500 rpm for 15 min at room temperature. Microbiological pretesting was carried out. Microbiological stability of the shower gels was evaluated by tests using microcount® duo microbiological testers (Schülke & Mayr GmbH, Norderstedt, Germany). The test plates were stored at 28 °C, for 3 days for bacterial colony and fungi assays, and for 5 days for yeast and mold testing. After this time, the plates were visually evaluated and the number of microorganisms was determined, based on the test manufacturer’s template. They were tested for fungal and bacterial colonies and molds or yeasts.
Statistical analysis
For zein value, ability to emulsify fatty soils, foaming ability, foaming stability factor and viscosity data were reported as a mean ± standard deviation (SD), with 3 replicates for each sample (n = 3). For comparing the statistical significance of values, ANOVA and a Tukey HSD post hoc test were applied. Differences were considered significant with the p-value was < 0.05. The Statistica version 10 software (StatSoft, Tulsa, OK, USA) was used for calculations.
Results and discussion
Production of surfactin-rich digestate extract
Surfactin is a cyclic lipopeptide described as a powerful biosurfactant and is natively produced by Bacillus subtilis. It can be easily detected with UV lamp due to the presence of carbonyl chromophore (210 nm) in peptide ring. It can be successfully separated from other sample components with the use of C18 resin containing columns. Additionally the ease of injection preparation makes HPLC–UV the method of choice in surfactin qualitative and quantitative measurements in liquid samples. Surfactin is a group of structural analogues so for the quantitative analysis their peak’s areas were integrated and expressed as overall surfactin in sample (presented in Supplementary Material in Figure S1). During shake-flask fermentation process, Bacillus subtilis natto KB1 (which is wild, non-GMO strain) produces surfactin and secretes it to the external environment. Modified Landy medium and used parameters turned out to be most effective method to obtain good surfactin’s yield based on the previous research (Lewińska 2021). After 72 h of fermentation, obtained liquid broth was centrifuged to remove bacteria cells and HPLC–UV analysis showed the surfactin yield equal 2.21 g/L. Such solution was subjected to reversed osmosis (RO), but before that step ultrafiltration was necessary to remove all residual cells and solid impurities. Then reversed osmosis was performed with stable crossflow set to 0.5 L/min. After 30 min, the concentrated solution was isolated in which surfactin’s concentration achieved 14.9 g/L. It means that RO allowed sevenfold concentration. Then the solution was diluted with propanediol to produce final extract with 10 g/L of surfactin which was used for cosmetic formulations.
Confirmation of the presence of surfactin in the extract
Confirmation of surfactin was carried out through direct injection into an ESI–MS/MS in both negative and positive ionization modes. The fragmentation pattern by ESI–MS/MS analysis of recognized peaks was used to confirm the molecular identity. Analysis in positive mode revealed two set of specific m/z peaks. The first set of peaks belonged to the surfactin family, with strong signals at m/z 1016.6, 1030.5, 1044.5, 1058.5, 1072. 6 and 1086.5 Da corresponding to sodiated surfactin homologues pseudomolecular ions [M + Na]+. Second set of peaks corresponded to protonated pseudomolecular ions [M + H]+ of surfactin at m/z 994.5, 1008.6, 1022.5, 1036.5 and 1050.6 Da.
Negative analysis mode confirmed set of peaks belonged to the surfactin family, with strong signals of [M-H]− at m/z 992.6, 1006.6, 1020.7, 1034.7 and 1048.6 Da. The set of sodiated peaks [M + Na–H]− with signals at m/z 1014.6, 1028.6, 1042.6, 1056.6 and 1072.6 Da was also detected. The presence of sodiated pseudomolecular ions in negative mode indicated very strong cation chelation ability of surfactin.
Full scan in positive and negative mode revealed surfactin isomers peaks with a mass difference of 14 Da, corresponding to CH2 group. Moreover, protonated and sodiated surfactin’s homologs were fragmented by the collision-induced dissociation (CID)—MS/MS. Their detected amino acid sequences were as follows: glutamic acid–leucine–leucine–valine–aspartic acid–leucine–leucine, but differed in the fatty acid moiety, which consisted β-hydroxy fatty acids of different lengths: C12, C13, C14, C15 and C16. This could be verified by the base fragment ion, m/z 685 Da, common to each precursor ion, as it was a cleavage product of glutamic acid–leucine and fatty acids–leucine, with the net charge retained in the hexapeptide residue (leucine–leucine–valine–aspartic acid–leucine–leucine). Another abundant fragment, common to every species analyzed, was observed at m/z 441 Da. It was the cleavage product of the leucine–leucine–valine–aspartic acid tetrapeptide residue. The fragment ions of fatty acid in various detected precursor ions differed by 14 mass units (Da). For example, the fragment m/z 356.3 Da and its dehydrated form (m/z 338.3 Da) from the precursor ion at m/z 1022.5 Da were 14 Da smaller than their equivalents (m/z 370.26 and 352.26 Da) from the precursor ion with m/z 1036.5 Da.
The MS/MS data from the series of precursor ions at m/z 992.6, 1006.6, 1020.6 and 1034.7 Da in negative mode revealed the loss of Leu-Leu-Asp residues (m/z 339.2) from the C terminus generating a complementary lipopeptide fragment with values of m/z 650.4, 664.4, 678.5 and 692.4 Da, respectively. The loss of the β-hydroxy fatty acid from the resulting lipopeptide chain was evidenced by the fragment ion m/z 452.3 (Glu-Leu-Leu-Val) and a further loss of glutamate from the N terminus by the fragment ion m/z 323.2 Da (Leu-Leu-Val).
The corresponding sodiated pseudomolecular ions [M + Na+-H+]− at m/z 672.5, 686.4, 700.4 and 714 Da was also indicated with difference in product ion with a m/z of 23 Da. The set of mass peaks, with an interval of 14 Da, proved the presence of surfactin as a mixture of lipopeptide homologues with different length acyl chain corresponding to the number of carbon atoms: C12, C13, C14, C15 and C16. (Table 1). Chromatogram of separated surfactin homologues is presented in Fig. 1. The mass spectra obtained in positive and negative mode are presented in Supplementary Material in Figure S2 and S3, respectively.
Formulation and manufacturing technology of cosmetic prototypes
For the purpose of the study, shower gel formulations were developed based on ingredients listed in Table 2. The variable analyzed in the product prototypes was the concentration of the digestate extract.
The base (C1) for the shower gel production with digestate extract consists of (water, disodium cocoyl glutamate—6% w/w, xanthan gum—1% w/w and propanediol) in the proportion described in Table 2. Our new product was composed on the base and addition of 4% w/w (C2), 40% w/w (C3) and 80%w/w (C4) (Table 2). In addition to C1 (bases), water and propanediol was added to maintain the appropriate proportions of compounds used to produce gels.
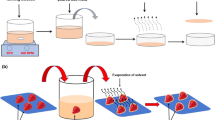
The cosmetic prototypes were obtained using an MZUTL 5 homogenizing mixer (Urlinski, Warsaw, Poland). A total of 5 L of cosmetic formulation was obtained per single batch. The digestate extract and water were introduced into the mixer. Agitation (50 rpm) and heating (1 °C/min) were started. The contents were heated to 40 °C. Other ingredients were added in a sequence specified in the formula. The components were mixed until a homogeneous mixture was obtained. To maintain a constant concentration of propanediol in each sample (due to its presence in the digestate extract), it was added to the prototypes, as necessary, to achieve a constant concentration of 20%. The variable analyzed in the study was the concentration of surfactin in the digestate extract. A polymeric thickener (xanthan gum) was incorporated into the formulation by dispensing it onto the surface of the liquid during agitation. The ingredients were mixed until a clear homogeneous solution was obtained. In the final step, the pH was adjusted with lactic acid to a value of ~ 5.5.
The samples were left to stand for 24 h and stored at room temperature until the system was completely deaerated. Their stability was then tested. The formulated shower gel prototypes exhibited full physicochemical and microbiological stability during the period of sample storage under laboratory conditions: mechanical loading test and preliminary microbiological tests. Preliminary tests found that the products did not require any conventional (commonly used) preservative systems owing to the presence of propanediol in the formulations. Consequently, the prototypical shower gels were not stabilized with any preservatives. The main task of the preservative is to keep the product in the same microbiological purity in which it was produced. Unfortunately preservatives are known to cause physical instability in dispersed systems. Additionally, as usually they are toxic, irritating and allergenic, modern and ecological cosmetics are looking for components with antimicrobial functions (Nowak et al. 2021; Herman 2019; Dréno et al. 2019; Halla et al. 2018). Ferment from biotechnological processes are rich in natural substances with these properties (Majchrzak et al. 2022; Lee et al. 2020; Herman 2019). The surfactin-rich ferment used in our research was enriched with propanediol, which is an emerging class of multifunctional—ingredients that besides antimicrobial properties (Herman 2019; Kerdudo et al. 2015), possess skin-caring properties (Tse et al. 2021). In addition, the surfactin found in the extract also has antimicrobial properties. Previous studies have described its antimicrobial activities against various pathogenic bacteria, such as Enterococcus faecalis, Staphylococcus areus and Pseudomonas aeruginosa (Chen et al. 2022; Fernandes et al. 2007). However, recent studies have questioned the general claim of antimicrobial properties of surfactin (Lilge et al. 2022).
Safety assessment of prototype cosmetics
Eco-friendly approach to the development of cosmetics requires the pursuit of sustainability goals which involve primarily the use of naturally derived ingredients in the process of cosmetic product formulation. Efforts to develop cosmetics that are consistent as closely as possible with the concept of sustainability should also take into account the aspect of safety in use and, more specifically, the potential impact of the product on the consumer (Dini et al. 2021; Boom et al. 2019, Barbulova et al. 2015). Cosmetics based on raw materials of natural origin are considered to be fully safe (Fonseca-Santos et al. 2015) and within the territory of the European Union, all marketed cosmetics are subject to a rigorous safety assessment in accordance with the legislation in force (Pauwels and Rogiers 2010). Even though commercially available products are certified as safe, there are occasional reports of adverse reactions experienced by consumers after using them. These include skin irritation (Mijaljica et al. 2022; Seweryn 2018; Ananthapadmanabhan et al. 2004), contact allergies (Alani et al. 2013), sensation of dry skin or other intolerances associated with the contact of a given cosmetic with the skin (Mijaljica et al. 2022; Seweryn 2018; Ananthapadmanabhan et al. 2004).
As part of the study, the formulated prototypes of cleansing cosmetics were analyzed with regard to the risk of skin irritation following their application. The skin irritation effect of the cosmetic prototypes was evaluated with a method based on the zein value (ZV) which, according to state-of-the-art knowledge, is a measure of the irritant effect induced by cleansing cosmetics on the skin (Cohen et al. 2016; Wardet al. 1998; Pezron et al. 1996). The results are shown in Fig. 2.
The analyzed prototypes of cleansing cosmetics are characterized by relatively low ZV values, ranging from 15 to 80 mg N/100 mL. Based on the literature, these values indicate that the prototypical cosmetics have no skin irritation properties (Pezron et al. 1996). It is important to note that, compared to the reference formulation C1, an increase in the concentration of the surfactin-rich digestate extract in the cleansing cosmetic prototypes led to a significant decrease in the ZV value. The lowest ZV, i.e., 15 mg N/100 mL, was obtained for the sample C4 containing the studied extract at the highest concentration. The decrease in the value of the zein number, and hence the probability of skin irritation developing after the application of the cosmetic prototypes on the skin, is linked to the use of the surfactin-rich digestate extract in the formulation. The irritant effect of cleansing cosmetics is associated predominantly with potential electrostatic interactions between the surfactants present in the form of monomers and the proteins building the stratum corneum. In addition, surfactants may potentially interfere with the liquid crystalline structure of the intercellular cement in the stratum corneum, which may damage the stratum corneum and impair the epidermal barrier. This type of action is mainly attributed to ionic (including anionic) surfactants, which are the primary ingredients in cleansing cosmetics. The established methods for reducing the irritant effect of cosmetics formulated with anionic surfactants mostly involve efforts to stabilize the micellar structure of surfactants in the bulk phase, increasing the size of aggregates forming in the solution or binding surfactant monomers in the surfactant–polymer system (Morris et al. 2022, 2019; Seweryn et al. 2018). In practice, it is possible to reduce the skin irritation effect of anionic surfactants by adding to the formulation anionic surfactants with proven low irritant potential (Seweryn and Bujak 2018; Klimaszewska et al. 2017), another type of surfactant (nonionic or amphoteric) (Klimaszewska et al. 2022), macromolecular compounds (including polymers or protein hydrolysates) (Bujak et al. 2015), hydrophobic substances (Wasilewski et al. 2016; Mukherjee et al. 2015) or plant-based extracts (Nizioł-Łukaszewska 2017).
The solution proposed by the authors was aimed at achieving a reduction in the irritant effect of the formulation through the use of a surfactin-rich digestate extract. In addition to containing a significant proportion of a biosurfactant, this raw material is a source of many cosmetologically valuable ingredients, including proteins, sugars, mineral salts and small amounts of hydrophobic components. Both surfactin, which has been shown in the literature to have a low skin irritation potential (Fei et al. 2020; Vecino et al. 2017), and the remaining ingredients may contribute to a significant decrease in the irritant effect of the entire cosmetic formulation by various mechanisms (increase in micelle size and stability, formation of complexes between macromolecular substances and surfactants leading to the binding of free monomers in the bulk phase). This effect was observed in the studies performed for the formulated cosmetic prototypes. Despite the addition of surfactants (in the form of surfactin in the digestate extract) into the formulation, a decrease in the irritant effect was noted, even though an increase in the concentration of conventional surfactants in aqueous solutions normally amplifies the irritant effect (Seweryn 2018). The observed decrease in the zein number, corresponding to a reduction in the skin irritation effect of cosmetics, is consistent with the findings of another study conducted by the Authors (Wasilewski et al. 2022a) to determine the possible applications of a levan-rich digestate extract. The study found that a decrease in the skin irritation potential of cosmetics is most likely attributable to the levan–surfactant interactions, the formation of levan–surfactant complexes and the presence of other ingredients with important cosmetic properties.
Evaluation of functional properties of prototype cosmetics
In addition to aspects related to safety in use, and specifically the potential to cause skin irritation and drying, comprehensive evaluation of the quality of cleansing cosmetics by consumers also includes product properties associated with its intended function. Cleansing cosmetics are expected to deliver appropriate washing performance and good foaming properties (Seweryn and Bujak 2018; Seweryn et al. 2018). The formulated prototypes of cleansing cosmetics were evaluated to determine their ability to emulsify fatty soils and foaming capacity. The results are shown in Figs. 3 and 4.
Cosmetics are subject to rigorous quality assessment by consumers. With respect to body wash cosmetics, consumers expect not only appropriate performance related specifically to their intended purpose, but also specific features that prevent excessive skin drying after the process of skin washing. The skin drying effect which may be observed after the application of cleansing cosmetics is attributed to excessive removal of fatty components from the skin’s surface. This effect is closely linked to the skin washing process and the ability of the washing bath to emulsify and solubilize hydrophobic components from the skin. The greatest risks are associated with the extraction of components of the intercellular cement of the stratum corneum (Morris et al. 2022; Ananthapadmanabhan et al. 2013; Corazza et al. 2010), changes in the pH of the skin due to disturbances of the bacterial flora and disruptions affecting the hydrolipidic film and the components of the natural moisturizing factor (Mijaljica et al. 2022; Ananthapadmanabhan et. al. 2013; Ananthapadmanabhan et al. 2004). These processes may damage the integrity of the epidermal barrier, leading to excessive water loss from the epidermis and causing epidermal dryness (Green et al. 2022; Lukić et al. 2021; Ananthapadmanabhan et al. 2013; Corazza et al. 2010). The ability of cleansing cosmetics to emulsify fatty soils can be evaluated both as a parameter of product functionality and as a measure of safety, related to the potential negative impact of such cosmetics on the skin. Emulsification is one of the sub-steps of the body washing process that allows effective removal of soiling from the surface of the skin. By determining the value of this parameter, it is possible to evaluate the washing performance of cleansing cosmetics (Seweryn and Bujak 2018). On the other hand, excessively powerful emulsification of hydrophobic soiling from the surface of the skin may lead to the leaching of valuable lipid components from the epidermis. The values describing the ability of the prototypical cleansing cosmetics to emulsify fatty soils were found to be in the range of 15–11 g/L. The parameter reached the highest value in the reference formulation C1, while the lowest value was detected in the prototype C4 formulated with the highest evaluated concentration of the digestate extract. As the results show, incorporating the digestate extract into the cosmetic composition leads to a decrease in the evaluated parameter, which may have an impact on the performance of the cosmetic in terms of its intended use (washing effect). He developed cleansing formulations contain mild anionic surfactant disodium cocoyl glutamate which is known to be gentle for skin. The favorable safety profile (mildness/non-irritancy) of surfactin-rich shower gels could be probably attributed to their decreased ability to emulsify hydrophobic substances leading to the reduced skin drying upon cleansing process (which also stands for reference sample), but even more importantly to the mechanisms/substances responsible for the decrease in skin irritation potential (zein value).
The ability of body wash cosmetics to generate foam is an important factor contributing to consumers' assessment of product quality. In the opinion of consumers, the washing bath should preferably generate copious amounts of foam exhibiting desirable sensory properties (thick and velvety) and stability. Crucially, foaming ability is a cosmetic parameter that does not translate in any way into the performance of cosmetic products. Its sole function is to enhance the consumer's experience during the washing process. The results of the foaming ability test performed for the prototypical cleansing cosmetics containing the surfactin-rich extract are shown in Fig. 4.
Foam volumes generated by the studied prototypical cosmetics ranged between 280 and 360 cm3. Compared to the reference formulation C1, a decrease in the evaluated parameter is noted as the concentration of the extract added to the formulation increases. The lowest foaming ability was determined for the cosmetic prototype C4 containing the extract at a concentration of 80%. Evaluation of foam stability showed a similar relationship, with a decrease in foam stability (up to 62%) observed solely in the formulation C4. For the remaining prototypes evaluated in the study, similar stability values as in the reference sample C1 were determined (85%).
In cleansing cosmetics, viscosity is another relatively important parameter contributing to perceived product quality. In industrial practice, the adjustment of this parameter in cosmetics of this type, which are surfactant-based systems, is typically accomplished through the addition of sodium chloride or polymeric rheology modifier. Obtaining the desired viscosity, especially in shower gels, is a prerequisite for positive consumer experience in terms of product dispensation from the package, spreading it on the skin and combining with water during the washing process. The rheological properties of cosmetics also have a bearing on their environmental safety. If cosmetic products are difficult to dispense, considerable quantities may stick to the walls of the package, reducing the efficiency of product use. What follows is that such cosmetics should be susceptible to shearing forces (package squeezing and shaking) that temporarily decrease the viscosity of products, thus facilitating their skin application (Wasilewski et al. 2022a; Gallegos and Franco 1999; Wasilewski and Seweryn 2016). The rheological behavior of model shower gels is shown in Fig. 5.
Viscosity and shear stress showed a nonlinear trend with increasing shear rate, indicating non-Newtonian behavior of all the model shower gels tested. It was observed that the apparent viscosity of the tested cosmetics prototypes decreased gradually with increasing shear rate value, which characterizes the shear-thinning pseudoplastic materials. The apparent viscosity increased with increasing surfactin digest content. However, the change in apparent viscosity at the same shear rate (in the range of 0.1 to 50 s−1) between samples was not that significant (p > 0.05) The shear stress increases continuously with shear rate for all the samples. The shear-thinning behavior of investigated shower gel prototypes was fitted to the power-law model (Eq. 4).
where η is the apparent viscosity (mPa), γ̇ is the shear rate (s−1), n is the flow behavior index (dimensionless) and K is the consistency index (Pa·sn). For pseudoplastic fluids, n < 1 (Fan et al. 2022, García-Ochoa et al. 2000, Rottava et al. 2009). Table 3 shows the parameters of the power-law model for investigated shower gel prototypes.
When it comes to cleansing cosmetics, the purchasing decisions of consumers are determined to a major extent by their visual perceptions (Amberg and Fogarassy 2019). This is why most conventional cosmetics of this type are formulated with the addition of specially selected dyes that add color to the product to make it appear more visually appealing to consumers. Additives of this type are usually synthetic raw materials of organic origin, whose safety is increasingly being called into question (Guerra et al. 2018). The trend not to use dyes is particularly evident in cosmetics that are marketed with the claims of being natural. This is motivated by the growing number of reports concerning adverse impacts of these ingredients on human health and symptoms of intolerance experienced by consumers. Cases involving various skin effects such as contact allergy have been reported (Borowska and Brzóska 2015). Consequently, following the trend toward naturalness, most natural cosmetics are free of such additives, and their color is derived from the raw materials used in the manufacturing process. The prototypical shower gels analyzed in the present study are dye-free and their color, as perceived by consumers, depends on the raw materials used. For the Authors, it was particularly important to establish the effects of the surfactin-rich digestate extract on the visual qualities of the formulated prototypes. To evaluate this aspect, an appropriate study was carried out, with the findings reported below. To determine the color difference of the tested model cosmetics, colorimetric assays were performed, and the results obtained are presented in Table 4.
The addition of surfactin-rich digest extract changed the appearance of the base cosmetic from colorless to colored products. In addition, the comparison of the L*, a* and b* values indicated that the concentration of extract differentiated the color of cosmetics prototypes.
A cosmetic without extract was characterized by the highest L* value and the lowest a* and b* values. As concentration of digest extracts increased, the L* values decreased, but a* and b* increased indicating that the products became more red and more yellow with increasing concentration. Moreover, the chroma (C*) values increased with extract concentration, demonstrating a significant change in the color saturation of the model cosmetics with the addition of the fermented extract to the base sample (without adding of the extracts) and with the increase in its concentration in the products prototypes. The changes in Ho values between the cosmetics prototypes indicated a change the color tonality in relation to the extract concentration. The calculated color difference, ΔE, compared to the C1 base cosmetic, showed good coloring power for C2, C3 and C4 products, with the highest ΔE value obtained for C4 cosmetics with the highest concertation of surfactin-rich digest extract. As indicated by literature data (Wan et al. 2019; Mokrzycki and Tatol 2011) if ΔE values are higher than 5, the color is perceived by the naked eye and perceived as a color effect.
The color differences between the cosmetics prototypes were clearly visible, with ΔE values of 6,18; 26.97 and 37.91 for C2, C3 and C4, respectively. Figure 6 shows the appearance of the shower gel prototypes.
Conclusions
In line with the concept of sustainable development and new trends in the cosmetics industry, the underlying assumption of the study was to formulate and produce fully functional shower gels based on ingredients of natural origin. Special attention was paid to the possibility of using a surfactin-rich digestate extract as an all-natural raw material obtained through a biotechnological process. To this end, prototypical shower gels were designed and formulated, and subsequently studied to assess their quality. The study results showed that the formulated cosmetic prototypes exhibited satisfactory quality characteristics. The digestate extract incorporated into the formulations was not found to impair any cosmetic quality parameters related to functionality. All the parameters evaluated in the formulated shower gels (viscosity, foaming properties and ability to emulsify fats) reached similar values as in the reference formulation containing no extract. The analysis of parameters related to the safety in use or, more specifically, the potential for skin irritation, none of the shower gels showed any irritant effect. The addition of the extract to the product formulation causes a significant (ca. 70%) decrease in the zein value, which is a measure of the severity of skin irritation effects. Colorimetric analysis of the formulated shower gels demonstrates that the addition of the surfactin-rich digestate extract changes the color of the samples toward the natural hue derived from the raw material used.-Consequently, there is no need to incorporate dyes into cosmetic formulations. The studies showed that it was feasible to formulate cleansing cosmetics consistent with the concept of sustainability based on ingredients of natural origin, including raw materials obtained through biotechnological processes, offering appropriate functional properties and highly safe to use in terms of their effects on users’ skin.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Akbari S, Abdurahman NH, Yunus RM, Fayaz F, Alara OR (2018) Biosurfactants—a new frontier for social and environmental safety: a mini review. Biotechnol Res Innov 2:81–90. https://doi.org/10.1016/j.biori.2018.09.001
Alani JI, Davis MDP, Yiannias JA (2013) Allergy to cosmetics: a literature review. Dermatitis 24:283–290. https://doi.org/10.1097/DER.0b013e3182a5d8bc
Amberg N, Fogarassy C (2019) Green consumer behavior in the cosmetics market. Resources 8:137. https://doi.org/10.3390/resources8030137
Ananthapadmanabhan KP, Moore DJ, Subramanyan K, Misra M, Meyer F (2004) Cleansing without compromise: the impact of cleansers on the skin barrier and the technology of mild cleansing. Dermatol Ther 17:16–25. https://doi.org/10.1111/j.1396-0296.2004.04S1002.x
Ananthapadmanabhan KP, Mukherjee S, Chandar P (2013) Stratum corneum fatty acids: their critical role in preserving barrier integrity during cleansing. Int J Cosmet Sci 35:337–345. https://doi.org/10.1111/ics.12042
Antignac E, Nohynek GJ, Re T, Clouzeau J, Toutain H (2011) Safety of botanical ingredients in personal care products/cosmetics. Food Chem Toxicol 49:324–341. https://doi.org/10.1016/j.fct.2010.11.022
Barbulova A, Colucci G, Apone F (2015) New trends in cosmetics: by-products of plant origin and their potential use as cosmetic active ingredients. Cosmetics 2:82–92. https://doi.org/10.3390/cosmetics2020082
Bezerra KGO, Rufino RD, Luna JM, Sarubbo LA (2018) Saponins and microbial biosurfactants: potential raw materials for the formulation of cosmetics. Biotechnol Prog 34:1482–1493. https://doi.org/10.1002/btpr.2682
Bjerk TR, Severino P, Jain S, Marques C, Silva AM, Pashirova T, Souto EB (2021) Biosurfactants: properties and applications in drug delivery. Biotechnol Ecotoxicol Bioeng 8:115. https://doi.org/10.3390/bioengineering8080115
Bom S, Jorge J, Ribeiro HM, Marto J (2019) A step forward on sustainability in the cosmetics industry: a review. J Clean Prod 225:270–290. https://doi.org/10.1016/j.jclepro.2019.03.255
Borowska S, Brzóska MM (2015) Metals in cosmetics: implications for human health. J Appl Toxicol 35:551–572. https://doi.org/10.1002/jat.3129
Bujak T, Wasilewski T, Nizioł-Łukaszewska Z (2015) Role of macromolecules in the safety of use of body wash cosmetics. Colloids Surf B Biointerfaces 135:497–503. https://doi.org/10.1016/j.colsurfb.2015.07.051
Chen X, Lu Y, Shan M, Zhao H, Lu Z, Lu Y (2022) A mini-review: mechanism of antimicrobial action and application of surfactin. World J Microbiol Biotechnol 38(8):143. https://doi.org/10.1007/s11274-022-03323-3
Cohen L, Martin M, Soto F, Trujillo F, Sanchez E (2016) The effect of counterions of linear alkylbenzene sulfonate on skin compatibility. J Surf Det 19:219–222. https://doi.org/10.1007/s11743-015-1765-8
Corazza M, Lauriola MM, Zappaterra M, Bianchi A, Virgili A (2010) Surfactants, skin cleansing protagonists. J Eur Acad Dermatol Venereol 24:1–6. https://doi.org/10.1111/j.1468-3083.2009.03349.x
Dini I, Laneri S (2021) The new challenge of green cosmetics: natural food ingredients for cosmetic formulations. Molecules 26:3921. https://doi.org/10.3390/molecules26133921
Domżał-Kędzia M, Lewińska A, Jaromin A, Weselski M, Pluskota R, Łukaszewicz M (2019) Fermentation parameters and conditions affecting levan production and its potential applications in cosmetics. Bioorg Chem 93:102787. https://doi.org/10.1016/j.bioorg.2019.02.012
Dréno B, Zuberbier T, Gelmetti C, Gontijo G, Marinovich M (2019) Safety review of phenoxyethanol when used as a preservative in cosmetics. J Eur Acad Dermatol Venereol 33:15–24. https://doi.org/10.1111/jdv.15944
Fan Y, Zhang L, Wang X, Wang K, Wang L, Wang Z, Xue F, Zhu J, Wang C (2022) Rheological thixotropy and pasting properties of food thickening gums orienting at improving food holding rate. Appl Rheol 32(1):100–121. https://doi.org/10.1515/arh-2022-0127
Fei D, Zhou GW, Yu ZQ, Gang HZ, Liu JF, Yang SZ, Ye RQ, Mu BZ (2020) Low-toxic and nonirritant biosurfactant surfactin and its performances in detergent formulations. J Surf Deterg 23:109–118. https://doi.org/10.1002/jsde.12356
Fernandes PAV, Arruda IRD, Santos AFABD, Araújo AAD, Maior AMS, Ximenes EA (2007) Antimicrobial activity of surfactants produced by Bacillus subtilis R14 against multidrug-resistant bacteria. Braz J Microbiol 38(4):704–709. https://doi.org/10.1590/S1517-83822007000400022
Ferreira A, Vecino X, Ferreira D, Cruz JM, Moldes AB, Rodrigues LR (2017) Novel cosmetic formulations containing a biosurfactant from Lactobacillus paracasei. Colloids Surf B Biointerfaces 155:522–529. https://doi.org/10.1016/j.colsurfb.2017.04.026
Fonseca-Santos B, Corrêa MA, Chorilli M (2015) Sustainability, natural and organic cosmetics: consumer, products, efficacy, toxicological and regulatory considerations. Braz J Pharm Sci 51:17–26. https://doi.org/10.1590/S1984-82502015000100002
Furtado IF, Sydney EB, Rodrigues SA, Sydney AC (2022) Xanthan gum: applications, challenges, and advantages of this asset of biotechnological origin. Biotechnol Res Innov J 6:e202204. https://doi.org/10.4322/biori.202205
Gallegos C, Franco JM (1999) Rheology of food, cosmetics and pharmaceuticals. Curr Opin Colloid Interface Sci 4:288–293. https://doi.org/10.1016/S1359-0294(99)00003-5
Ganesan NG, Rangarajan V (2021) A kinetics study on surfactin production from Bacillus subtilis MTCC 2415 for application in green cosmetics. Biocatal Agric Biotechnol 33:102001. https://doi.org/10.1016/j.bcab.2021.102001
García-Ochoa F, Santos VE, Casas JA, Gómez E (2000) Xanthan gum: production, recovery, and properties. Biotechnol Adv 18:549–579. https://doi.org/10.1016/S0734-9750(00)00050-1
Goyal N, Jerold F (2021) Biocosmetics: technological advances and future outlook. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-17567-3
Green M, Feschuk AM, Kashetsky N, Maibach HI (2022) “Normal” TEWL-how can it be defined? A systematic review. Exp Dermatol 31:1618–1631. https://doi.org/10.1111/exd.14635
Guerra E, Llompart M, Garcia-Jares C (2018) Analysis of dyes in cosmetics: challenges and recent developments. Cosmetics 5:47. https://doi.org/10.3390/cosmetics5030047
Halla N, Fernandes IP, Heleno SA, Costa P, Boucherit-Otmani Z, Boucherit K, Rodrigues AE, Ferreira ICFR, Barreiro MF (2018) Cosmetics preservation: a review on present strategies. Molecules 23:1571. https://doi.org/10.3390/molecules23071571
Herman A (2019) Antimicrobial ingredients as preservative booster and components of self-preserving cosmetic products. Curr Microbiol 76:744–754. https://doi.org/10.1007/s00284-018-1492-2
Jahan R, Bodratti AM, Tsianou M, Alexandridis P (2020) Biosurfactants, natural alternatives to synthetic surfactants: physicochemical properties and applications. Adv Colloid Interface Sci 275:102061. https://doi.org/10.1016/j.cis.2019.102061
Kapoor R, Singh AB, Misra R (2019) Green cosmetics—Changing young consumer preference and reforming cosmetic industry. Int J Recent Technol Eng 8:12932–12939. https://doi.org/10.35940/ijrte.D6927.118419
Kerdudo A, Fontaine-Vive F, Dingas A, Faure C, Fernandez X (2015) Optimization of cosmetic preservation: water activity reduction. Int J Cosmet Sci 37:31–40. https://doi.org/10.1111/ics.12164
Klimaszewska E, Wieczorek D, Lewicki S, Stelmasiak M, Ogorzałek M, Szymański Ł, Tomasiuk R, Markuszewski L (2022) Effect of new surfactants on biological properties of liquid soaps. Molecules 27:5425. https://doi.org/10.3390/molecules27175425
Klimaszewska E, Seweryn A, Czerwonka D, Piotrowska U, Ogorzalek M (2017) Improvement of the safety in use of babies cosmetics through appropriate selection of surfactants. Przem Chem 96:2509–2513. https://doi.org/10.15199/62.2017.12.23
Kolling C, Ribeiro JLD, de Medeiros JF (2022) Performance of the cosmetics industry from the perspective of corporate social responsibility and design for sustainability. Sustain Prod Consum 30:171–185. https://doi.org/10.1016/j.spc.2021.12.002
Lee S, Lee J, Yu H, Lim J (2018) Synthesis of environment friendly biosurfactants and characterization of interfacial properties for cosmetic and household products formulations. Colloids Surf A 536:224–233. https://doi.org/10.1016/j.colsurfa.2017.05.001
Lee SW, Lim JM, Mohan H, Seralathan KK, Park YJ, Lee JH, Oh BT (2020) Enhanced bioactivity of Zanthoxylum schinifolium fermented extract: anti-inflammatory, anti-bacterial, and anti-melanogenic activity. J Biosci Bioeng 129:638–645. https://doi.org/10.1016/j.jbiosc.2019.12.003
Lewińska A (2021) Optimizing the process design of oil-in-water nanoemulsion for delivering poorly soluble cannabidiol oil. Processes 9:1180. https://doi.org/10.3390/pr9071180
Lewińska A, Domżał-Kędzia M, Kierul K, Bochynek M, Pannert D, Nowaczyk P, Łukaszewicz M (2021) Targeted hybrid nanocarriers as a system enhancing the skin structure. Molecules 26:1063. https://doi.org/10.3390/molecules26041063
Lilge L, Ersig N, Hubel P, Aschern M, Pillai E, Klausmann P, Pfannstiel J, Henkel M, Morabbi Heravi K, Hausmann R (2022) Surfactin shows relatively low antimicrobial activity against Bacillus subtilis and other bacterial model organisms in the absence of synergistic metabolites. Microorganisms 10(4):779. https://doi.org/10.3390/microorganisms10040779
Lourith N, Kanlayavattanakul M (2009) Natural surfactants used in cosmetics: glycolipids. Int J Cosmet Sci 31:255–261. https://doi.org/10.1111/j.1468-2494.2009.00493.x
Luft L, Confortin TC, Todero I, Zabot GL, Mazutti MA (2020) An overview of fungal biopolymers: bioemulsifiers and biosurfactants compounds production. Crit Rev Biotechnol 40:1059–1080. https://doi.org/10.1080/07388551.2020.1805405
Lukić M, Pantelić I, Savić SD (2021) Towards optimal ph of the skin and topical formulations: from the current state of the art to tailored products. Cosmetics 8:69. https://doi.org/10.3390/cosmetics8030069
Majchrzak W, Motyl I, Śmigielski K (2022) Biological and cosmetical importance of fermented raw materials: an overview. Molecules 27:4845. https://doi.org/10.3390/molecules27154845
Mani I (2020) Microbial production of vitamins. In: Singh V, Singh A, Bhargava P, Joshi M, Joshi CM (eds) Engineering of microbial biosynthetic pathways. Springer, Singapore, pp 143–152
Mathur H, Beresford TP, Cotter PD (2020) Health benefits of lactic acid bacteria (LAB) fermentates. Nutrients 12:1679. https://doi.org/10.3390/nu12061679
Mijaljica D, Spada F, Harrison IP (2022) Skin cleansing without or with compromise: soaps and syndets. Molecules 27:2010. https://doi.org/10.3390/molecules27062010
Mokrzycki WS, Tatol M (2011) Colour difference DE—a survey. Mach Graph vis 20:383
Morris SA, Thompson RT, Glenn RW, Ananthapadmanabhan KP, Kasting GB (2019) Mechanisms of anionic surfactant penetration into human skin: investigating monomer, micelle and submicellar aggregate penetration theories. Int J Cosmet Sci 41:55–66. https://doi.org/10.1111/ics.12511
Morris SA, Kasting GB, Ananthapadmanabhan KP (2022) Surfactant equilibria and its impact on penetration into stratum corneum. Curr Opin Colloid Interface Sci 59:101579. https://doi.org/10.1016/j.cocis.2022.101579
Mukherjee S, Yang L, Vincent C, Lei X, Ottaviani MF, Ananthapadmanabhan KP (2015) A comparison between interactions of triglyceride oil and mineral oil with proteins and their ability to reduce cleanser surfactant-induced irritation. Int J Cosmet Sci 37:371–378. https://doi.org/10.1111/ics.12205
Nizioł-Łukaszewska Z, Bujak T (2018) Saponins as natural raw materials for increasing the safety of bodywash cosmetic use. J Surf Det 21:767–776. https://doi.org/10.1002/jsde.12168
Nizioł-Łukaszewska Z, Osika P, Wasilewski T, Bujak T (2017) Hydrophilic dogwood extracts as materials for reducing the skin irritation potential of body wash cosmetics. Molecules 22:320. https://doi.org/10.3390/molecules22020320
Nohynek GJ, Antignac E, Re T, Toutain H (2010) Safety assessment of personal care products/cosmetics and their ingredients. Toxicol Appl Pharmacol 243:239–259. https://doi.org/10.1016/j.taap.2009.12.001
Nowak K, Jabłońska E, Ratajczak-Wrona W (2021) Controversy around parabens: alternative strategies for preservative use in cosmetics and personal care products. Environ Res 198:110488. https://doi.org/10.1016/j.envres.2020.110488
Pauwels M, Rogiers V (2010) Human health safety evaluation of cosmetics in the EU: a legally imposed challenge to science. Toxicol Appl Pharmacol 243:260–274. https://doi.org/10.1016/j.taap.2009.12.007
Pezron I, Galet L, Clausse D (1996) Surface interaction between a protein monolayer and surfactants and its correlation with skin irritation by surfactants. J Colloid Interface Sci 180:285–289. https://doi.org/10.1006/jcis.1996.0301
Rincón-Fontán M, Rodríguez-López L, Vecino X, Cruz JM, Moldes AB (2018) Design and characterization of greener sunscreen formulations based on mica powder and a biosurfactant extract. Powder Technol 327:442–448. https://doi.org/10.1016/j.powtec.2017.12.093
Rottava I, Batesini G, Silva MF, Lerin L, de Oliveira D, Padilha FF, Toniazzo G, Mossi A, Cansian RL, Luccio M, Treichel H (2009) Xanthan gum production and rheological behavior using different strains of Xanthomonas sp. Carbohyd Polym 77:65–71. https://doi.org/10.1016/j.carbpol.2008.12.001
Savic SM, Cekic ND, Savic SR, Ilic TM, Savic SD (2021) ‘All‐natural’anti‐wrinkle emulsion serum with Acmella oleracea extract: a design of experiments (DoE) formulation approach, rheology and in vivo skin performance/efficacy evaluation. Int J Cosmet Sci 43:530–546. https://doi.org/10.1111/ics.12726
Seweryn A (2018) Interactions between surfactants and the skin – theory and practice. Adv Colloid Interface Sci 256:242–255. https://doi.org/10.1016/j.cis.2018.04.002
Seweryn A, Bujak T (2018) Application of anionic phosphorus derivatives of alkyl polyglucosides for the production of sustainable and mild body wash cosmetics. ACS Sustain Chem Eng 6:17294–17301. https://doi.org/10.1021/acssuschemeng.8b04711
Seweryn A, Wasilewski T, Bocho–Janiszewska A (2018) Correlation between sequestrant type and properties of mild soap-based hand washing products. Ind Eng Chem Res 57:12683–12688. https://doi.org/10.1021/acs.iecr.8b02910
Sikora E, Michorczyk P, Olszańska M, Ogonowski J (2015) Supercritical CO 2 extract from strawberry seeds as a valuable component of mild cleansing compositions. Int J Cosmet Sci 37:574–578. https://doi.org/10.1111/ics.12234
Singh RD, Kapila S, Ganesan NG, Rangarajan V (2022) A review on green nanoemulsions for cosmetic applications with special emphasis on microbial surfactants as impending emulsifying agents. J Surf Deterg 25:303–319. https://doi.org/10.1002/jsde.12571
Sirilun S, Sivamaruthi BS, Kumar N, Kesika P, Peerajan S, Chaiyasut C (2016) Lactobacillus fermented plant juice as a potential ingredient in cosmetics: formulation and assessment of natural mouthwash. Asian J Pharm Clin Res 9(Suppl 3):52–56. https://doi.org/10.22159/ajpcr.2016.v9s3.14707
Sivamaruthi BS, Chaiyasut C, Kesika P (2018) Cosmeceutical importance of fermented plant extracts: a short review. Int J Appl Pharm 10:31–34. https://doi.org/10.22159/IJAP.2018V10I4.26355
Tse TJ, Nelson FB, Reaney MJ (2021) Analyses of commercially available alcohol-based hand rubs formulated with compliant and non-compliant ethanol. Int J Environ Res Public Health 18:3766. https://doi.org/10.3390/ijerph18073766
Vecino X, Cruz JM, Moldes AB, Rodrigues LR (2017) Biosurfactants in cosmetic formulations: trends and challenges. Crit Rev Biotechnol 37:911–923. https://doi.org/10.1080/07388551.2016.1269053
Velamakanni RP, Sree BS, Vuppugalla P, Velamakanni RS, Merugu R (2022) Biopolymers from microbial flora. In: Nadda AK, Sharma S, Bhat R (eds) Biopolymers. Springer Series on Polymer and Composite Materials. Springer, Cham, pp 151–172
Wan H, Yu Ch, Yu H, Guo X, Luo L, Pan H, Zheng T, Wang J, Cheng T, Zhang Q (2019) Determination of flavonoids and carotenoids and their contributions to various colors of rose cultivars (Rosa spp.). Front Plant Sci 10:1–14. https://doi.org/10.3389/fpls.2019.00123
Ward RK, Hubbard AW, Sulley H, Garle MJ, Clothier RH (1998) Human keratinocyte cultures in an in vitro approach for the assessment of surfactant-induced irritation. Toxicol in Vitro 12:163–173. https://doi.org/10.1016/S0887-2333(97)00098-2
Wasilewski T, Seweryn A, Krajewski M (2016) Improvement in the safety of use of hand dishwashing liquids through the addition of hydrophobic plant extracts. J Surf Deterg 19:1315–1326. https://doi.org/10.1007/s11743-016-1868-x
Wasilewski T, Hordyjewicz-Baran Z, Zarębska M, Stanek N, Zajszły-Turko E, Tomaka M, Bujak T, Nizioł-Łukaszewska Z (2022a) Sustainable green processing of grape pomace using micellar extraction for the production of value-added hygiene cosmetics. Molecules 27:2444. https://doi.org/10.3390/molecules27082444
Wasilewski T, Seweryn A, Pannert D, Kierul K, Domżał-Kędzia M, Hordyjewicz-Baran Z, Łukaszewicz M, Lewińska A (2022b) Application of Levan-rich digestate extract in the production of safe-to-use and functional natural body wash cosmetics. Molecules 27:2793. https://doi.org/10.3390/molecules27092793
Wasilewski T, Seweryn A (2016) Effect of the hydrophobic plant extract and sodium chloride content on the rheological properties of hand dishwashing liquids. Przem Chem 95:778–783. https://doi.org/10.15199/2F62.2016.4.13
Ziemlewska A, Nizioł-Łukaszewska Z, Bujak T, Zagórska-Dziok M, Wójciak M, Sowa I (2021) Effect of fermentation time on the content of bioactive compounds with cosmetic and dermatological properties in Kombucha Yerba Mate extracts. Sci Rep 11:18792. https://doi.org/10.1038/s41598-021-98191-6
Acknowledgements
We are thankful to the financial support provided for these studies by the National Centre for Research and Development, Poland. Grant No. POIR.01.01.01-00-1433/19.
Author information
Authors and Affiliations
Contributions
AS and TW contributed to the study conception and design. Material preparation, data collection and analysis were performed by AS, TW, ZHB, MB, DP, MÅ and AL. Resourcing, investigation and data curation were done by AS, ZHB, MB and AL The first draft of the manuscript was written by AS, TW, ZHB and AL, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Seweryn, A., Wasilewski, T., Hordyjewicz-Baran, Z. et al. Implementation of sustainable development goals in the cosmetics industry based on the example of cleansing cosmetics containing a surfactin-rich digestate extract. Clean Techn Environ Policy 25, 3111–3125 (2023). https://doi.org/10.1007/s10098-023-02562-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10098-023-02562-8