Abstract
Restoration of natural forests previously replaced by plantations is a widespread challenge for forestry in Chile and elsewhere. However, there is little documented evidence for successful restoration, either through active or passive approaches. In this study, we aimed at (1) determining the potential for passive restoration in first-rotation Pinus radiata plantations through natural regeneration of native tree species and (2) identifying drivers of this advance regeneration. Across different regions in south-central Chile, we established nearly 260 plots to assess regeneration and environmental conditions along 26 transects running from plantations into adjacent natural forests. The regeneration was exclusively composed by native species, except for 7 individuals of P. radiata. Mean density and diversity of seedlings were significantly higher in natural forests than in plantations, but this was not the case for sapling density, and no differences in sapling diversity were supported. Additionally, significant differences in regeneration composition between plantations and natural forests were found only at two of the eight study sites. Compared to climatic and soil chemical variables, which varied mostly at regional scales, local environmental conditions showed little influence on regeneration, possibly due to the structural homogeneity of plantations. Yet, the significantly higher basal area, litter thickness and gap fraction of plantations compared to natural forests suggest that these factors may explain differences at the seedling stage. Our study indicates that the use of appropriate harvesting methods that maintain advance regeneration may facilitate the transition from plantations to native forests through passive restoration. The use this approach should be further investigated through analyzing regeneration’s response to different forms of plantation harvesting.
Similar content being viewed by others
Highlights
-
First large-scale assessment of native species regeneration in pine plantations in Chile.
-
Similar sapling density and diversity in plantations and natural secondary forests.
-
Advance regeneration in plantations can facilitate passive forest restoration.
Introduction
Given the high productivity of plantation forests, it has been suggested that they can contribute to biodiversity conservation by reducing the pressure for wood production from natural and semi-natural forests (Paquette and Messier 2010). Although this may hold true under certain policy and regulatory conditions, it is clearly not the case when plantations are established through conversion of natural forests (Pirard and others 2016), which has occurred in several regions in the world (Schroth and others 2002; Chen and others 2005; Domec and others 2015; Heilmayr and others 2016). For that reason, plantations that have been established through conversion of natural ecosystems in recent decades are usually deemed not eligible for certification programs, and their restoration to more natural conditions is encouraged (FSC 2015; PEFC 2018). Instead of replacing native forests, plantations are nowadays expected to be established on degraded lands, where in addition to providing wood and fiber products, they can enhance environmental and social benefits (Bauhus and others 2010; Baral and others 2016; Silva and others 2019; NGP 2020).
Following a long process of extensive deforestation and land degradation, a rapid expansion of commercial tree plantations took place in Chile, especially since 1975 along the Coastal Range, as a response to policies promoting afforestation and the development of the forest industry (Lara and Veblen 1993; Otero 2006; Niklitschek 2007; Prado 2015). These plantations were mainly of Pinus radiata D. Don (Monterey pine), Eucalyptus globulus Labill. (blue gum) and E. nitens (Dean & Maiden) Maiden (shining gum). From these species, P. radiata is currently naturalized and considered potentially invasive (Bustamante and Simonetti 2005; PNUD 2017). Although they were partly established on degraded lands to contribute to soil protection (Lara and Veblen 1993; Prado 2015), a substantial proportion was also established following clearing of natural forests and shrublands (Lara and Veblen 1993; Prado 2015; Heilmayr and others 2016). The natural forests of this region show high levels of endemism and form part of one the 24 world biodiversity hotspots described by Myers and others (2000). They also comprise some of the most threatened and least protected terrestrial ecosystems in the country (Pliscoff and Fuentes-Castillo 2011; Smith-Ramírez and others 2019). In response to requirements of forest certification schemes adopted by some of the main forest companies, there are now commitments to restore at least 35,000 ha of natural forests and shrublands that were replaced by plantations since 1994 (WWF 2013; Forestal Mininco S.A. 2017; Forestal Arauco S.A. 2018). However, the available experience for this form of restoration is limited, and the development of silvicultural approaches to facilitate this conversion remains an important restoration challenge (Bannister and others 2016; Salas and others 2016).
Plantation forests in Chile are mainly harvested through clearcutting (Salas and others 2016), which suggests that restoration to native forests may commence following this form of harvesting. However, plantations can also support regeneration processes that may accelerate succession toward target ecosystems (Parrotta and others 1997; Brockerhoff and others 2013; McAlpine and others 2020). By providing suitable microclimatic conditions, supporting habitat or improving habitat connectivity for seed dispersers, recovering soil organic matter and fertility, and suppressing competitive weeds and shrubs, plantations can facilitate the establishment of species that are characteristic of the original natural forests (Parrotta and others 1997; Lemenih and Bongers 2010; Geldenhuys and others 2017). At the same time, however, closed plantation stands may not provide sufficient light or soil water for the establishment of a range of native tree species (Huber and Trecaman 2004; Tzu-Yang and others 2015; Kremer and others 2021).
Some studies have shown that P. radiata plantations in Chile usually develop an understory of native trees and shrubs intermixed with exotic species, during their approximately 20-year rotations (Ramírez and others 1984; Eguiguren 1995; Estades and Escobar 2005; Gómez and others 2009; Heinrichs and others 2018). Stand attributes such as canopy cover, distance to seed sources and the coppicing ability of the main tree species appear to play a relevant role in the abundance and growth of this advance regeneration (Becerra and Montenegro 2013; García and others 2016; Promis and others 2019; Kremer and others 2021). This suggests that restoration of native forests from P. radiata plantations may follow a passive approach based on natural advance regeneration (Holl and Aide 2011; Balestrin and others 2020), which may be combined with active interventions for the partial or complete removal of the canopy (Chan 2006; Cummings and others 2007; Seiwa 2012). However, previous studies describing the understory of P. radiata plantations were mostly constrained to specific locations and did not necessarily focus on stands that were established through conversion of natural forests or on tree species regeneration. To address this knowledge deficit, we have studied i) the potential of first-rotation P. radiata plantations established through clearing of natural forest for a passive restoration approach based on natural regeneration of native tree species and ii) the influence of environmental factors on the establishment of this regeneration. We hypothesized that advance regeneration of native tree species in P. radiata plantations is partly representative of that in natural forests. For this purpose, we carried out a large-scale survey of P. radiata plantations across different landscapes to assess natural regeneration of tree species compared to adjacent remnants of natural forest.
Methods
Study Area
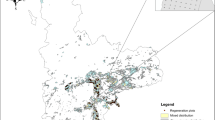
The study was carried out along the Coastal Range of south-central Chile between the regions of Ñuble and Los Ríos (~ 36º00’S— ~ 40º00’S) (Figure 1). This mountain range is characterized by steep slopes reaching more than 50% and altitudes up to 1,525 m a.s.l. that decrease southwards (Melnick and others 2009; Luzio 2010). The relatively deep soils (> 1 m) originated mainly from metamorphic bedrock and in the southern part also from volcanic ash that was deposited over the metamorphic complex (Luzio 2010). The climate is characterized by warm Mediterranean conditions in the north, to wet-temperate in the south. In addition to this latitudinal variation, precipitation decreases from west to east, together with an increase in thermal amplitude as oceanic influence decreases (Luebert and Pliscoff 2005). Depending mainly on altitude and latitude, mean annual precipitation ranges from 1,000 to 2,248 mm year−1, with up to 5 months of summer drought. Minimum and maximum mean monthly temperatures vary spatially from 7.1 to 8.8ºC, and from 16.1 to 18.7ºC, respectively, with 2–17 frost days per year (AGRIMED 2017). The natural forests of this region have the highest richness of vascular plant species across the country and show high levels of endemism, harboring 45 locally endemic vascular plants, and several animal species that are sometimes restricted to only a few locations (Bannister and others 2012, Smith-Ramírez and others 2019). Yet, these forests are poorly represented in protected areas, and owing to extensive land-use change, some forest types are now confined to small patches that represent a minimum share of their original cover (Pliscoff and Fuentes-Castillo 2011; Echeverría and others 2019). The natural forests of the study area belong to the Deciduous Forest Region, which is characterized by the dominance of the Nothofagus genus. Nothofagus obliqua (Mirb.) Oerst. is continuously present amongst the dominant species, sharing the upper canopy layer with Nothofagus glauca (Phil.) Krasser in the north, and with Nothofagus dombeyi (Mirb.) Oerst. and sometimes Nothofagus alpina (Poepp & Endl.) Oerst. in the south. Companion tree species from other genera usually include Persea lingue (Ruiz & Pav.) Nees and Gevuina avellana Molina, together with scleropyllous species such as Cryptocarya alba (Molina) Looser and Peumus boldus Molina in the north and with species more characteristic of the evergreen forest type in the south (Luebert and Pliscoff 2006). The regeneration of these forests is mainly driven by small- to medium-scale disturbances such as treefall gaps, as opposed to the large-scale disturbances experienced in the Andes. The lower frequency and spatial scale of these disturbances allow the establishment of more shade-tolerant species, leading to communities with higher tree species richness (Veblen and others 1981).
Map of the study area, across Chile’s administrative regions. Blue triangles represent the sites sampled. Areas in dark green represent plantations of Pinus radiata, whereas areas in light green represent eucalypt plantations (based on CONAF 2017).
Currently, plantations of P. radiata that are managed for saw logs and pulp wood are the most wide-spread land use across this region (Salas and others 2016; Echeverría and others 2019). Where direct conversion of natural forests to P. radiata plantations occurred, the natural forest was cleared mainly through clearcutting and burning (Lara and Veblen 1993). Studies on the ecology of P. radiata plantations to the north of our study region have described the composition of their understory as a subset of the communities of the natural ecosystems (Ramírez and others 1984; Estades and Escobar 2005).
Sampling Method
We sampled 18- to 22-year-old plantations of Pinus radiata that had been established after clearing natural forests and were adjacent to remnants of natural forest. We chose plantations approaching harvesting age, because their understory should have reached the maximum structural and compositional development possible under the current management regime (Estades and Escobar 2005; Heinrichs and others 2018; Forbes and others 2019).
Eight different sites were sampled within a region extending approximately 430 km from north to south, aiming to represent different vegetation sub-types and a variety of soil and climatic conditions of natural forests to be restored in the Deciduous Forest Region (Figure 1). The plantation stands at different sites are described in Table 1, and further details on the sites’ potential vegetation, soil and climate types are provided in Table S1. Our samples consisted of straight, 135 m-long transects, which ran perpendicular to the border between P. radiata plantations and remnants of natural forest. In total, 26 transects were established in groups of one to six per site, at distances of 130 to 2,700 m from each other within sites. The number of transects sampled at each site was determined on the basis of the site’s internal environmental variation and accessibility, and the suitability of the landscape spatial configuration to the sampling design. In all plantations, the natural forest had been clearcut and the logging slash burned. The P. radiata stands had been established with initial densities of approximately 1,300 seedlings ha−1 and were managed for sawlog production, which includes at least one thinning operation.
Each transect consisted of 10 sampling points established every 15 m, except for two transects that had seven and nine plots in total, respectively, given the steep slopes that sometimes hindered the access. The edge between the plantation and the natural forest was a relatively straight line of at least 100 m, at the center of which one sampling point (E) was located. From the remaining sampling points, 6 were established inside the plantation at 15, 30, 45, 60, 75 and 90 m from the edge (P1, P2, P3, P4, P5 and P6, respectively) and three inside the natural forest at 15, 30 and 45 m from the edge (N1, N2 and N3, respectively). The minimum distance to any non-forest land use was 90 m for sampling points in the P. radiata plantations and 45 for those in the natural forests (Figure 2).
Field Measurements
At each sampling point, natural regeneration of all tree species was quantified in a circular 2 m-radius plot. All seedlings (height between 5 and 150 cm; diameter at breast height (dbh) < 5 cm) and saplings (height > 150 cm; dbh < 5 cm) were counted and identified at species level. Seedlings were classified into small (5–20 cm height) and large (20–150 cm height). Additionally, dbh and species of all trees (dbh > 5 cm) were recorded within a 7 m-radius (154 m2) around the sampling point, which was assumed to be the area of influence of the neighboring pine trees on the regeneration in the 2 m-radius plots. At each sampling point, altitude and slope were measured, and a hemispherical photograph was taken to estimate light parameters at 1.3 m above the 2 m-radius plot. These photographs were taken using a fisheye converter and avoiding direct sunlight. We visually estimated the percentage cover of understory below 1.3 m height, and the percentage ground cover of deadwood, leaf litter and pine needles on the ground. Mean depth of the litter layer was measured at four points that were systematically distributed within the 2 m-radius plots.
Soil samples were extracted within the 2 m-radius plots at the transect points P1, P3 and P5 (plantation) and N1 and N3 (natural forest), from the upper 10 cm at the same four locations used for the measurements of litter layer depth. The samples were then bulked to form one composite sample for each of these forest types per transect. All measurements and samplings were conducted between March and June 2018.
Sample Analysis
Hemispherical photographs were analyzed with WinSCANOPY DSLR 2008a (Regent Instruments, Quebec, QC) to quantify the daily total, direct and diffuse photosynthetic photon flux density (PPFD) above and below canopy. Following drying and sieving (2 mm), soil samples were analyzed for total N and organic carbon content with a Leco-Truspec CN analyzer and for pH with a portable pH-meter (WTW, Weilheim, Germany). Following extraction of soils with 1 M NH4Cl, cation exchange capacity (CEC) in the extractant was measured using inductively coupled plasma optical emission spectroscopy (ICP-OES) (Spectro, Kleve, Germany) (Weinzierl and Dietze 2000; König 2005).
Climatic and Landscape Data
Climatic parameters and landscape metrics were calculated for each transect. Climatic data from the Climate and Resilience Research Centre (CR2) were used, which were based on locally calibrated models of monthly precipitation and temperature at a 0.05º latitude–longitude resolution (approximately 5 km) (CR2 2018). We used data from 1995–2017 (that is,, the approximate time span between the establishment of the P. radiata plantations and the sampling) to calculate mean values of annual precipitation, monthly temperature, and minimum and maximum annual temperature. Additionally, based on satellite imagery, the Euclidean distance from each sampling point within plantations to natural forest was calculated. We also calculated the proportion of natural forest area and the total length of border between plantations and natural forests within radii of 100, 150, 200 and 250 m around the sampling points. These distances were chosen based on previously reported data on dispersal distances from natural forest into adjacent P. radiata plantations in south-central Chile (García and others 2016).
Data Analysis
Generalized linear mixed effects models (GLMM) were used to examine the influence of the three different environments (P. radiata plantation, adjacent natural forest and transition zone among both types, hereafter the “stand type”) on regeneration density of native tree species. Separate sets of models were fitted to predict total regeneration density, seedling density and sapling density. For each response variable, the full model included stand type and vegetation sub-type as fixed effects, and transect and site as random effects, with transects nested within sites. The interaction between stand type and vegetation sub-type was also included among the fixed effects. Several versions of the full models were fitted by successively excluding predictors until the only remaining factor was stand type (Table S2). To account for observed zero-inflation, we included additional model variants that incorporated zero-inflation i) constant across the dataset, ii) dependent on the stand type, iii) dependent on the vegetation sub-type and iv) dependent on both stand type and vegetation sub-type (Brooks and others 2017).
The models for total density and for sapling density were fitted with a negative binomial distribution and a log link function, given that overdispersion was observed in both cases, whereas the models for seedling density were fitted with a Poisson distribution and the log link function (Brooks and others 2017). All models were fitted using the “glmmTMB” R-package (Magnusson and others 2020). From each set of models, the best-fit model was selected with the Akaike information criterion (AIC), using the “bblme” R-package (Bolker and others 2020). Wald tests were used to test the significance of each response variable, and Tukey tests were conducted for multiple pairwise comparisons among stand types. Additionally, Mann–Whitney U tests were carried out to compare species-specific seedling and sapling abundances between plantations and natural forests and interpret the influence of the stand type on the basis of biological traits of these species. Edge plots were not included in this analysis, given the low sample sizes for single species.
To compare the diversity of the regeneration of tree species among P. radiata plantations and adjacent natural forests, for each stand type we calculated rarefaction and extrapolation (R/E) curves of the three most widely used members of the family of Hill numbers: species richness (q = 0), the exponential of Shannon entropy (q = 1) and the inverse Simpson concentration (q = 2), with higher values of q indicating increasing sensitivity to species relative abundances (Hsieh and others 2016). The corresponding curves were calculated with the iNEXT R-package (Hsieh and Chao 2020), using a sampling-unit approach to describe the sampling effort (diversity vs. number of plots) (Hsieh and others 2016). Separate curves were calculated for total regeneration, seedlings and saplings. R/E curves of Hill numbers represent a unified framework for estimating species diversity and making statistical comparisons of different assemblages while controlling for the effect of the sampling effort (Chao and others 2014; Hsieh and others 2016). Ninety-five percent confidence intervals were calculated for each curve, with 50 bootstrap replications. If the confidence intervals of the corresponding curves do not overlap for any sample size in the comparison range, significant differences are guaranteed among the expected diversities. However, partially overlapping intervals do not guarantee non-significance (Schenker and Gentleman 2001; Chao and others 2014). Given that this type of comparison is partly based on extrapolations of diversity values to higher sample sizes, the edge plots (between plantations and natural forests) were excluded from these analyses, since their reference (effective) sample size (n = 26) would have not allowed reliable extrapolations. Additionally, we calculated R/E curves individually for each site to determine differences in diversity among stand types at the local scale. Given the lower number of plots available for each site, in this case the sampling effort of the R/E curves was based on individuals instead of plots (diversity vs. number of individuals), and no separate analysis was conducted for seedlings and saplings.
To compare the tree species composition of advance regeneration in plantations and natural forests, we conducted a non-metric multidimensional scaling analysis (NMDS) and a permutational analysis of variance (PERMANOVA) to test whether differences among them were significant (“Vegan” R-package; Oksanen and others 2019). These analyses were conducted for all observations (plots) together and with observations grouped by site. Only plots from plantations and natural forest were included, and rare species (< 3% frequency across plantation and natural forest plots) were excluded. Dissimilarity matrices were calculated with the Jaccard index, which relies exclusively on presence data (Borcard and others 2011), and 999 permutations were conducted in each case.
Mann–Whitney U tests were used to compare site conditions (basal area, light parameters, understory cover, litter thickness and soil chemical properties) between plantations, edge plots and natural forest.
A canonical correspondence analysis (CCA) was conducted to identify the main drivers of natural regeneration in plantations. A cluster analysis to identify associations between natural tree communities from different transects revealed no clear grouping among tree species communities in natural forests (Figure S1). Hence, no specific grouping of the data was conducted. Rare species (< 3% frequency across plantation plots) were also excluded from this analysis. Given the high number of potential explanatory variables, we removed redundant ones from the analysis. When explanatory variables accounting for a same stand attribute were highly correlated (Spearman’s Rho correlation coefficient (ρ) > 0.75; p-value < 0.05), we calculated for each variable of that group the mean ρ of its correlations with the other variables and selected the one with the highest value. On the basis of the resulting set of explanatory variables, further selection of non-redundant variables was carried out through forward selection, using the “ordistep” function from the “vegan” R-package (Borcard and others 2011; Oksanen and others 2019). The variables originally measured and those finally included in the CCA analysis are shown in Table S3. We performed permutation tests (n = 999) to check the significance of the complete CCA model and of each constrained axis and explanatory variable individually. We ranked the explanatory variables according to their importance for regeneration, based on the weighted average of absolute values of their scores along the main significant canonical axes (that is, those with eigenvalues > 0.30) (ter Braak and Verdonschot 1995). The weighting was based on the corresponding eigenvalue of each canonical axis.
For all statistical tests, null-hypotheses were rejected with p-values < 0.05.
Results
Abundance of Regeneration and Diameter Distribution of Native Trees
The best-fit models predicting total regeneration density (seedlings and saplings) and seedling density included the variables stand type, vegetation subtype, and the interaction among both predictors as fixed effects, and transect identity as a random effect. The best-fit model for sapling density included only stand type as a fixed effect and transect identity as a random effect. According to the corresponding models, stand type significantly affected both total regeneration density and seedling density (p-value < 0.05), although it did not affect sapling density. Total regeneration density was significantly lower in plantations than in edge plots and natural forests. Seedling density showed a similar pattern, although it was also significantly higher in natural forests than in edge plots. In contrast, sapling density showed no significant differences among stand types (Figure 3; Table S4).
Whereas the regeneration in plantations was dominated by saplings (> 150 cm) (64%), small seedlings below 20 cm dominated the regeneration in natural forests, although in the latter case the distribution among height classes was more even. At the edge, the distribution of height classes among regenerating plants was intermediate between plantations and natural forests (Figure 4).
Seedlings of several species (C. alba, Eucrhyphia cordifolia Cav., G. avellana, L. sempervirens (Ruiz & Pav.) Tul., Myrceugenia planipes (Hook & Arn.) Berg, P. lingue, Aextoxicon punctatum Ruiz & Pav. and Embothrium coccineum J. R. Forst. & G. Forst.) were significantly more abundant in natural forests than in plantations. However, in most cases these differences were no longer present at the sapling stage, with the exceptions of A. punctatum and E. coccineum, which had very low seedling and sapling densities in plantations, whereas E. coccineum also had a very low occurrence. The only species with significantly higher sapling abundance in plantations compared to natural forest was Aristotelia chilensis (Molina) Stuntz, which also showed the highest excess of sapling over seedling density in the plantations (Figure 5). The overall abundance of P. radiata regeneration was negligible, with only seven individuals found in total.
Abundance of tree species regeneration in P. radiata plantations and natural forest stands, as seedlings (black bars) and saplings (gray bars). Rare species (< 3% total frequency) are not represented. Stars next to density bars indicate significant differences in density among plantations and natural forests for a single species, based on Mann–Whitney U tests. *p-value < 0.05; **p-value < 0.01; ***p-value < 0.001.
As expected, the tree diameter structure differed considerably among the three stand types. In plantations, pine trees showed a unimodal size distribution, whereas trees of native species were only present as small individuals, mostly below 10 cm dbh (Figure S3). In contrast, the tree diameter distribution of natural forests showed a negative exponential pattern, with an almost three-fold higher density of small trees (< 10 cm dbh) than in plantations. At the edge, stand structure was intermediate between plantations and native forests (Figure S3). Shade-intolerant, semi-tolerant and shade-tolerant native tree species were present in the understory of plantations, with N. obliqua (intolerant), P. lingue (shade tolerant) and G. avellana (semi-tolerant) making 24, 18 and 14% of the basal area, respectively.
Regeneration Diversity and Composition
Species richness (q = 0) of total tree regeneration (seedlings and saplings) was similar among plantations and natural forests. However, Shannon (q = 1) and Simpson diversities (q = 2) (both of which incorporate relative species abundance for calculating diversity, as opposed to species richness) were significantly higher in natural forests than in plantations. Whereas the same pattern was observed for seedlings, for saplings there was no evidence for significant differences in these indices between the stand types (Figure 6). In most study sites, richness of regenerating tree species (q = 0; seedlings and saplings) appeared to be higher in natural forests than in plantations. However, only at two sites (Natral; Labranza) there was evidence for significant differences, and only in one of them (Natral) for all the range of sample sizes. For Shannon and Simpson diversity (q = 1 and q = 2, respectively), differences were significant at four (Los Corrales; Los Guanacos; Labranza; Natral) and at three sites, respectively (Los Corrales; Los Guanacos; Labranza) (Figure 7).
Overall species diversity of tree regeneration in plantations and natural forests (green and blue curves, respectively), expressed through rarefaction (interpolation) and extrapolation sampling curves of the first three Hill numbers: species richness (q = 0), the exponential of Shannon entropy (q = 1), and the inverse Simpson concentration (q = 2). In each case, diversity is expressed as units of effective number of species (that is, the number of equally abundant species that would be needed to give the same value of the corresponding diversity measure). The reference (effective) sample sizes are represented by dots. The solid segments of the curves represent the interpolated diversity values, whereas the dashed segments represent diversity extrapolated up to double the reference sample sizes of P. radiata stands (312). Shaded areas around the curves represent 95% confidence intervals obtained by 50 bootstrap replications. Sections of the curves in which confidence intervals do not overlap indicate significant differences with p-value < 0.05, although partially overlapping intervals do not guarantee non-significance (Schenker and Gentleman 2001; Chao and others 2014).
Species diversity of tree regeneration in plantations and natural forests (green and blue, respectively) across sites, expressed through rarefaction (interpolation) and extrapolation sampling curves of the first three Hill numbers: species richness (q = 0), the exponential of Shannon entropy (q = 1), and the inverse Simpson concentration (q = 2). For interpretation see Figure 6.
The PERMANOVA test showed a significant difference in composition of tree regeneration among plantations and natural forests (Table 2), whereas this difference was not apparent in the NMDS ordination, which showed a clear overlap between the communities of both environments (Figure 8). Additionally, the PERMANOVA indicated that stand type had only low explanatory power (R2 = 0.03) compared to the site (R2 = 0.22), which also had a significant effect on regeneration composition. According to the site-specific PERMANOVA tests, the difference in composition between plantations and adjacent natural forests was significant only at two sites (Los Corrales; Labranza) (Table 2). In addition, except for those two sites, the site-specific NMDS ordinations showed a clear overlap among the groups of plots from the corresponding stand types (Figure 9).
NMDS ordination of seedling and sapling communities in plantations and natural forests. Green dots represent plots from plantations, whereas blue squares represent plots from natural forests. The graphical distance between two plots represents their Jaccard distance. Three ordination dimensions were used to guarantee convergence within 999 iterations.
NMDS ordinations of the communities of natural regeneration in plantations and their adjacent natural forest across different study sites. Green dots represent plots from the P. radiata plantations, whereas blue squares represent plots from natural forests. The graphical distance between two plots represents their Jaccard distance. All ordinations were two-dimensional except for those of Labranza and La Piragua—Peña Labrada, for which a third dimension was included to guarantee convergence within 999 iterations.
Environmental Conditions of the Stand Types
Tree basal area was significantly higher in P. radiata plantations than in edge plots and natural forests. Yet, mean PPFD below the canopy was significantly lower in natural forests than in plantations, while gap fraction showed no significant differences. Understory cover, in contrast, was significantly higher in natural forests than in edge plots and plantations. The litter layer thickness was significantly higher in plantations than in edge plots and natural forests, whereas soil total N content and CEC were significantly higher in natural forests than in edge plots and plantations (Table 3).
Drivers of Regeneration in P. radiata Plantations
The set of 13 explanatory variables finally included in the model (Table S3) explained 38.5% of the variance in the seedling and sapling community, with an adjusted R2 (unbiased by random correlations) of 0.32. The ANOVA-type permutations showed that the complete CCA model and the first six CCA ordination axes were significant (p-value < 0.05). Canonical axes 1 to 4 had eigenvalues > 0.30, and from the total variance, 16.8% was explained by the first two canonical axes, CCA1 and CCA2. Each of them represented 8.9 and 7.9% of the total variance, and 23.1 and 20.5% of the total explained variance, respectively. All explanatory variables included in the model were significantly related to the species data, except for the percentage of uncovered mineral soil (Table 4). The highest weighted means of scores along the first four canonical axes were shown by the mean lowest annual temperature (0.46), altitude (0.43) and soil pH (0.36). The variables following in importance were related to climate and soil parameters, while PPFD and the variables related to connectivity to natural forest showed only limited importance in the ordination, with average scores below 0.25 (Table 4).
The first canonical axis of the ordination (CCA1) was mostly correlated with minimum annual temperature, altitude, and soil pH, while the second axis (CCA2) was mostly correlated with maximum, minimum and mean annual temperature, soil pH and cation exchange capacity (CEC), and precipitation (Table 4; Figure 10). In the ordination diagram, maximum, minimum and mean annual temperatures increased in the opposite direction of altitude (Figure 10). CEC also increased in this direction. Species showed a strong differentiation along this gradient. The sclerophyllous Lithraea caustica Hook. & Arn., Peumus boldus Molina and Cryptocarya alba (Molina) Looser, and a group of species formed by A. punctatum, Amomyrtus sp, E. cordifolia, Drymis winteri Forst. and M. planipes appear to be linked to higher temperatures and lower altitude, while seeming especially sensitive to annual minimum temperatures. The species Maytenus boaria Molina, Lomatia hirsuta (Lam.) Diels, and N. obliqua were most abundant on the opposite side of this gradient (that is, at lower temperatures and higher altitude). A second distinguishable gradient was the one described by pH and precipitation, with each of these variables increasing in opposite directions. Most species were concentrated at the center of this gradient, while the group of sclerophyllous species was associated with lower precipitation and higher pH values, and the group formed by A. punctatum, Amomyrtus sp, E. cordifolia, D. winteri, and M. planipes appeared to be linked to higher precipitation and lower soil pH. At the same time, these species show higher occurrences at higher soil total N and organic carbon content, both of which appeared correlated. The group of sclerophyllous species, together with M. boaria, L. hirsuta and N. obliqua, was found at the opposite side of this gradient, simultaneously characterized by lower precipitation and lower soil acidity, and particularly, lower values of total N and organic carbon content. Lomatia dentata (Ruiz & Pav.) R. Br., Luma apiculata (A. P. de Candolle) Burret, Aristotelia chilensis (Molina) Stuntz, G. avellana and P. lingue showed little segregation across the diagram, either indicating that they find their optimum in the mid-range of the variables represented, or that they are present everywhere along the gradients described.
Bi-plot of the canonical correspondence analysis, representing regenerating tree species and explanatory variables along the first two canonical axes (CCA1 and CCA2; eigenvalues 0.62 and 0.55, respectively). The axes were scaled based on the species scores (for this reason they differ from those shown in Table 4, where they are scaled based on the variables’ scores). Explanatory variables are represented by blue arrows, whereas species scores are represented by black dots. The arrows point at the direction of maximum variation for the corresponding explanatory variable. The orthogonal projection of the tip of an arrow on a canonical axis indicates the correlation of the corresponding explanatory variable with the axis. The orthogonal projection of a species on an axis indicates the score of the species along the axis, while its projection on an explanatory variable shows the optimum of the species along that variable (ter Braak and Verdonschot 1995; Borcard and others 2011). Only significant variables according to the permutation analysis are represented. Ap = Aextoxicon punctatum; Asp = Amomyrtus sp; Ac = Aristotelia chilensis; Ca = Cryptocarya alba; Dw = Drymis winteri; Ec = Eucryphia cordifolia; Ga = Gevuina avellana; Lc = Lithraea caustica; Ld = Lomatia dentata; Lh = Lomatia hirsuta; La = Luma apiculata; Mb = Maytenus boaria; Mp = Myrceugenia planipes; No = Nothofagus obliqua; Pb = Peumus boldus; Pl = Persea lingue.
Discussion
Abundance of Advance Regeneration and Diameter Distribution of Native Trees
In P. radiata plantations, there was a consistently lower density of total regeneration than in the adjacent natural forests. The edge plots represented a transition zone, with intermediate abundances between both stand types. However, the difference between plantations and natural forests was no longer significant at the sapling stage, given the higher density of saplings vs. seedlings in plantations, and the reverse situation in natural forests.
The declining abundance with increasing height of advance regeneration observed in the natural forest is a common pattern of young, naturally regenerated forests. This can be explained by continuous recruitment on the one side, and high mortality owing to intensive competition among seedlings, browsing, and declining shade tolerance on the other side (Kneeshaw and others 2006; Collet and Le Moguedec 2007). The opposite trend observed in plantations suggests little recruitment of new seedlings after a certain stand age on the one side, and persistence and low mortality of saplings on the other side, possibly due to a high re-sprouting potential. Thus, the cohort of taller seedlings and saplings may have mostly originated during an earlier phase of stand development with more favorable conditions, less competition from P. radiata trees and a more recent seedbank from the original forest (Galloway and others 2017). This may have been the case of A. chilensis, for example (the only species with significantly higher abundance in plantations than in natural forest), which was likely favored by its capacity to rapidly colonize open areas, and to persist after canopy closure (Muñoz and others 2013). In plantations, there are a number of factors that may impede recruitment of seedlings or increase their mortality. Seedbank depletion over time and lower seed dispersal rates into plantations may lead to lower total germination (Kremer and Bauhus 2020). Additionally, basal area was significantly higher in plantations than in natural forests. Although this did not lead to differences in canopy cover, higher root competition and evapotranspiration rates could be leading to low soil water availability, inhibiting seed germination or seedling establishment (Huber and Trecaman 2004; Granados and others 2016). The thick litter layer (> 4 cm) may also prevent germination of newly dispersed species with small-sized seeds (Loydi and others 2013), whereas the generally lower soil fertility may affect establishment and growth (Fenner and Thompson 2005; Katijima and Myers 2008). Lower understory cover may lead to more browsing of new seedlings by livestock and deer (Simonetti and others 2013; Forbes and others 2016a; Burger and others 2019). However, the variety of shade tolerances, dispersal mechanisms, and seed and fruit sizes and types among different tree species showing higher seedling densities in natural forests than in plantations suggest that reduced seedling establishment in plantations is not associated with specific plant traits. Yet, more and better standardized quantitative trait information of different tree species than is available now may allow interpreting the processes behind the overall influence of plantations on seedling density more precisely.
Although some saplings of native species seemed to develop into small trees (< 15 cm dbh), native species were absent from higher diameter classes in the plantation stands. This is most likely attributable to the limited age of plantations (approximately 20 years). Also, while thinning of plantations may provide opportunities for establishment of new seedlings through soil scarification and canopy opening, it can also destroy existing, older regeneration. The low seedling densities observed in plantations suggest that the latest thinning operations, which usually occur around the 12th year after plantation establishment, did not stimulate additional regeneration of native species. However, determining the ages of seedlings and saplings in plantations would be necessary to confirm this.
Regeneration Diversity and Composition
In addition to a lower abundance, the community of regenerating tree species showed a lower diversity in plantations than in natural forests. However, this contrast in diversity was also caused by differences at the seedling stage, not the sapling stage. The difference among seedling communities, in turn, can be largely attributed to differences in the relative abundance of species (that is, their evenness) rather than to their total number, since there was little evidence for differences in species richness at this stage, whereas both Shannon and Simpson diversity indicated significant differences. This suggests that most, if not all native species found in natural forests, can still regenerate in plantations, although some are more disadvantaged than others.
Drivers of Regeneration in P. radiata Plantations
The canonical correspondence analysis explained only a small proportion of variation in the assemblages of regenerating species with their environment, despite the significance of the model and of most of the explanatory variables included. However, since the proportions of explained variation are usually low for environmental data (generally < 10%), the importance of the gradients represented by the CCA should be rather judged based on the eigenvalues of the ordination axes (that is, their relevance for the ordination) and their ecological interpretability (ter Braak and Verdonschot 1995). Given that both the first and second CCA axes (that is, the linear combinations of environmental variables that best explain the composition and abundance of regeneration) were simultaneously related to similar explanatory variables, they could not be independently associated with any specific environmental gradient. The gradients with highest influence on regeneration were represented by climatic variables (temperature and precipitation), altitude, and soil chemical properties (pH, organic carbon, CEC and total N). In contrast, variables related to canopy structure or connectivity to natural forest remnants showed little influence. Yet, canopy structure and distance to remnants of natural forest have been usually reported as important determinants of natural regeneration, both in plantations in general (Kremer and Bauhus 2020), and specifically in P. radiata plantations (García and others 2016; Forbes and others 2016b; Forbes and others 2019; Kremer and others 2021). In our study, the particularly low levels of gap fraction in plantations (4% on average) were most likely limiting for establishment and growth of seedlings and saplings. Nevertheless, the lack of environmental variation within structurally homogenous plantations may have hindered the expression of any possible relationships between abundance, diversity and composition of regeneration, and light availability. This may explain the diverging results of previous studies conducted in P. radiata plantations, where environmental variation was either experimentally manipulated (Forbes and others 2016b; Kremer and others 2021) or deliberately captured through stratified sampling of stands of different ages (Forbes and others 2019). The low influence of the connectivity to natural forest remnants also contrasts with the previous finding that seed rain in P. radiata plantations is positively affected by the proximity of natural forests (García and others 2016). This may suggest that regeneration in Chilean P. radiata plantations is less dependent on seed rain than on legacies for vegetative reproduction, as was frequently observed in the field. However, since the origin of seedlings and saplings (vegetative vs. generative) was often difficult to ascertain, this variable could not be incorporated in our analyses.
Given the spatial resolution of climate data employed (approximately 5 km), the temperature and precipitation gradients shown by the CCA output most likely reflect only the landscape-scale variation in these factors. The same may apply to the observed pH gradient, given its usual association with soil parent material and precipitation (Sadzawka and others 1995; Blume and others 2010, p.192). Although these overriding patterns restrict the analysis of conditions for regeneration of native species at the site scale, they suggest that a set of sites with a similarly wide range of values along these gradients should be included among restoration plans to guarantee the presence of different species and floristic associations across the landscape. At the same time, however, it may imply that sites with intermediate conditions along these gradients may provide suitable conditions for a high richness of native species in advance regeneration of plantations, especially considering that most species were concentrated near the central values of both gradients. Meanwhile, species located at the extremes of these gradients (for example, Maytenus boaria for the temperature-altitude and Lithraea caustica for precipitation-pH) were neither frequent nor abundant in plantations and natural forests (that is, the reference ecosystems) and thus may not be representative of the ecosystems to restore.
Similar to climatic variables, the segregation of species along the gradient described by organic C and total N contents seems to reflect a latitudinal pattern, rather than a site-scale variation (Figure S3). In contrast, CEC did not show a clear latitudinal pattern (Figure S3), suggesting its variability has a stronger local component. However, apart from the sclerophyllous species group, no other species showed requirements of CEC above average. It is therefore unlikely that soil chemical properties were limiting regeneration of tree species.
Despite the apparently low effect of structural attributes of plantations on regeneration, differences in site conditions between natural forests and plantations suggest that site-level interventions (for example, removal of canopy trees, livestock exclusion, soil amendments) could promote recruitment, in view of the higher density and diversity of seedlings in natural forest stands when compared to plantations. However, these results are rather explorative, and specific guidelines for restoration should be based on experimental manipulations of variables that are potentially limiting survival and establishment of native tree species.
Potential for Passive Restoration
The current structure and composition of the sampled native forest remnants are indicative for lowland Nothofagus secondary forests, which gradually develop a unimodal diameter distribution before gap creation enables a higher establishment of more shade-tolerant species (Veblen 1985; Donoso 1993). If left to develop naturally, these stands will likely progress toward states of higher structural and functional diversity. The high similarity in advance regeneration between P. radiata plantations and sampled remnant natural forests at the level of saplings suggests that the advance regeneration found in plantations could form the basis for a passive restoration approach toward secondary native forests. It may be argued that our results apply only to stands in relatively close proximity to native forests, and that plantation stands at greater distance from potential seed sources than the stands sampled here may show lower abundance of natural regeneration. However, as discussed above, our results revealed limited influence of the distance and connectivity to natural remnants on regeneration abundance and composition, suggesting that abundance and composition of the regeneration in plantations are largely determined by legacies for vegetative reproduction rather than by ongoing seed dispersal. Yet, the effect of proximity to natural forests may increase after total or partial canopy removal, and thus, it should be further evaluated under such conditions.
Given the variety of shade tolerances of the regenerating species present (Figure 5), partial harvesting approaches that promote microsite variability and light heterogeneity, while maintaining advance regeneration, may be ecologically most appropriate to promote an overall development of native species. Partial canopy removal in the form of strip cuttings has already shown clear ecological advantages over clearcutting or full canopy retention for planted seedlings of native tree species in Chilean P. radiata plantations (Kremer and others 2021), and a similar response may thus be expected from natural regeneration (Chan 2006; Cummings and others 2007; Seiwa 2012). However, after a prolonged period of suppression (~ 20 years until harvesting age), a proportion of the advance regeneration in plantations may no longer be able to respond to release (Coban and others 2016), in which case the establishment of new plants would be required. Given the low abundance of seedlings in plantations, the recruitment of new seedlings would rely on germination from the seedbank, dispersal from nearby native forest remnants and an increased seed production from retained small native trees following plantation removal (Válio and Scarpa 2001; McAlpine and Drake 2003; García and others 2016). In view of the variety of species with different shade tolerance traits, a range of different responses of regeneration to total or partial canopy removal should be expected. Eventually, further emergence of sprouts from stumps and roots could also be expected (Kormanik and Brown 1967; Deal and others 2003; Promis and others 2019). Nevertheless, these responses are usually species specific (Berdanier and Clark 2016), and the sprouting potential of stumps of native tree species may be weakened after two decades following harvesting (Šplíchalová and others 2012), in which case enrichment planting may be necessary. To determine the overall response of natural regeneration to canopy interventions, further research involving different harvesting approaches with different levels of canopy removal would be required.
Conclusions
Our results suggest that commercially mature (ca. 20 yr. old) first rotation P. radiata plantations established after clearing of natural forests harbor advance regeneration of native tree species that is largely representative of nearby second-growth natural forests. Obviously, the regeneration layer shows a high persistence and capacity to recover after plantation establishment. Our findings, based on a comprehensive sampling of the plantations to be managed for restoration, support the idea that these stands have the potential to develop into forests dominated by native tree species by relying on advance regeneration below their canopies. Thus, a complete stand re-initiation through plantation clearing and subsequent planting may not be necessary, eventually reducing the total time required to restore the natural vegetation. However, given the differences in density and diversity of seedlings between plantations and natural forests, a continuous recruitment of most species of the native tree community would depend on the supplementary establishment of new plants, either through enrichment planting, increased germination or re-sprouting following complete or partial canopy removal of plantations. Since the germination and re-sprouting potentials are unknown, further research on the response of these processes following canopy openings is required. While our study was designed to capture the variation in typical, commercially mature P. radiata plantations in the study region in south-central Chile and allowed us to confirm the potential for passive restoration of these plantations, we did not specifically explore the biological or economical limits of this restoration approach with pines as a nurse crop. Hence, we emphasize the need to explore these limits including the response of advance regeneration to contrasting levels of canopy cover. In addition to determining optimal harvesting approaches, this would also enable studying the influence of the environmental variables assessed here under a variety of conditions.
References
AGRIMED. 2017. Atlas agroclimático de Chile. Santiago: Universidad de Chile.
Balestrin B, Moreira A, Procknow D, Camargo B, Degrandi M, Puhl J, de Moraes M. 2020. Natural regeneration as an indicator of ecological restoration by applied nucleation and passive restoration. Ecol Eng 157:105991.
Bannister JR, Donoso PJ, Mujica R. 2016. Silviculture as a tool for restoration of temperate forests. Bosque 37:229–235.
Baral H, Guariguata MR, Keenan RJ. 2016. A proposed framework for assessing ecosystem goods and services from planted forests. Ecosyst Services 22, Part B:260–268.
Bauhus J, Pokorny B, Van der Meer P, Kanowski P, Kanninen M. 2010. Ecosystem good and services – the key for sustainable plantations. Bauhus J, Van der Meer P, Kanninen M, editors. Ecosystem goods and services from plantation forests. London: Earthscan. p205–227.
Becerra PI, Montenegro G. 2013. The widely invasive tree Pinus radiata facilitates regeneration of native woody species in a semi-arid ecosystem. Appl Veg Sci 16:173–183.
Berdanier AB, Clark JS. 2016. Divergent reproductive allocation trade-offs with canopy exposure across tree species in temperate forests. Ecosphere 7:1–10.
Blume H-P, Brümmer GW, Horn R, Kandeler E, Kögel-Knabner I, Kretzschmar R, Stahr K, Wilke B-M. 2010. Scheffer/Schachtschabel: Lehrbuch der Bodenkunde, 17th edn. Heidelberg: Springer Spektrum.
Bolker B, Team RDC, Giné-Vázquez I. 2020. bbmle: Tools for General Maximum Likelihood Estimation. https://cran.r-project.org/web/packages/bbmle/bbmle.pdf.
Borcard D, Gillet F, Legendre P. 2011. Numerical ecology with R. New York: Springer.
Brockerhoff EG, Jactel H, Parrotta JA, Ferraz SFB. 2013. Role of eucalypt and other planted forests in biodiversity conservation and the provision of biodiversity-related ecosystem services. For Ecol Manag 301:43–50.
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A, Skaug HJ, Machler M, Bolker BM. 2017. glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400.
Burger A, Bannister JR, Galindo N, Vargas-Gaete R, Vidal OJ, Schlegel B, Burger A, Bannister JR, Galindo N, Vargas-Gaete R, Vidal OJ, Schlegel B. 2019. Evidencia de ramoneo por el ciervo nativo y casi amenazado Pudu puda en plantaciones de restauración en la Isla de Chiloé, Chile. Gayana Botánica 76:24–33.
Bustamante RO, Simonetti JA. 2005. Is Pinus radiata invading the native vegetation in central Chile? Demographic responses in a fragmented forest. Biol Invas 7:243–249.
Chan SS, Larson DJ, Maas-Hebner KG, Emmingham WH, Johnston SR, Mikowski DA (2006) Overstory and understory development in thinned and underplanted Oregon Coast Range Douglas-fir stands. Can J Res 36:2696–2711.
Chao A, Gotelli NJ, Hsieh TC, Sander EL, Ma KH, Colwell RK, Ellison AM. 2014. Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67.
Chen G, Yang Y, Xie J, Guo J, Gao R, Qian W. 2005. Conversion of a natural broad-leafed evergreen forest into pure plantation forests in a subtropical area: effects on carbon storage. Ann for Sci 62:659–668.
Coban S, Colak AH, Rotherham ID. 2016. Interactions between canopy cover density and regeneration cores of older saplings in Scots pine (Pinus sylvestris L.) stands. Forest Systems 25:073.
Collet C, Le Moguedec G. 2007. Individual seedling mortality as a function of size, growth and competition in naturally regenerated beech seedlings. Forestry 80:359–370.
CONAF. 2017. Superficies de uso de suelo regional. https://www.conaf.cl/nuestros-bosques/bosques-en-chile/catastro-vegetacional/.
CR2. 2018. Productos grillados - Centro de Ciencia del Clima y la Resiliencia - CR2. http://www.cr2.cl/datos-productos-grillados/.
Cummings J, Reid N, Davies I, Grant C. 2007. Experimental manipulation of restoration barriers in abandoned eucalypt plantations. Restor Ecol 15:156–167.
Deal RL, Barbour RJ, Mcclellan MH, Parry DL. 2003. Development of epicormic sprouts in Sitka spruce following thinning and pruning in south-east Alaska. Forestry 76:401–412.
Dellasala D, Martin A, Spivak R, Schulke T, Bird B, Criley M, van Daalen C, Kreilick J, Brown R, Aplet G. 2003. A citizen’s call for ecological forest restoration: forest restoration principles and criteria. Ecol Rest 21:14–23.
Domec J-C, King JS, Ward E, Christopher Oishi A, Palmroth S, Radecki A, Bell DM, Miao G, Gavazzi M, Johnson DM, McNulty SG, Sun G, Noormets A. 2015. Conversion of natural forests to managed forest plantations decreases tree resistance to prolonged droughts. For Ecol Manag 355:58–71.
Donoso C. 1993. Bosques templados de Chile y Argentina. Variación, estructura y dinámica, 4th edn. Santiago: Editorial Universitaria.
Echeverría C, Fuentes R, Heilmayr. 2019. Cambios de uso y cobertura del suelo en la Cordillera de la Costa del centro-sur de Chile entre 1986 y 2011. Smith-Ramírez C, Squeo FA, editors. Biodiversidad y ecología de los bosques costeros de Chile. Santiago: Universidad de Los Lagos Editorial. p471–486.
Eguiguren F. 1995. Caracterización florística del sotobosque en plantaciones de Pinus radiata D. Don de la provincia de Arauco, VIII región. Undergraduate thesis, Universidad de Chile.
Estades C, Escobar MA. 2005. Los ecosistemas de las plantaciones de pino de la Cordillera de la Costa. Smith-Ramírez C, Armesto JJ, Valdovinos C, editors. Historia, biodiversidad y ecología de los bosques costeros de Chile. Santiago: Editorial Universitaria. p600–616.
Fenner M, Thompson K. 2005. The ecology of seeds. Cambridge: Cambridge University Press.
Forbes AS, Norton DA, Carswell F. 2019. Opportunities and limitations of exotic Pinus radiata as a facilitative nurse for New Zealand indigenous forest restoration. New Zealand J for Sci 49:1–14.
Forbes AS, Norton DA, Carswell FE. 2016a. Tree fern competition reduces indigenous forest tree seedling growth within exotic Pinus radiata plantations. For Ecol Manag 359:1–10.
Forbes AS, Norton DA, Carswell FE. 2016b. Artificial canopy gaps accelerate restoration within an exotic Pinus radiata plantation. Restor Ecol 24:336–345.
Forestal Arauco S.A. 2018. Plan de Restauración Bosque Nativo Arauco. https://www.arauco.cl/chile/wp-content/uploads/sites/14/2018/04/EG14.9_Plan_de_Restauracio%25CC%2581n_de_Bosque_Nativo_Arauco_2018.03.26_v1.pdf.
Forestal Mininco S.A. 2017. Plan de ordenación Forestal Mininco S.A. http://www.mininco.cl/SGAEX/pages/abrearchivo2.asp?arch=archivos/41/Plan_Ordenacion_2017.pdf.
FSC. 2015. FSC Principles and Criteria for Forest Stewardship. https://fsc.org/en/about-us.
Galloway AD, Holmes PM, Gaertner M, Esler KJ. 2017. The impact of pine plantations on fynbos above-ground vegetation and soil seed bank composition. South Afr J Bot 113:300–307.
García V, Simonetti J, Becerra P. 2016. Lluvia de semillas, depredación de semillas y germinación de especies nativas en plantaciones de Pinus radiata en Chile centro-sur: efecto de la distancia a bosque nativo y presencia de sotobosque. Bosque 37:359–367.
Geldenhuys CJ, Atsame-Edda A, Mugure MW. 2017. Facilitating the recovery of natural evergreen forests in South Africa via invader plant stands. For Ecosyst 4:21.
Gómez P, Hahn S, San Martín J. 2009. Estructura y composición florística de un matorral bajo plantaciones de Pinus radiata D. Don En Chile. Central. Gayana Botánica 66:256–268.
Granados ME, Vilagrosa A, Chirino E, Vallejo VR. 2016. Reforestation with resprouter species to increase diversity and resilience in Mediterranean pine forests. For Ecol Manag 362:231–240.
Hånel B. 2008. Regeneration of Picea abies forests on highly productive peatlands—Clearcutting or selective cutting? Sandinavian J for Res 8:518–527.
Heilmayr R, Echeverria C, Fuentes R, Lambin EF. 2016. A plantation-dominated forest transition in Chile. Appl Geogr 75:71–82.
Heinrichs S, Pauchard A, Schall P. 2018. Native Plant Diversity and Composition Across a Pinus radiata D.Don Plantation Landscape in South-Central Chile—The Impact of Plantation Age, Logging Roads and Alien Species. Forests 9:567.
Hsieh TC, Chao KHM and A. 2020. iNEXT: Interpolation and Extrapolation for Species Diversity. https://cran.r-project.org/web/packages/iNEXT/iNEXT.pdf.
Hsieh TC, Ma KH, Chao A. 2016. iNEXT: an R package for rarefaction and extrapolation of species diversity (Hill numbers). Methods Ecol Evol 7:1451–1456.
Huber A, Trecaman R. 2004. Eficiencia del uso del agua en plantaciones de Pinus radiata en Chile. Bosque 25:33–43.
Holl KD, Aide TM. 2011. When and where to actively restore ecosystems? For Ecol Manag 261:1558–1563.
Katijima K, JA Myers. 2008. Seedling ecophysiology: strategies toward achievement of positive net carbon balance. Leck MA, Parker TV, Simpson RL, Simpson RS, editors. Seedling ecology and evolution. Cambridge: Cambridge University Press. p172–188.
Kneeshaw DD, Kobe RK, Coates KD, Messier C. 2006. Sapling size influences shade tolerance ranking among southern boreal tree species. J Ecol 94:471–480.
König N 2005. Handbuch Forstliche Analytik. Bundesministerium für Verbraucherschutz, Ernährung und Landwirtschaft. Göttingen: Niedersächsische Forstliche Versuchsanstalt.
Kormanik PP, Brown CL. 1967. Root Buds and the Development of Root Suckers in Sweetgum. For Sci 13:338–345.
Kremer KN, Bannister JR, Bauhus J. 2021. Restoring native forests from Pinus radiata plantations: Effects of different harvesting treatments on the performance of planted seedlings of temperate tree species in central Chile. For Ecol Manag 479:118585.
Kremer KN, Bauhus J. 2020. Drivers of native species regeneration in the process of restoring natural forests from mono-specific, even-aged tree plantations: a quantitative review. Restor Ecol 28:1074–1086.
Lara A, Veblen. 1993. Forest plantations in Chile: a successful model? Smith A, editor. Afforestation: policies, planning and progress. London: Belhaven Press. p118–139.
Lemenih M, Bongers F. 2010. The role of plantation forests in fostering ecological restoration: Experiences from East Africa. Bongers F, Tennigkeit T, editors. Degraded forests in Africa: Management and restoration. New York: Earthscan. p171–220.
Loydi A, Eckstein RL, Otte A, Donath TW. 2013. Effects of litter on seedling establishment in natural and semi-natural grasslands: a meta-analysis. J Ecol 101:454–464.
Luebert F, Pliscoff P. 2005. Bioclimas de la Cordillera de la Costa del centro-sur de Chile. Smith-Ramírez C, Armesto JJ, Valdovinos C, editors. Historia, biodiversidad y ecología de los bosques costeros de Chile. Santiago: Editiorial Universitaria. p60–74.
Luebert F, Pliscoff P. 2006. Sinopsis bioclimática y vegetacional de Chile. Santiago: Editorial Universitaria.
Luzio W. 2010. Suelos de Chile. Santiago: Universidad de Chile.
Magnusson A, Skaug H, Nielsen A, Berg C, Kristensen K, Maechler M, Bentham K van, Bolker B, Sadat N, Lüdecke D, Lenth R, O’Brien J, Brooks M. 2020. glmmTMB: Generalized Linear Mixed Models using Template Model Builder. https://cran.r-project.org/web/packages/glmmTMB/glmmTMB.pdf.
McAlpine K, Drake D. 2003. The effects of small-scale environmental heterogeneity on seed germination in experimental treefall gaps in New Zealand. Plant Ecol 165:207–215.
McAlpine K, Lamoreaux S, Timmins SM. 2020. Understory vegetation provides clues to succession in woody weed stands, New Zealand. J Ecol 45:3418.
Melnick D, Bookhagen B, Strecker MR, Echtler HP. 2009. Segmentation of megathrust rupture zones from fore-arc deformation patterns over hundreds to millions of years, Arauco peninsula, Chile. J Geophys Res 114:B01407.
Muñoz A, González M, Díaz-Hormazábal I, Riquelme NI. 2013. Aristotelia chilensis (Mol) Stunz. Donoso C, editor. Las especies arbóreas de los bosques templados de Chile y Argentina - Autoecología. 2nd edn. Valdivia: Marisa Cuneo Ediciones. p166–173.
Myers N, Mittermeier RA, Mittermeier CG, Da Fonseca GA, Kent J. 2000. Biodiversity hotspots for conservation priorities. Nature 403:853–858.
NGP. 2020. New Generation plantations - 2020 review. https://drive.google.com/file/d/1gLujfexmpxN4Zt0elYhb6FnoWYjhbHkP/view.
Niklitschek ME. 2007. Trade Liberalization and land use changes: explaining the expansion of afforested land in Chile. For Sci 53:385–394.
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H. 2019. vegan: Community Ecology Package. https://cran.r-project.org/web/packages/vegan/vegan.pdf.
Otero L. 2006. La huella del fuego. Santiago: Pehuén.
Paquette A, Messier C. 2010. The role of plantations in managing the world’s forests in the Anthropocene. Front Ecol Environ 8:27–34.
Parrotta JA, Turnbull JW, Jones N. 1997. Catalyzing native forest regeneration on degraded tropical lands. For Ecol Manag 99:1–7.
PEFC. 2018. Sustainable Forest Management – Requirements. https://cdn.pefc.org/pefc.org/media/2019-01/b296ddcb-5f6b-42d8-bc98-5db98f62203e/6c7c212a-c37c-59ee-a2ca-b8c91c8beb93.pdf.
Pirard R, Dal Secco L, Warman R. 2016. Do timber plantations contribute to forest conservation? Environ Sci Policy 57:122–130.
Pliscoff P, Fuentes-Castillo T. 2011. Representativeness of terrestrial ecosystems in Chile’s protected area system. Environ Conserv 38:303–311.
PNUD. 2017. Catálogo de las especies exóticas asilvestradas/naturalizadas en Chile. Laboratorio de Invasiones Biológicas: Universidad de Concepción, Santiago de Chile.
Prado JA. 2015. Plantaciones Forestales - Más allá de los árboles. Santiago: Colegio de Ingenieros Forestales.
Promis A, Olivares S, Acuña S, Cruz G. 2019. Respuesta temprana de la regeneración de plantas leñosas después del incendio forestal denominado “Las Máquinas” en la Región del Maule, Chile. Gayana Botánica 76:257–262.
Ramírez C, Figueroa H, Carrillo R, Contreras DC. 1984. Estudio fitosociológico de los estratos inferiores en un bosque de pino (Valdivia, Chile). Bosque 5:65–81.
Sadzawka J, Peralta M, Ibarra M, Peralta JM, Fuentes JP. 1995. Características químicas de suelos forestales chilenos. Bosque 16:9–28.
Salas C, Donoso PJ, Vargas R, Arriagada CA, Pedraza R, Soto DP. 2016. The forest sector in Chile: An overview and current challenges. J for 114:562–571.
Schenker N, Gentleman JF. 2001. On judging the significance of differences by examining the overlap between confidence intervals. Am Stat 55:182–186.
Schroth G, D’Angelo SA, Teixeira WG, Haag D, Lieberei R. 2002. Conversion of secondary forest into agroforestry and monoculture plantations in Amazonia: consequences for biomass, litter and soil carbon stocks after 7 years. For Ecol Manag 163:131–150.
Seiwa K, Yukino E, Hishita M, Masaka K. 2012. Effects of thinning intensity on species diversity and timber production in a conifer (Cryptomeria japonica) plantation in Japan. J for Res 17:468–478.
Silva LN, Freer-Smith P, Madsen P. 2019. Production, restoration, mitigation: a new generation of plantations. New for 50:153–168.
Simonetti J, Grez A, Estades CF. 2013. Providing habitat for native mammals through understory enhancement in forestry plantations. Conserv Biol 27:1117–1121.
Smith-Ramírez C, Teillier S, Jiménez J, Barahona RM, Parra LE, Jerez V. 2019. Plantas y animales endémicos de la Cordillera de la Costa de Chile. Smith-Ramírez C, Squeo FA, editors. Biodiversidad y Ecología de los Bosques Costeros de Chile. Santiago: Universidad de Los Lagos Editorial. p393–416.
Šplíchalová M, Adamec Z, Kadavý J, Kneifl M. 2012. Probability model of sessile oak (Quercus petraea (Matt.) Liebl.) stump sprouting in the Czech Republic. Eur J for Res 131:1611–1618.
Ter Braak CJF, Verdonschot PFM. 1995. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat Sci 57:255–289.
Tzu-Yang L, Vadeboncoeur MA, Kuo-Chuan L, Ming-Zhen C, Huang M-Y, Lin T-C. 2015. Understorey plant community and light availability in conifer plantations and natural hardwood forests in Taiwan. Appl Veg Sci 2015:591–602.
Válio IFM, Scarpa FM. 2001. Germination of seeds of tropical pioneer species under controlled and natural conditions. Braz J Bot 24:79–84.
Veblen T. 1985. Stand dynamics in Chilean Nothofagus forests. Picket S, White P, editors. The ecology of natural disturbance and patch dynamics. Orlando: Academic Press, Inc. p35–51.
Veblen TT, Donoso C, Schlegel FM, Escobar B. 1981. Forest dynamics in South-Central Chile. J Biogeogr 8:211–247.
Weinzierl W, Dietze G. 2000. Bestimmung der austauschbaren Kationen ohne Veränderung des natürlichen Boden-pH, auch effektive Kationenaustauschkapazität genannt. Freiburg: Landesamt für Geologiie, Rohstoffe und Bergbau.
WWF. 2013. Informe Técnico de sustitución de bosque nativo y matorral arborescente en el patrimonio de Forestal MASISA S.A. http://awsassets.panda.org/downloads/informe_tecnico_de_sustitucion_de_bosque_nativo_y_matorral_arborescente_en_el_patrimonio_1.pdf.
Acknowledgements
Klaus Kremer received a DAAD-CONICYT scholarship, which supported his doctoral studies at the Faculty of Environment and Natural Resources, University of Freiburg. Additionally, the fieldwork of this study was conducted with the support of the Georg-Ludwig-Hartig Stiftung and the SuFoRun project. We are thankful to Forestal Arauco S.A. for their commitment to this study, particularly to Jocelyn Gallegos, Boris Fica and Gustavo Escobar. We also thank Jeanette Nothstein, Daniela Gatica, Alex Kremer, Cristabel Durán, Mercedes Salas, Georgios Skiadaresis, Daniel Uteau and Jan Bannister. We are especially grateful to Luz Ramírez for her continued support.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kremer, K., Promis, Á. & Bauhus, J. Natural Advance Regeneration of Native Tree Species in Pinus radiata Plantations of South-Central Chile Suggests Potential for a Passive Restoration Approach. Ecosystems 25, 1096–1116 (2022). https://doi.org/10.1007/s10021-021-00704-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-021-00704-x