Abstract
Objective
This systematic review aimed to compare the effects of immersive and non-immersive virtual reality on upper extremity function in stroke survivors by employing a network meta-analysis approach.
Data sources
MEDLINE, Embase, CINAHL Plus, APA PsycINFO, and Scopus were searched. Virtual reality was used for upper extremity rehabilitation; dose-matched conventional rehabilitation was used for comparison. Fugl-Meyer Assessment was used to assess upper extremity function. Searches were limited to English language randomized controlled trials.
Methods
Two independent reviewers conducted study selection, data extraction, and quality assessment. Methodological quality was assessed using the Physiotherapy Evidence Database scale. A random-effects frequentist network meta-analysis was conducted by assuming a common random-effects standard deviation for all comparisons in the network.
Results
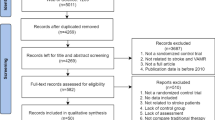
Twenty randomized controlled trials with 813 participants were included, with each study evaluated as good quality. Immersive virtual reality systems were most effective at improving upper extremity function, followed by non-immersive virtual reality systems, then non-immersive gaming consoles of Microsoft Kinect and Nintendo Wii. Conventional rehabilitation was least effective. Immersive virtual reality was estimated to induce 1.39 (95% confidence interval (CI): 0.25, 2.53) and 1.38 (95% CI: 0.55, 2.20) standard mean differences of improvements in upper extremity function, compared to Nintendo Wii intervention and conventional rehabilitation, respectively.
Conclusion
This systematic review and network meta-analysis highlights the superior effects of immersive virtual reality to non-immersive virtual reality systems and gaming consoles on upper extremity motor recovery.



Similar content being viewed by others
Notes
“Cochrane Highly Sensitive Search Strategy for identifying randomized trials in MEDLINE: sensitivity-maximizing version (2008 revision); Ovid format” from Lefebvre C, Glanville J, Briscoe S, Featherstone R, Littlewood A, Marshall C, Metzendorf M-I, Noel-Storr A, Paynter R, Rader T, Thomas J, Wieland LS. Technical Supplement to Chapter 4: Searching for and selecting studies. In: Higgins JPT, Thomas J, Chandler J, Cumpston MS, Li T, Page MJ, Welch VA (eds). Cochrane handbook for systematic reviews of interventions version 6.3 (updated February 2022). Cochrane, 2022. Available from: www.training.cochrane.org/handbook.
Cochrane Highly Sensitive Search Strategy for identifying controlled trials in Embase: (2020 revision); Embase.com format from Lefebvre C, Glanville J, Briscoe S, Featherstone R, Littlewood A, Marshall C, Metzendorf M-I, Noel-Storr A, Paynter R, Rader T, Thomas J, Wieland LS. Technical Supplement to Chapter 4: Searching for and selecting studies. In: Higgins JPT, Thomas J, Chandler J, Cumpston MS, Li T, Page MJ, Welch VA (eds). Cochrane handbook for systematic reviews of interventions version 6.3 (updated February 2022). Cochrane, 2022. Available from: www.training.cochrane.org/handbook.
“Cochrane CINAHL-Plus filter” from Lefebvre C, Glanville J, Briscoe S, Featherstone R, Littlewood A, Marshall C, Metzendorf M-I, Noel-Storr A, Paynter R, Rader T, Thomas J, Wieland LS. Technical Supplement to Chapter 4: Searching for and selecting studies. In: Higgins JPT, Thomas J, Chandler J, Cumpston MS, Li T, Page MJ, Welch VA (eds). Cochrane handbook for systematic reviews of interventions version 6.3 (updated February 2022). Cochrane, 2022. Available from: www.training.cochrane.org/handbook.
Watson RJ, Richardson PH. Identifying randomized controlled trials of cognitive therapy for depression: comparing the efficiency of Embase, Medline and PsycINFO bibliographic databases. Br J Med Psychol. 1999 Dec;72 ( Pt 4):535–42.
Searching Scopus for “Randomised control trials,” NUS library, accessed June 6, 2022 https://libguides.nus.edu.sg/c.php?g=145717&p=2470589.
References
Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Delling FN (2020) Heart disease and stroke statistics—2020 update: a report from the American Heart Association. Circulation 141:e139-596
Kwakkel G, Kollen BJ, van der Grond J, Prevo AJ (2003) Probability of regaining dexterity in the flaccid upper limb: impact of severity of paresis and time since onset in acute stroke. Stroke 34:2181–2186
Hatem SM, Saussez G, Della Faille M, Prist V, Zhang X, Dispa D, Bleyenheuft Y (2016) Rehabilitation of motor function after stroke: a multiple systematic review focused on techniques to stimulate upper extremity recovery. Front Hum Neurosci 10:442
Massetti T, Da Silva TD, Crocetta TB, Guarnieri R, De Freitas BL, Bianchi Lopes P, Watson S, Tonks J, de Mello Monteiro CB (2018) The clinical utility of virtual reality in neurorehabilitation: a systematic review. J Cent Nerv Syst Dis 10:1179573518813541
Levin MF (2011) Can virtual reality offer enriched environments for rehabilitation? Expert Rev Neurother 11:153–155
Hao J, Yao Z, Harp K, Gwon DY, Chen Z, Siu K (2022) Effects of virtual reality in the early-stage stroke rehabilitation: a systematic review and meta-analysis of randomized controlled trials. Physiother Theory Pract. https://doi.org/10.1080/09593985.2022.2094302
Hao J, Xie H, Harp K, Chen Z, Siu K (2022) Effects of virtual reality intervention on neural plasticity in stroke rehabilitation: a systematic review. Arch Phys Med Rehabil 103:523–541
Mekbib DB, Han J, Zhang L, Fang S, Jiang H, Zhu J, Roe AW, Xu D (2020) Virtual reality therapy for upper limb rehabilitation in patients with stroke: a meta-analysis of randomized clinical trials. Brain Inj 34:456–465
Slater M, Wilbur S (1997) A framework for immersive virtual environments (FIVE): Speculations on the role of presence in virtual environments. Presence: Teleoperators Virtual Environ 6:603–616
Rose T, Nam CS, Chen KB (2018) Immersion of virtual reality for rehabilitation—review. Appl Ergon 69:153–161
Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, Ioannidis JP, Straus S, Thorlund K, Jansen JP (2015) The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 162:777–784
Verhagen AP, De Vet HC, De Bie RA, Kessels AG, Boers M, Bouter LM, Knipschild PG (1998) The Delphi list: a criteria list for quality assessment of randomized clinical trials for conducting systematic reviews developed by Delphi consensus. J Clin Epidemiol 51:1235–1241
Cashin AG, McAuley JH (2019) Clinimetrics: Physiotherapy Evidence Database (PEDro) Scale. J Physiother 66:59
Maher CG, Sherrington C, Herbert RD, Moseley AM, Elkins M (2003) Reliability of the PEDro scale for rating quality of randomized controlled trials. Phys Ther 83:713–721
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Rücker G (2012) Network meta-analysis, electrical networks and graph theory. Research Synth Methods 3:312–324
Rücker G, Schwarzer G (2014) Reduce dimension or reduce weights? Comparing two approaches to multi-arm studies in network meta-analysis. Stat Med 33:4353–4369
Rücker G, Schwarzer G (2015) Ranking treatments in frequentist network meta-analysis works without resampling methods. BMC Med Res Methodol 15:1–9
Chaimani A, Salanti G (2012) Using network meta-analysis to evaluate the existence of small-study effects in a network of interventions. Res Synth Methods 3:161–176
Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G (2013) Graphical tools for network meta-analysis in STATA. PLoS ONE 8:e76654
Viechtbauer W, Viechtbauer MW (2015) Package ‘metafor’. The Comprehensive R Archive Network. http://cran.r-project.org/web/packages/metafor/metafor.pdf. Accessed 10 Feb 2023
Rücker G, Schwarzer G, Krahn U, König J, Schwarzer MG (2015) Package ‘netmeta’. Network meta-analysis using frequentist methods. http://cran.r-project.org/web/packages/metafor/metafor.pdf. Accessed 10 Feb 2023
Henrique PP, Colussi EL, De Marchi AC (2019) Effects of exergame on patients’ balance and upper limb motor function after stroke: a randomized controlled trial. J Stroke Cerebrovasc Dis 28:2351–2357
Hsu H, Kuo L, Lin Y, Su F, Yang T, Lin C (2022) Effects of a virtual reality–based mirror therapy program on improving sensorimotor function of hands in chronic stroke patients: a randomized controlled trial. Neurorehabil Neural Repair 36:335–345
Huang C, Chiang W, Yeh Y, Fan S, Yang W, Kuo H, Li P (2022) Effects of virtual reality-based motor control training on inflammation, oxidative stress, neuroplasticity and upper limb motor function in patients with chronic stroke: a randomized controlled trial. BMC Neurol 22:1–14
Mekbib DB, Debeli DK, Zhang L, Fang S, Shao Y, Yang W, Han J, Jiang H, Zhu J, Zhao Z (2021) A novel fully immersive virtual reality environment for upper extremity rehabilitation in patients with stroke. Ann N Y Acad Sci 1493:75–89
Ögün MN, Kurul R, Yaşar MF, Turkoglu SA, Avci Ş, Yildiz N (2019) Effect of leap motion-based 3D immersive virtual reality usage on upper extremity function in ischemic stroke patients. Arq Neuropsiquiatr 77:681–688
Kiper P, Szczudlik A, Agostini M, Opara J, Nowobilski R, Ventura L, Tonin P, Turolla A (2018) Virtual reality for upper limb rehabilitation in subacute and chronic stroke: a randomized controlled trial. Arch Phys Med Rehabil 99:834–842
Kottink AI, Prange GB, Krabben T, Rietman JS, Buurke JH (2014) Gaming and conventional exercises for improvement of arm function after stroke: a randomized controlled pilot study. Games Health J 3:184–191
Oh Y, Kim G, Han K, Won YH, Park S, Seo J, Ko M (2019) Efficacy of virtual reality combined with real instrument training for patients with stroke: a randomized controlled trial. Arch Phys Med Rehabil 100:1400–1408
Piron L, Turolla A, Agostini M, Zucconi CS, Ventura L, Tonin P, Dam M (2010) Motor learning principles for rehabilitation: a pilot randomized controlled study in poststroke patients. Neurorehabil Neural Repair 24:501–508
Shin J, Kim M, Lee J, Jeon Y, Kim S, Lee S, Seo B, Choi Y (2016) Effects of virtual reality-based rehabilitation on distal upper extremity function and health-related quality of life: a single-blinded, randomized controlled trial. J Neuroeng Rehabil 13:1–10
Shin S, Lee H, Chang WH, Ko SH, Shin Y, Kim Y (2022) A smart glove digital system promotes restoration of upper limb motor dunction and enhances cortical hemodynamic changes in subacute stroke patients with mild to moderate weakness: a randomized controlled trial. J Clin Med 11:7343
Ain QU, Khan S, Ilyas S, Yaseen A, Tariq I, Liu T, Wang J (2021) Additional effects of Xbox kinect training on upper limb function in chronic stroke patients: a randomized control trial. Healthcare 9:242
Hung J, Chou C, Chang Y, Wu C, Chang K, Wu W, Howell S (2019) Comparison of Kinect2Scratch game-based training and therapist-based training for the improvement of upper extremity functions of patients with chronic stroke: a randomized controlled single-blinded trial. Eur J Phys Rehabil Med 55:542–550
Kim W, Cho S, Park SH, Lee J, Kwon S, Paik N (2018) A low cost kinect-based virtual rehabilitation system for inpatient rehabilitation of the upper limb in patients with subacute stroke: a randomized, double-blind, sham-controlled pilot trial. Medicine 97:e11173
Lee M, Son J, Kim J, Pyun S, Eun S, Yoon B (2016) Comparison of individualized virtual reality-and group-based rehabilitation in older adults with chronic stroke in community settings: a pilot randomized controlled trial. Eur J Integr Med 8:738–746
Choi JH, Han EY, Kim BR, Kim SM, Im SH, Lee SY, Hyun CW (2014) Effectiveness of commercial gaming-based virtual reality movement therapy on functional recovery of upper extremity in subacute stroke patients. Ann Rehabil Med 38:485–493
da Silva Ribeiro NM, Ferraz DD, Pedreira É, Pinheiro Í, da Silva Pinto AC, Neto MG, Dos Santos LRA, Pozzato MGG, Pinho RS, Masruha MR (2015) Virtual rehabilitation via Nintendo Wii® and conventional physical therapy effectively treat post-stroke hemiparetic patients. Top Stroke Rehabil 22:299–305
Junior VAdS, Santos MdS, Ribeiro NMdS, Maldonado IL (2019) Combining proprioceptive neuromuscular facilitation and virtual reality for improving sensorimotor function in stroke survivors: a randomized clinical trial. J Cent Nerv Syst Dis 11:1179573519863826
Kong K, Loh Y, Thia E, Chai A, Ng C, Soh Y, Toh S, Tjan S (2016) Efficacy of a virtual reality commercial gaming device in upper limb recovery after stroke: a randomized, controlled study. Top Stroke Rehabil 23:333–340
Térémetz M, Garcia A, Hanneton S, Roby-Brami A, Roche N, Bensmail D, Lindberg P, Robertson JV (2022) Improving upper-limb and trunk kinematics by interactive gaming in individuals with chronic stroke: a single-blinded RCT. Ann Phys Rehabil Med 65:101622
Dias S, Sutton AJ, Ades AE, Welton NJ (2013) Evidence synthesis for decision making 2: a generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med Decis Making 33:607–617
Gladstone DJ, Danells CJ, Black SE (2002) The Fugl-Meyer assessment of motor recovery after stroke: a critical review of its measurement properties. Neurorehabil Neural Repair 16:232–240
Lange B, Koenig S, Chang C, McConnell E, Suma E, Bolas M, Rizzo A (2012) Designing informed game-based rehabilitation tasks leveraging advances in virtual reality. Disabil Rehabil 34:1863–1870
Bohil CJ, Alicea B, Biocca FA (2011) Virtual reality in neuroscience research and therapy. Nat Rev Neurosci 12:752–762
Juliano JM, Spicer RP, Vourvopoulos A, Lefebvre S, Jann K, Ard T, Santarnecchi E, Krum DM, Liew S (2020) Embodiment is related to better performance on a brain–computer interface in immersive virtual reality: A pilot study. Sensors 20:1204
Pekna M, Pekny M, Nilsson M (2012) Modulation of neural plasticity as a basis for stroke rehabilitation. Stroke 43:2819–2828
Maier M, Rubio Ballester B, Duff A, Duarte Oller E, Verschure PF (2019) Effect of specific over nonspecific VR-based rehabilitation on poststroke motor recovery: a systematic meta-analysis. Neurorehabil Neural Repair 33:112–129
Laver KE, Lange B, George S, Deutsch JE, Saposnik G, Crotty M (2017) Virtual reality for stroke rehabilitation. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD008349.pub4
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Consent statement
Not applicable.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix 1. Search strategies in each database
Appendix 1. Search strategies in each database
Medline (Ovid) Footnote 1
1 "virtual realit$".ab,ti.
2 "virtual environment$".ab,ti.
3 "augmented realit$".ab,ti.
4 "mixed realit$".ab,ti.
5 "video gam$".ab,ti.
6 exergam$.ab,ti.
7 "serious gam$".ab,ti.
8 Kinect.ab,ti.
9 Wii.ab,ti.
10 (head mounted adj5 devic$).ab,ti.
11 (head mounted adj5 displa$).ab,ti.
12 "smart glass$".ab,ti.
13 smartglass$.ab,ti.
14 arm$.ab,ti.
15 hand$.ab,ti.
16 "upper limb$".ab,ti.
17 "upper extremit$".ab,ti.
18 $stroke.ab,ti.
19 "cerebral vascular acciden$".ab,ti.
20 "cerebral vascular event$".ab,ti.
21 "cerebrovascular acciden$".ab,ti.
22 "cerebrovascular event$".ab,ti.
23 hemiplegi$.ab,ti
24 hemiparesis.ab,ti.
25 randomized controlled trial.pt.
26 controlled clinical trial.pt.
27 randomized.ab.
28 placebo.ab.
29 drug therapy.fs.
30 randomly.ab.
31 trial.ab.
32 groups.ab.
33 exp virtual reality/
34 exp augmented reality/
35 exp Video Games/
36 exp exergaming/
37 exp Smart Glasses/
38 exp Upper Extremity/
39 exp Stroke/
40 exp animals/ not humans.sh.
41 1 or 2 or 3 or 4 or 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13
42 14 or 15 or 16 or 17
43 18 or 19 or 20 or 21 or 22 or 23 or 24
44 25 or 26 or 27 or 28 or 29 or 30 or 31 or 32
45 33 or 34 or 35 or 36 or 37
46 41 or 45
47 38 or 42
48 39 or 43
49 44 not 40
50 46 and 47 and 48 and 49
Embase Footnote 2
#1 'virtual realit*':ab,ti
#2 'virtual environment*':ab,ti
#3 'augmented realit*':ab,ti
#4 'mixed realit*':ab,ti
#5 'video gam*':ab,ti
#6 exergam*:ab,ti
#7 'serious gam*':ab,ti
#8 Kinect:ab,ti
#9 wii:ab,ti
#10 ('head mounted' NEAR/5 devic*):ab,ti
#11 ('head mounted' NEAR/5 displa*):ab,ti
#12 'smart glass*':ab,ti
#13 smartglass*:ab,ti
#14 arm*:ab,ti
#15 hand*:ab,ti
#16 'upper limb*':ab,ti
#17 'upper extremit*':ab,ti
#18 stroke:ab,ti
#19 'post-stroke':ab,ti
#20 poststroke:ab,ti
#21 'cerebral vascular acciden*':ab,ti
#22 'cerebral vascular event*':ab,ti
#23 'cerebrovascular acciden*':ab,ti
#24 'cerebrovascular event*':ab,ti
#25 hemiplegi*:ab,ti
#26 hemiparesis:ab,ti
#27 'randomized controlled trial'/de
#28 'controlled clinical trial'/de
#29 random*:ti,ab,tt
#30 'randomization'/de
#31 'intermethod comparison'/de
#32 placebo:ti,ab,tt
#33 compare:ti,tt OR compared:ti,tt OR comparison:ti,tt
#34 (evaluated:ab OR evaluate:ab OR evaluating:ab OR assessed:ab OR assess:ab) AND (compare:ab OR compared:ab OR comparing:ab OR comparison:ab)
#35 (open NEXT/1 label):ti,ab,tt
#36 ((double OR single OR doubly OR singly) NEXT/1 (blind OR blinded OR blindly)):ti,ab,tt.
#37 'double blind procedure'/de
#38 (parallel NEXT/1 group*):ti,ab,tt
#39 crossover:ti,ab,tt OR 'cross over':ti,ab,tt
#40 ((assign* OR match OR matched OR allocation) NEAR/6 (alternate OR group OR groups OR intervention OR interventions OR patient OR patients OR subject OR subjects OR participant OR participants)):ti,ab,tt
#41 assigned:ti,ab,tt OR allocated:ti,ab,tt
#42 (controlled NEAR/8 (study OR design OR trial)):ti,ab,tt
#43 volunteer:ti,ab,tt OR volunteers:ti,ab,tt
#44 'human experiment'/de
#45 trial:ti,tt
#46 ((random* NEXT/1 sampl* NEAR/8 ('cross section*' OR questionnaire* OR survey OR surveys OR database OR databases)):ti,ab,tt) NOT ('comparative study'/de OR 'controlled study'/de OR 'randomised controlled':ti,ab,tt OR 'randomized controlled':ti,ab,tt OR 'randomly assigned':ti,ab,tt)
#47 'cross‐sectional study' NOT ('randomized controlled trial'/de OR 'controlled clinical study'/de OR 'controlled study'/de OR 'randomised controlled':ti,ab,tt OR 'randomized controlled':ti,ab,tt OR 'control group':ti,ab,tt OR 'control groups':ti,ab,tt)
#48 'case control*':ti,ab,tt AND random*:ti,ab,tt NOT ('randomised controlled':ti,ab,tt OR 'randomized controlled':ti,ab,tt)
#49 'systematic review':ti,tt NOT (trial:ti,tt OR study:ti,tt)
#50 nonrandom*:ti,ab,tt NOT random*:ti,ab,tt
#51 'random field*':ti,ab,tt
#52 ('random cluster' NEAR/4 sampl*):ti,ab,tt
#53 review:ab AND review:it NOT trial:ti,tt
#54 'we searched':ab AND (review:ti,tt OR review:it)
#55 'update review':ab
#56 (databases NEAR/5 searched):ab
#57 (rat:ti,tt OR rats:ti,tt OR mouse:ti,tt OR mice:ti,tt OR swine:ti,tt OR porcine:ti,tt OR murine:ti,tt OR sheep:ti,tt OR lambs:ti,tt OR pigs:ti,tt OR piglets:ti,tt OR rabbit:ti,tt OR rabbits:ti,tt OR cat:ti,tt OR cats:ti,tt OR dog:ti,tt OR dogs:ti,tt OR cattle:ti,tt OR bovine:ti,tt OR monkey:ti,tt OR monkeys:ti,tt OR trout:ti,tt OR marmoset*:ti,tt) AND 'animal experiment'/de
#58 'animal experiment'/de NOT ('human experiment'/de OR 'human'/de)
#59 'virtual reality'/exp
#60 'virtual environment'/exp
#61 'augmented reality'/exp
#62 'mixed reality'/exp
#63 'video game'/exp
#64 'serious game'/exp
#65 'head-mounted display'/exp
#66 'hand'/exp
#67 'upper limb'/exp
#68 'cerebrovascular accident'/exp
#69 'cerebrovascular event'/exp
#70 'hemiplegia'/exp
#71 'hemiparesis'/exp
#72 #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13
#73 #14 OR #15 OR #16 OR #17
#74 #18 OR #19 OR #20 OR #21 OR #22 OR #23 OR #24 OR #25 OR #26
#75 #27 OR #28 OR #29 OR #30 OR #31 OR #32 OR #33 OR #34 OR #35 OR #36 OR #37 OR #38 OR #39 OR #40 OR #41 OR #42 OR #43 OR #44 OR #45
#76 #46 OR #47 OR #48 OR #49 OR #50 OR #51 OR #52 OR #53 OR #54 OR #55 OR #56 OR #57 OR #58
#77 #59 OR #60 OR #61 OR #62 OR #63 OR #64 OR #65
#78 #66 OR #67
#79 #68 OR #69 OR #70 OR #71
#80 #72 OR #77
#81 #73 OR #78
#82 #74 OR #79
#83 #75 NOT #76
#84 #80 AND #81 AND #82 AND #83
CINAHL Plus Footnote 3
S1 TI (virtual realit*) OR AB (virtual realit*)
S2 TI (virtual environment*) OR AB (virtual environment*)
S3 TI (augmented realit*) OR AB (augmented realit*)
S4 TI (mixed realit*) OR AB (mixed realit*)
S5 TI (video gam*) OR AB (video gam*)
S6 TI (exergam*) OR AB (exergam*)
S7 TI (serious gam*) OR AB (serious gam*)
S8 TI Kinect OR AB Kinect
S9 TI Wii OR AB Wii
S10 TI ((head mounted) N5 (devic*)) OR AB ((head mounted) N5 (devic*))
S11 TI ((head mounted) N5 (displa*)) OR AB ((head mounted) N5 (displa*))
S12 TI (smart glass*) OR AB (smart glass*)
S13 TI (smartglass*) OR AB (smartglass*)
S14 TI (arm*) OR AB (arm*)
S15 TI (hand*) OR AB (hand*)
S16 TI (upper limb*) OR AB (upper limb*)
S17 TI (upper extremit*) OR AB (upper extremit*)
S18 TI stroke OR AB stroke
S19 TI post-stroke OR AB post-stroke
S20 TI poststroke OR AB poststroke
S21 TI (cerebral vascular acciden*) OR AB (cerebral vascular acciden*)
S22 TI (cerebral vascular event*) OR AB (cerebral vascular event*)
S23 TI (cerebrovascular acciden*) OR AB (cerebrovascular acciden*)
S24 TI (cerebrovascular event*) OR AB (cerebrovascular event*)
S25 TI (hemiplegi*) OR AB (hemiplegi*)
S26 TI hemiparesis OR AB hemiparesis
S27 MH randomized controlled trials
S28 MH double‐blind studies
S29 MH single‐blind studies
S30 MH random assignment
S31 MH pretest‐posttest design
S32 MH cluster sample
S33 TI (randomised OR randomized)
S34 AB (random*)
S35 TI (trial)
S36 MH (sample size) AND AB ( assigned OR allocated OR control)
S37 MH (placebos)
S38 PT (randomized controlled trial)
S39 AB (control W5 group)
S40 MH (crossover design) OR MH (comparative studies)
S41 AB (cluster W3 RCT)
S42 MH animals +
S43 MH (animal studies)
S44 TI (animal model*)
S45 MH (human)
S46 MH (Virtual Reality +)
S47 MH (Augmented Reality)
S48 MH (Video Games +)
S49 MH (Smart Glasses)
S50 MH (Upper Extremity +)
S51 MH (Stroke +)
S52 MH (Hemiplegia)
S53 S1 OR S2 OR S3 OR S4 OR S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13
S54 S14 OR S15 OR S16 OR S17
S55 S18 OR S19 OR S20 OR S21 OR S22 OR S23 OR S24 OR S25 OR S26
S56 S27 OR S28 OR S29 OR S30 OR S31 OR S32 OR S33 OR S34 OR S35 OR S36 OR S37 OR S38 OR S39 OR S40 OR S41
S57 S42 OR S43 OR S44
S58 S57 NOT S45
S59 S56 NOT S58
S60 S46 OR S47 OR S48 OR S49
S61 S51 OR S52
S62 S53 OR S60
S63 S50 OR S54
S64 S55 OR S61
S65 S59 AND S62 AND S63 AND S64
APA PsycInfo Footnote 4
S1 TI (virtual realit*) OR AB (virtual realit*)
S2 TI (virtual environment*) OR AB (virtual environment*)
S3 TI (augmented realit*) OR AB (augmented realit*)
S4 TI (mixed realit*) OR AB (mixed realit*)
S5 TI (video gam*) OR AB (video gam*)
S6 TI (exergam*) OR AB (exergam*)
S7 TI (serious gam*) OR AB (serious gam*)
S8 TI Kinect OR AB Kinect
S9 TI Wii OR AB Wii
S10 TI ((head mounted) N5 (devic*)) OR AB ((head mounted) N5 (devic*))
S11 TI ((head mounted) N5 (displa*)) OR AB ((head mounted) N5 (displa*))
S12 TI (smart glass*) OR AB (smart glass*)
S13 TI (smartglass*) OR AB (smartglass*)
S14 TI (arm*) OR AB (arm*)
S15 TI (hand*) OR AB (hand*)
S16 TI (upper limb*) OR AB (upper limb*)
S17 TI (upper extremit*) OR AB (upper extremit*)
S18 TI stroke OR AB stroke
S19 TI post-stroke OR AB post-stroke
S20 TI poststroke OR AB poststroke
S21 TI (cerebral vascular acciden*) OR AB (cerebral vascular acciden*)
S22 TI (cerebral vascular event*) OR AB (cerebral vascular event*)
S23 TI (cerebrovascular acciden*) OR AB (cerebrovascular acciden*)
S24 TI (cerebrovascular event*) OR AB (cerebrovascular event*)
S25 TI (hemiplegi*) OR AB (hemiplegi*)
S26 TI hemiparesis OR AB hemiparesis
S27 SU.EXACT(Treatment Effectiveness Evaluation)
S28 SU.EXACT.EXPLODE(Treatment Outcomes)
S29 SU.EXACT(Placebo)
S30 SU.EXACT(Followup Studies)
S31 (placebo*)
S32 (random*)
S33 (comparative stud*)
S34 (clinical N3 trial*)
S35 (research N3 design)
S36 (evaluat* N3 stud*)
S37 (prospectiv* N3 stud*)
S38 (singl* OR doubl* OR trebl* OR tripl*) N3 (blind* OR mask*)
S39 DE (Virtual Reality) OR DE (Augmented Reality)
S40 MM (Computer Games)
S41 MM "Arm (Anatomy)"
S42 DE "Hand (Anatomy)" OR DE "Fingers (Anatomy)" OR DE "Palm (Anatomy)"
S43 MM (Cerebrovascular Accidents)
S44 MM (Hemiparesis) OR MM (Hemiplegia)
S45 S1 OR S2 OR S3 OR S4 OR S5 OR S6 OR S7 OR S8 OR S9 OR S10 OR S11 OR S12 OR S13
S46 S14 OR S15 OR S16 OR S17
S47 S18 OR S19 OR S20 OR S21 OR S22 OR S23 OR S24 OR S25 OR S26
S48 S27 OR S28 OR S29 OR S30 OR S31 OR S32 OR S33 OR S34 OR S35 OR S36 OR S37 OR S38
S49 S39 OR S40
S50 S41 OR S42
S51 S43 OR S44
S52 S45 OR S49
S53 S46 OR S50
S54 S47 OR S51
S55 S48 AND S52 AND S53 AND S54
Scopus Footnote 5
( ( ( TITLE ( "virtual realit*") OR ABS ( "virtual realit*"))) OR ( ( TITLE ( "virtual environment*") OR ABS ( "virtual environment*"))) OR ( ( TITLE ( "augmented realit*") OR ABS ( "augmented realit*"))) OR ( ( TITLE ( "mixed realit*") OR ABS ( "mixed realit*"))) OR ( ( TITLE ( "video gam*") OR ABS ( "video gam*"))) OR ( ( TITLE ( exergam*) OR ABS ( exergam*))) OR ( ( TITLE ( "serious gam*") OR ABS ( "serious gam*"))) OR ( ( TITLE ( kinect) OR ABS ( kinect))) OR ( ( TITLE ( wii) OR ABS ( wii))) OR ( ( TITLE ( "head mounted" W/5 devic*) OR ABS ( "head mounted" W/5 devic*))) OR ( ( TITLE ( "head mounted" W/5 displa*) OR ABS ( "head mounted" W/5 displa*))) OR ( ( TITLE ( "smart glass*") OR ABS ( "smart glass*"))) OR ( ( TITLE ( smartglass*) OR ABS ( smartglass*)))) AND ( ( ( TITLE ( arm*) OR ABS ( arm*))) OR ( ( TITLE ( hand*) OR ABS ( hand*))) OR ( ( TITLE ( "upper limb*") OR ABS ( "upper limb*"))) OR ( ( TITLE ( "upper extremit*") OR ABS ( "upper extremit*")))) AND ( ( ( TITLE ( *stroke) OR ABS ( *stroke))) OR ( ( TITLE ( "cerebral vascular acciden*") OR ABS ( "cerebral vascular acciden*"))) OR ( ( TITLE ( "cerebral vascular event*") OR ABS ( "cerebral vascular event*"))) OR ( ( TITLE ( "cerebrovascular acciden*") OR ABS ( "cerebrovascular acciden*"))) OR ( ( TITLE ( "cerebrovascular event*") OR ABS ( "cerebrovascular event*"))) OR ( ( TITLE ( hemiplegi*) OR ABS ( hemiplegi*))) OR ( ( TITLE ( hemiparesis) OR ABS ( hemiparesis)))) AND ( ( INDEXTERMS ( "clinical trials" OR "clinical trials as a topic" OR "randomized controlled trial" OR "Randomized Controlled Trials as Topic" OR "controlled clinical trial" OR "Controlled Clinical Trials" OR "random allocation" OR "Double-Blind Method" OR "Single-Blind Method" OR "Cross-Over Studies" OR "Placebos" OR "multicenter study" OR "double blind procedure" OR "single blind procedure" OR "crossover procedure" OR "clinical trial" OR "controlled study" OR "randomization" OR "placebo")) OR ( TITLE-ABS-KEY ( ( "clinical trials" OR "clinical trials as a topic" OR "randomized controlled trial" OR "Randomized Controlled Trials as Topic" OR "controlled clinical trial" OR "Controlled Clinical Trials as Topic" OR "random allocation" OR "randomly allocated" OR "allocated randomly" OR "Double-Blind Method" OR "Single-Blind Method" OR "Cross-Over Studies" OR "Placebos" OR "cross-over trial" OR "single blind" OR "double blind" OR "factorial design" OR "factorial trial"))) OR ( TITLE-ABS ( clinical AND trial* OR trial* OR rct* OR random* OR blind*)))
Figure 4
Figure 5
Table 4
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hao, J., He, Z., Yu, X. et al. Comparison of immersive and non-immersive virtual reality for upper extremity functional recovery in patients with stroke: a systematic review and network meta-analysis. Neurol Sci 44, 2679–2697 (2023). https://doi.org/10.1007/s10072-023-06742-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-06742-8