Abstract
Free air CO2 enrichment (FACE) experiments in aggrading temperate forests and plantations have been initiated to test whether temperate forest ecosystems act as sinks for anthropogenic emissions of CO2. These FACE experiments have demonstrated increases in net primary production and carbon (C) storage in forest vegetation due to increased atmospheric CO2 concentrations. However, the fate of this extra biomass in the forest floor or mineral soil is less clear. After 6 years of FACE treatment in a short-rotation poplar plantation, we observed an additional sink of 32 g C m−2 y−1 in the forest floor. Mineral soil C content increased equally under ambient and increased CO2 treatment during the 6-year experiment. However, during the first half of the experiment the increase in soil C was suppressed under FACE due to a priming effect, that is, the additional labile C increased the mineralization of older SOM, whereas during the second half of the experiment the increase in soil C was larger under FACE. An additional sink of 54 g C m−2 y−1 in the top 10 cm of the mineral soil was created under FACE during the second half of the experiment. Although, this FACE effect was not significant due to a combination of soil spatial variability and the low number of replicates that are inherent to the present generation of forest stand FACE experiments. Physical fractionation by wet sieving revealed an increase in the C and nitrogen (N) content of macro-aggregates due to FACE. Further fractionation by density showed that FACE increased C and N contents of the light iPOM and mineral associated intra-macro-aggregate fractions. Isolation of micro-aggregates from macro-aggregates and subsequent fractionation by density revealed that FACE increased C and N contents of the light iPOM, C content of the fine iPOM and C and N contents of the mineral associated intra-micro-aggregate fractions. From this we infer that the amount of stabilized C and N increased under FACE treatment. We compared our data with published results of other forest FACE experiments and infer that the type of vegetation and soil base saturation, as a proxy for bioturbation, are important factors related to the size of the additional C sinks of the forest floor–soil system under FACE.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Temperate forest ecosystems are hypothesized to be large sinks for anthropogenic emissions of CO2 due to regrowth and CO2 fertilization (Houghton 2003; Houghton and others 1998; Janssens and others 2003; Schimel 1995). To test this hypothesis, free air CO2 enrichment (FACE) experiments in aggrading temperate forests and plantations have been initiated, including: Duke Forest (treatment initiation in 1996), NC, USA (Pinus taeda); Aspen FACE (1997), Rhinelander, WI, USA (Populus tremuloides, Acer saccharum, Betula papyrifera); Oak Ridge (1998), TN, USA (Liquidambar styraciflua); POP-EuroFACE (1999), Italy (Populus alba, Populus nigra, Populus euramericana); Swiss Canopy Crane web-FACE (1999; Fagus, Quercus, Carpinus, Tilia); Stillberg treeline FACE (2001), Switzerland (Larix decidua, Pinus uncinata); Bangor FACE (2005, no published data yet), Wales, UK (Betula pendula, Alnus glutinosa, Fagus sylvatica). These FACE experiments have demonstrated increases in net primary production (NPP) and C storage in forest vegetation due to increased atmospheric CO2 concentrations (Calfapietra and others 2003; DeLucia and others 1999; Gielen and others 2005; Hamilton and others 2002; Handa and others 2005, 2006; Hättenschwiler and others 2002; Karnosky and others 2003; Liberloo and others 2006a, b; Norby and others 2002, 2005). In contrast, Körner and others (2005) merely observed an increased flux of non-structural carbon (C) through mature forest trees under FACE without an increase in stem growth and leaf litter production.
In general, the aboveground biomass contributes through senescence and dieback to the forest litter layer. This litter may in part be incorporated into the mineral soil, ranging from negligible amounts to almost all depending on bioturbation in the top soil. Belowground biomass contributes through root turnover to the mineral soil and if present also to the lower layers of the forest floor. The extra C uptake due to increased atmospheric CO2 concentrations may therefore, next to forest vegetation, also be stored in forest floor litter and mineral forest soil.
At the Duke Forest, Schlesinger and Lichter (2001) observed a significant additional sink of 183 g C m−2 in the forest floor after 3 years of FACE treatment. However, C content of the mineral soil was not affected by FACE. Later on, Lichter and others (2005) reported an additional sink of 52 g C m−2 y−1 in the forest floor under FACE at the Duke Forest averaged over a period of 6 years. But also after 6 years, FACE did not affect C and nitrogen (N) contents of the mineral soil.
At Rhinelander, elevated CO2 increased leaf litter production by 36% in aspen and doubled birch leaf litter production in the aspen-birch stand relative to control (Karnosky and others 2003). FACE also increased the mass of dead fine roots by 140% beneath aspen and by 340% beneath aspen-birch (King and others 2001). Stable soil C formation under FACE was 50% greater than under FACE + increased O3 concentration after 4 years, where the combination with increased O3 treatment largely canceled the extra C input into the soil due to FACE alone (Loya and others 2003).
Soil C of the top 5 cm of the mineral soil at Oak Ridge increased by, respectively, 35 and 255 g C m−2 under ambient and elevated CO2 over a period of 5 years (Jastrow and others 2005). The difference during these 5 years, that is, the accrual of 220 g C m−2, together with the replacement of pretreatment soil C in the FACE rings of 221 g C m−2 (determined by using stable C isotopes) resulted in a total input of new FACE-derived soil C of 441 g C m−2. FACE did not affect soil C contents below 5-cm depth.
During the first rotation (1999–2001) of the POP-EuroFACE project, total soil C content increased under ambient CO2 and FACE treatment, respectively, with 12 and 3%, that is, 484 and 107 g C m−2 (Hoosbeek and others 2004). We estimated the input of new C with the C3/C4 stable isotope (δ13C) method. Respectively, 704 and 926 g C m−2 of new C was incorporated under control and FACE during the 3-year experiment. Although more new C was incorporated under FACE, the increase in total C was suppressed under FACE. We hypothesized that these seemingly opposite effects may have been caused by a priming effect of the newly incorporated litter, where priming effect is defined as the stimulation of SOM decomposition caused by the addition of labile substrates. In 2002, the experiment continued with a second 3-year rotation. Chemical fractionation revealed an increase in the labile C fraction at 0–10-cm depth due to FACE treatment (Hoosbeek and others 2006a), which is in agreement with the larger input of plant litter and root exudates under FACE (Liberloo and others 2006a; Lukac and others 2003). Also respiration measurements in combination with the application of a SOM model revealed that more metabolizable C was present in FACE soil (Hoosbeek and others 2007).
The fate of FACE induced additional C allocated belowground remains unclear (Jastrow and others 2005; Lichter and others 2005). Enhanced C transfer to the root system may result mainly in enhanced root respiration or, otherwise, in an increase in root dry matter, mycorrhizal activity, and subsequent transfer of C to soil C pools. The stability of soil organic matter is controlled by the chemical structure of the organic matter and the existence of protection offered by the soil matrix and minerals (Baldock and Skjemstad 2000; Davidson and Janssens 2006; Elliott 1986; Jastrow 1996; Krull and others 2003; Six and others 2002; Van Veen and Kuikman 1990). The additional C input may affect population size and activity of soil fauna and flora, and may therefore also affect the formation of soil aggregates (Oades 1993; Prior and others 2004; Rillig and others 1999). Oades (1984, 1993) suggested a model of aggregate formation in which micro-aggregates (~100 μm in diameter) are formed within macro-aggregates (>250 μm in diameter). This model of the cycle of aggregate formation has been extended and applied by Jastrow (1996), Puget and others (1995), and Six and others (1998, 1999, 2001, 2002). Fresh plant remains entering the soil become sites for microbial activity and nucleation centers for aggregation. The enhanced microbial activity induces the binding of organic matter and soil particles into macro-aggregates. As the enclosed organic matter is decomposed, microbial and decomposition products become associated with mineral particles (Chenu and Stotzky 2002). This association results in the formation of micro-aggregates. Eventually, the binding agents in macro-aggregates degrade, resulting in a breakdown of macro-aggregates and the release of microbially processed organic matter and micro-aggregates. These micro-aggregates play a key role in the protection and stabilization of SOM by providing an environment in which organic residues may be bonded to mineral surfaces forming stable organo-mineral complexes.
After 6 years of CO2 enrichment at the Duke Forest FACE experiment, Lichter and others (2005) observed that the C content of the mineral top soil (0–15 cm) averaged over the FACE and control rings significantly increased during the experiment. Physical fractionation suggested that this increase occurred entirely within the free light fraction (LF) in which organic C is not protected against decomposition. Fractions in which soil C is protected to some degree, that is, coarse and fine intra-aggregate particulate organic matter (iPOM) and mineral-associated organic matter, were not affected by FACE.
At Oak Ridge, the proportion of soil C found in micro-aggregates averaged 58% in both FACE and ambient plots and did not change over time (Jastrow and others 2005). This implies that the extra FACE induced C input into the soil was protected in the same manner as under ambient conditions.
Physical fractionation of soil samples collected after 5 years of treatment at the POP-EuroFACE experiment revealed that FACE did not affect the weight, C and N contents of the macro- and micro-aggregate fractions, or the micro-aggregate fractions isolated from the macro-aggregates (Hoosbeek and others 2006b). Again, this means that the extra C input under FACE was in part protected and stabilized in a similar fashion as the C input under ambient conditions. Poplar genotype did however have an effect on aggregate formation, that is, under P. euramericana the formation of free micro-aggregates increased which means that more newly incorporated soil C was stabilized and protected.
Because N availability commonly limits forest productivity, some combinations of increased N uptake from the soil and more efficient use of the N already assimilated by trees are necessary to sustain the high rates of forest NPP under FACE (Finzi and others 2007). At the Duke Forest, Oak Ridge and Rhinelander FACE site the uptake of N increased under elevated CO2, yet fertilization studies at the Duke and Oak Ridge sites showed that tree growth and forest NPP were limited by N availability. In contrast, N-use efficiency increased under elevated CO2 during the first 3 years at the POP-EuroFACE site, where fertilization studies showed that N was not limiting to tree growth (Calfapietra and others 2007). In general, N is also needed for the long-term storage of C in stable organic matter fractions in the forest floor and mineral soil. After all, the C/N ratios of organic matter fractions are always lower than those of the originating plant biomass. In a FACE experiment on fertilized grassland in Switzerland, Van Groenigen and others (2002) found that, based on isotopic measurements, the sequestration of new C and N under FACE was highly correlated. They suggested that new N was used to sequester new C. Moreover, in a meta-analysis based on 65 studies, Van Groenigen and others (2006) found that soil C only increases under elevated CO2 when N is added at rates well above typical atmospheric deposition.
Over a period of 6 years, forest floor C content at Duke increased due to FACE, whereas N content was not affected (Lichter and others 2005). At the same time, C concentration did not change due to FACE, whereas N concentration decreased. As a consequence, forest floor C/N ratios increased from 44.4 to 51.5 under ambient CO2 and from 46.7 to 48.9 under FACE. During the same period, the C/N ratio of the mineral soil (0–15 cm) did not change under ambient CO2 (18.2 → 18.7), but increased under FACE (17.2 → 20.5).
At Oak Ridge the FACE-induced soil C accrual was accompanied by a significant increase in soil N, that is, FACE did not affect the C/N ratio of the mineral soil. Jastrow and others (2005) postulated that FACE also affected N cycling by some combinations of reducing N losses, stimulation of N fixation, and increasing N uptake through greater root exploration.
During the first rotation of the POP-EuroFACE experiment, FACE treatment significantly decreased N concentrations in leaf litter, but did not significantly increase annual leaf litter production (Cotrufo and others 2005). As a result, FACE reduced the input of N into the forest floor and soil. In an assessment of the N use by the poplar trees during the first rotation, Calfapietra and others (2007) found that FACE decreased the N concentration of most plant tissues, whereas biomass increased under FACE. As a result, FACE did not affect plant N pools and did not change the cumulative uptake of N by the trees. The observed loss of soil N under FACE during the third year may have been caused by a combination of increased N mineralization due to a priming effect as mentioned earlier (Hoosbeek and others 2004) and lower N input through leaf litter (Calfapietra and others 2007; Cotrufo and others 2005).
This article focuses on the forest floor and soil C and N dynamics during the second rotation. We hypothesize that FACE, through increased NPP, increases forest floor litter build up, and we expect an increase in litter C/N ratio due to changing litter quality. With respect to the mineral soil we hypothesize that FACE increases soil C content. We had to reject this hypothesis for the first rotation, but will test this original hypothesis again for the second rotation and for both rotations combined. The extra available substrate under FACE may either sustain the increased N mineralization or induce increased N immobilization. Furthermore, with respect to the stabilization and protection of SOM, we hypothesize that the FACE-induced presence of extra labile soil C increased the formation of macro-aggregates and subsequently increased the formation of micro-aggregates and stable organo-mineral complexes.
Methods
Site Description
The POPFACE experiment was established early 1999 on former agricultural fields near Viterbo (42°37′04″N, 11°80′87″E, alt. 150 m), Italy. The plantation and adjacent fields had been under forest until about 1950. Since then a variety of agricultural crops have been grown on these former forest soils until the inception of the POPFACE plantation. The annual precipitation is on average 700 mm with dry summers (xeric moisture regime). During November 1998, an initial soil survey took place. The loamy soils classified as Pachic Xerumbrepts and were described in detail by Hoosbeek and others (2004).
Nine hectares were planted with Populus x euramericana hardwood cuttings at a density of 0.5 trees m−2. Within this plantation three FACE and three control plots (30 × 30 m2) were randomly assigned under the condition of a minimum distance between the plots of 120 m to avoid CO2 cross contamination. These six plots were planted at a density of 1 tree m−2 using three different genotypes. The plots were divided into two parts by a physical resin-glass barrier (1-m deep in the soil) for differential N treatments in the two halves of each plot. However, because of the high inorganic N content of the soil, no fertilization treatment was applied during the first 3-year rotation of the experiment. Each half plot was divided into three sectors, where each sector was planted with one of the following genotypes: P. x euramericana Dode (Guinier) (=P. deltoides Bart. ex Marsh. × P. nigra L.) genotype I-214, P. nigra L. (Jean Pourtet) and a local selection of P. alba L. (genotype 2AS11).
C enrichment was achieved by injecting pure CO2 through laser-drilled holes in tubing mounted on six masts (Miglietta and others 2001). The FACE rings (octagons) within the FACE plots had a diameter of about 22 m. The elevated CO2 concentrations, measured at 1-min intervals, were within 20% deviation from the pre-set target concentration (560 μmol mol−1) for 91% of the time to 72.2% of the time, respectively, at the beginning and at the end of each rotation cycle of the plantation. The used CO2 gas had a δ13C value of −6‰, which is close to the ambient value and therefore not suitable as a tracer. The plantation was drip irrigated at a rate of 6 to 10 mm d−1 during the growing seasons.
The trees were coppiced after the first three growing seasons (1999–2001). The experiment continued with a second rotation under the name EuroFACE (2002–2004). A fertilization treatment was added to one-half of each experimental plot because soil analyses showed the occurrence of limiting conditions of N availability in the soil (Scarascia-Mugnozza and others 2006). The total amount of N supplied was 212 kg ha−1 y−1 in 2002 and 290 kg ha−1 y−1 during 2003 and 2004.
Forest Floor Litter
Litter samples were collected in October 2004, that is, at the end of the 6-year experiment. A PVC ring with an inner diameter of 19 cm was placed on top of the forest floor to serve as a template for cutting the litter. A sharp knife was used to cut a cylindrical sample. Despite the fact that three distinct litter layers could be identified, it was not possible to sample these separately. Within the cylinder, all litter was removed from the mineral soil using a brush and spoon. The samples were transported to the laboratory in a mobile refrigerator. In the laboratory the samples were dried and fractionated by dry sieving (<2, 2–8, >8 mm). These size fractions largely resembled the L (almost undecomposed litter), F (recognizable, but fragmented), and H (humified) layers as observed in field. C and N were determined by flash combustion in an elemental analyzer (EA 1108) (Van Lagen 1996).
Mineral Soil
Throughout the experiment soil samples were collected at the end of each growing season from each sector within the three control and three FACE plots. Bulk density samples were taken at 0–10 and 10–20 cm below the surface of the mineral soil with the help of a bulk density sampler that holds 300 cm3 metal rings. Adjacent to these samples, bulk samples were taken with a small spade for C and N analyses and fractionation. After transportation in a mobile refrigerator, the ring samples were dried at 105°C for 3 days, whereas the bulk samples were dried at room temperature. Bulk densities were calculated based on dry weight of the ring samples and ring volume. For C and N analyses sub-samples of the bulk samples were crushed by hand and live roots were removed. No carbonates were present in the soil. C and N were determined with an elemental analyzer (EA 1108) (Van Lagen 1996). Total soil organic C and N content are expressed as gram C or N per m2 per depth increment.
Physical Fractionation
For fractionation, one bulk sample per sector was collected with a small spade from the upper 10 cm of the mineral soil and air dried at room temperature. Before drying, large aggregates (>1 cm) were broken up along natural planes of weakness. The wet sieving procedure was described by Kemper and Rosenau (1986) and Pulleman and others (2003). Materials used included a wet sieving apparatus, 20 l buckets (used as wet sieving basins) and three 20-cm diameter sieves (2000, 250, and 53 μm mesh). The buckets were filled with demineralized water; the sieves were stacked, submerging one sieve at a time to prevent air bubbles from getting trapped under a sieve. The top sieve (2000 μm) was placed on top of the stack without touching the water at first. Dried soil material was placed on the top sieve, after which the stack of sieves was lowered until the material on the top sieve was just covered by water. The samples were left to slake for 5 min, followed by 2 min of wet sieving. The wet sieving apparatus gently lowers and lifts the sieves at a speed of about 30 repetitions per minute, over a distance of 3 cm. After sieving, floating material was aspirated and the sieves were lifted out of the water. Material that remained on the sieves was washed into beakers assigned to the specific fractions. The isolated fractions were dried at 40°C. Four fractions based on the following size classes were distinguished: smaller than 53 (clay and silt sized), 53–250 (micro-aggregates), 250–2000 (macro-aggregates), and larger than 2000 μm (large macro-aggregates). C and N contents were determined as described above.
Intra Macro-Aggregate Fractions
Breaking up the macro-aggregate fraction will, according to the aggregate formation model, result in the release of clay and silt (<53 μm), micro-aggregates (53–250 μm), and coarse POM plus sand (>250 μm). A “micro-aggregate isolator,” as described by Six and others (2002), was used to break up the macro-aggregates while minimizing the break down of the released micro-aggregates. Ten grams of macro-aggregates were immersed in deionized water on top of a 250-μm mesh screen and shaken with 50 glass beads (4-mm diameter). A continuous water flow through the device flushed all released micro-aggregates immediately onto a 53-μm sieve, thus avoiding further disruption. After complete breakup of the macro-aggregates, coarse iPOM and sand remained on the 250 μm mesh screen. The micro-aggregates and the clay and silt fraction were separated by the 53-μm sieve.
Density Fractionation
The macro- and micro-aggregates obtained by wet sieving and the micro-aggregates isolated from macro-aggregates were further analyzed by density fractionation to obtain the intra-aggregate light (LF) and heavy (HF) fractions. Five grams of dried soil material was suspended in 35 ml of a 1.85 g cm−3 sodium polytungstate solution (SPT) in 50 ml conical falcon tubes (Six and others 2002). The tubes were gently shaken ten times end over end. Material that remained on the cap and sides of the tubes was rinsed back into solution with more SPT solution and the volume was made up to the 40 ml mark. The tubes were placed under vacuum (−80 kPa) for 10 min. After this, the samples were left to rest for 20 min, tubes were balanced with SPT, capped, and centrifuged for 60 min at 1250 g (in this case: 2600 rpm).
Floating material (LF) was aspirated onto a pre-weighed glass fiber filter, SPT solution was decanted over the filter. The glass fiber filters containing the LF were rinsed with demineralized water, dried, and weighed. The HF was rinsed twice by adding demineralized water, shaking until all materials were suspended again and centrifuged. After centrifugation the solution was decanted.
The tubes with the washed HF were made up to the 40 ml mark with 0.5% sodium hexametaphosphate solution (NaHMP, dispersing agent) and shaken in a reciprocal shaker for 18 h. The dispersed HF was sieved over 2000, 250 and 53 μm sieves, respectively, to isolate the different fractions of iPOM and sand. The different fractions were collected and dried at 70°C. The fraction smaller than 53 μm also contained NaHMP, which does not evaporate with the water and amounted to approximately 0.2 g. SPT solution was recycled by passing it through a column of active C to remove remaining organic molecules in the solution and filtering it through glass fiber filters to remove possible precipitates (Six and others 1999).
Statistical Model
The POP-EuroFACE field experiment was set up as split-split-plot design with six rings (one within each of six randomly selected areas of 30 × 30 m2) as whole-plots (three FACE and three control plots). Each whole-plot was split into two sub-plots that each received one of two N-fertilization treatments. The sub-plots were split into three sub-sub-plots with different poplar genotypes. The whole-plots were assigned randomly within the poplar plantation, and the CO2 treatments were assigned randomly to these whole-plots. Within each whole-plot, the two N-treatments were assigned randomly to the two sub-plots, and within each sub-plot the three genotypes were assigned randomly to the sub-sub-plots. The number of replicates per treatment are therefore: CO2 treatment n = 6 (three ambient + three FACE); N treatment n = 12 (six unfertilized + six N-fertilized); Species n = 36 (12 P. alba + 12 P. nigra + 12 P. euramericana).
Two versions of the same general linear model (SPSS 12.0.1) were used for the analysis of, respectively, (1) data obtained at one point in time, and (2) data obtained in consecutive years (repeated measures anova). Version 1 was built with the following factors: CO2trmt (fixed); Ntrmt (fixed); Species (fixed); RingNr(random). The model (specified in the design statement) contained the following elements: CO2trmt, RingNr(CO2trmt), Ntrmt, CO2trmt × Ntrmt, Ntrmt × RingNr(CO2trmt), Species, CO2trmt × Species; Ntrmt × Species; CO2trmt × Ntrmt × Species, where RingNr(CO2trmt) indicates the nested structure of the data. The specification of RingNr as a random factor and as a factor nested within CO2trmt is necessary to obtain the appropriate ANOVA table and F-tests for the various main treatment factors and their interactions. For Version 2 of the model year was added as a fixed factor to the elements (including its interactions). Main or interaction effects were considered to be significant when the P-value of the F-test was <0.05.
Results
Forest Floor Litter
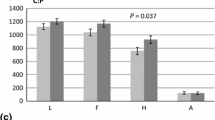
Between the initiation of the experiment, in November 1998 and November 2004, a forest floor with three distinguishable litter layers had built up (L, F, and H horizons). Under ambient CO2 the total accumulated C and N contents (L + F + H) were, respectively, 574 (se 48) g C m−2 and 38 (se 3) g N m−2 (Figures 1 and 2). The total C content under FACE was significantly (P = 0.022) larger, that is, 767 (se 52) g C m−2. The N content was larger as well, that is, 53 (se 4) g N m−2, but not significantly (P = 0.109). The C/N ratio of the total forest floor was not affected by FACE (Figure 3). The N-fertilization treatment and species did not affect total C and N contents, nor the C/N ratio.
Forest floor litter C content accumulated between November 1998 (bare soil) and November 2004. The error bars represent +SE and apply to the total (unfractionated) C content. Significant (P = 0.022) FACE effect on total litter C content. Significant (P = 0.005) FACE effect on L litter C content. Significant (P = 0.006) species effect on L litter C content. Possible (P = 0.063) FACE effect on H litter C content.
Forest floor litter N content accumulated between November 1998 (bare soil) and November 2004. The error bars represent +SE and apply to the total (unfractionated) N content. Possible (P = 0.109) FACE effect on total litter N content. Significant (P = 0.017) species effect on L litter N content. Possible (P = 0.101) FACE effect on H litter N content.
Forest floor litter C/N ratios per fraction. Error bars represent +SE per fraction. Possible (P = 0.098) species effect on L litter C/N ratio. Significant (P = 0.017) N-fertilization effect on F litter C/N ratio. Possible (P = 0.066)) N-fertilization effect on H litter C/N ratio. Significant (P = 0.011) species effect on H litter C/N ratio.
The C content of the to larger than 8 mm litter fraction, consisting primarily of unfragmented leaves and small branches, was larger under FACE (P = 0.005). The N content of this fraction was not affected by FACE. As a result, the C/N ratio was larger under FACE, although not significantly. N-fertilization did not affect the to larger than 8 mm litter fraction. Species affected both C and N contents of the to larger than 8 mm fraction (P = 0.006 and 0.017), with the highest C and N contents under P. euramericana and the lowest under P. nigra. The C/N ratio under P. alba was larger than for the other species (P = 0.098).
The 2–8 mm litter fraction, consisting primarily of fragmented and partly decomposed leaves and small branches, contains less C and N than the other litter fractions. Its C and N contents were not significantly affected by FACE, N-fertilization, or species. However, N-fertilization treatment decreased the C/N ratio (P = 0.017) of the 2–8 mm fraction.
The oldest litter fraction (<2 mm), representing decomposed and unrecognizable plant remains, is the largest forest floor fraction. Its C and N contents were enhanced by FACE although not significantly (P = 0.063 and 0.101). N-fertilization also increased C and N contents, but not significantly. There was no species effect on C and N contents. FACE did not affect the C/N ratio of the to smaller than 2 mm fraction. N-fertilization possibly decreased the C/N ratio (P = 0.066), while the lowest C/N ratio was observed under P. nigra (P = 0.011).
Mineral Soil Bulk Density
The soil bulk density changed during the experiment, but this change was not affected by species or either by CO2 or by N-fertilization treatments (Figure 4). After plowing and soil preparation during the fall of 1998, the soil settled during the following years (1999–2001). Subsequently, the bulk density decreased due to the addition of organic matter, biological activity, and aggregate formation.
Mineral Soil C and N Contents: First and Second Rotations
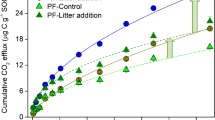
During the 6-year experiment soil C increased by, respectively, 301 and 308 g C m−2 under ambient CO2 and FACE at the 0–10-cm depth interval (Figure 5; Table 1). Soil N increased by, respectively, 28 and 18 g N m−2 under ambient CO2 and FACE at the same depth interval (Figure 6; Table 1). Soil C and N contents were not affected by CO2 and N treatments and species. Only the increase with time and the random factor Ring number were significant.
At the 10–20-cm depth interval, soil C increased by, respectively, 52 and 44 g C m−2 under ambient CO2 and FACE during the 6-year experiment (Figure 5; Table 1), whereas soil N increased and slightly decreased by, respectively, 6 and 0.4 g N m−2 (Figure 6; Table 1). Again, FACE had no significant effect. N fertilization suppressed the increase in soil C, but did not affect soil N content. The increase in soil C was significantly lower under P. euramericana. Also at the second depth interval, Ring number was significant for both soil C and N contents.
Mineral Soil C and N Contents: Second Rotation
During the second rotation (2001–2004), soil C increased under ambient CO2 and FACE by, respectively, 130 and 292 g C m−2 in the top 10 cm (Figure 5; Table 2). Although, the increase under FACE during this period was more than twice as much as under ambient CO2, the FACE effect was not significant. FACE did not affect soil N either (Figure 6; Table 2). Furthermore, soil C and N were not affected by N-fertilization and species, whereas the increase with time and the random factor Ring number were significant.
Also at the 10–20-cm depth interval the increase in soil C under FACE was twice as much as under ambient CO2, but again the effect was not significant (Figure 5; Table 2). Soil N was not affected by FACE either (Figure 6; Table 2). Species affected soil C content, whereas Ring number affected both soil C and N.
Physical Fractionation: Wet Sieving
C and N contents of the large macro-aggregates (>2000 μm) were larger under FACE than under ambient CO2, although not significantly (Table 3). N-fertilization and species did not affect the C and N contents of large macro-aggregates. C and N contents of the macro-aggregates (250–2000 μm) were significantly larger under FACE and N-fertilization. Also species affected C and N contents of the macro-aggregates with the highest and lowest values under, respectively, P. nigra and P. alba. The micro-aggregate fraction (53–250 μm) was not affected by CO2 and N treatment and species. C and N contents of the silt and clay sized fraction (<53 μm) were lower under FACE.
Intra-Macro-Aggregate Fractions
FACE increased C and N contents of the light iPOM and the mineral associated intra-macro-aggregate fractions (Table 4). The C and N contents of the coarse iPOM fractions were not affected by FACE, whereas the N content of the fine iPOM fraction decreased due to FACE. N-fertilization did not affect the C content of any of the intra-macro-aggregate fractions, but increased the N content of the coarse and fine iPOM fractions. The lowest C and N contents of the light and fine iPOM intra-macro-aggregate fractions were found under P. alba.
Intra-Micro-Aggregates Fractions of ‘Free’ Micro-Aggregates
C and N contents of the light iPOM intra-micro-aggregate fractions increased under FACE (Table 5). However, C content of the mineral-associated fraction decreased due to FACE. N-fertilization did not affect C and N contents of any of the intra-micro-aggregate fractions. Poplar genotype affected C and N contents of the light iPOM and C content of the fine iPOM fractions, where the largest fractions were found under P. euramericana.
Intra-Micro-Aggregates Fractions of Micro-Aggregates Isolated from Macro-Aggregates
FACE increased C and N contents of the light and mineral-associated iPOM fractions and C content of the fine iPOM fraction (Table 6). N-fertilization increased C content of the mineral-associated iPOM fraction. C and N contents of the light iPOM fraction were largest under P. euramericana, whereas C and N contents of the mineral associated iPOM fraction were lower under P. alba.
Discussion
Forest Floor Litter
Cotrufo and others (2005) observed a decrease in leaf litter N input under FACE during the first rotation. This decreased N input did not result in a lower C/N ratio under FACE of the total forest floor after 6 years. However, the C/N ratio of the most recent litter fraction (>8 mm) was larger under FACE, although not significantly. The C/N ratios of the older fractions were lower as compared with younger fractions and were not affected by FACE, probably due to N immobilization during decomposition as was observed by Cotrufo and others (2005).
The larger accumulated forest floor C content under FACE is also in agreement with the results of a decomposition experiment by Cotrufo and others (2005). They placed leaf litter produced under ambient CO2 in bags in ambient and FACE plots, and litter produced under FACE in ambient and FACE plots. They postulated that under FACE litter decomposed faster during the first phase of decomposition probably due to a FACE-induced priming effect. However, during the later stages of decomposition the remaining mass was higher under FACE due to lower leaf litter N input under FACE.
FACE created an additional sink of 193 g C m−2 in the forest floor over a period of 6 years, which is on average 32 g C m−2 y−1 (Table 7). This is less than the FACE-induced additional C sink of 52 g C m−2 in the forest floor of the Duke Forest. Despite larger NPP rates at POP-EuroFACE (Calfapietra and others 2003; Liberloo and others 2006a; Norby and others 2002), the smaller additional sink may be due to the difference in litter type, that is, deciduous versus coniferous, and the type of soil. Although measured and calculated with different methods, decomposition rates of poplar litter may be up to one order of magnitude larger than for loblolly pine litter (Cotrufo and others 2005; Lichter and others 2005). These high rates of decomposition for poplar litter will primarily apply to the youngest fractions, that is, the larger than 8 mm L and 2–8 mm F fractions. Once humified in the smaller than 2 mm H fraction, decomposition rates decline. The long-term sink potential of the forest floor litter layer will therefore largely be determined by this humified forest floor litter pool. Of the three distinguished fractions, the humified fraction was the largest for both C and N contents. These C and N contents were enhanced by FACE (P = 0.063 and 0.101) without affecting its C/N ratio. We infer that the forest floor as a whole, and in particular the humified fraction, will probably serve as an additional C sink under future increased atmospheric CO2 concentrations in poplar stands. Although, the mean residence time of organic matter in the forest floor is in general less than in the mineral soil (for example, Lichter and others 2005), due to the absence of stabilization mechanisms that may occur in mineral soils like aggregate formation and bonding of organic matter to mineral surfaces.
Field observations revealed the presence of many arthropods, whereas earthworms were virtually absent. Also, the boundary between the H and the A horizons was clear and showed negligible signs of mixing, that is, mixing of humified H material into the top of the mineral soil by bioturbation. We therefore infer that since the beginning of the experiment almost all aboveground litter accumulated in the forest floor, whereas the input of organic matter into the mineral soil is primarily due to root turnover. In addition, some dissolved organic matter may have leached from the forest floor litter layers into the mineral soil, but we estimated this quantity to be relatively small as compared with the other C fluxes involved.
Mineral Soil
Total soil organic matter content has been increasing in all plots, that is, under all treatments and poplar species, since the beginning of the experiment (Figure 5). This increase in SOM is largely due to the afforestation of agricultural land. During the first rotation (1999–2001), total soil C content increased more under ambient CO2 treatment than under FACE, whereas under FACE more new C was incorporated than under ambient CO2 (Hoosbeek and others 2004). These unexpected and opposite effects may have been caused by a priming effect. The extra available C under FACE probably increased the decomposition of old and new soil C (Hoosbeek and others 2006a, 2007).
However, during the second rotation (2002–2004) the increase in soil C was larger under FACE. Soil C increased under ambient CO2 and FACE by, respectively, 130 and 292 g C m−2 in the top 10 cm, which is an additional sink of 162 g C m−2 over 3 years, or on average 54 g C m−2 (0–10 cm) per year (Table 7). Although, the increase in soil C under FACE was more than twice as much as under ambient CO2, the FACE effect was not significant due to a combination of spatial variability and a low number of replicates.
FACE induced contrasting effects on soil C content during the first and second rotations. The loss of older C during the first rotation and the increased accumulation of new C during the second rotation under FACE resulted in about equal increases of soil C under ambient CO2 and FACE during the 6-year experiment.
Soil N content followed a similar trend as soil C for the first 5 years. Irrespective of treatment and species, the C/N ratio increased linearly from 9.3 in 1999 to 10.7 in 2003, but decreased in 2004 to 10.0. Lagomarsino and others (2008) monitored the microbial biomass during the second rotation and found that microbial biomass N (MBN) had by far the highest values in October 2004. When expressed as a fraction of total N (TN), microbial biomass N (MBN/TN) was significantly increased under FACE throughout the second rotation. Lagomarsino and others (2008) observed the preferential uptake of ammonium-N by microbial biomass and concluded that FACE enhanced the immobilization of ammonium-N into organic N. From these and earlier observations we infer that during the first rotation additional labile C under FACE induced a priming effect in the soil, that is, increased N mineralization, whereas during the second rotation additional labile C caused increased N immobilization under FACE.
At the Duke Forest, soil C content of the top 15 cm increased under ambient and elevated CO2 by, respectively, 430 and 592 g C m−2 over a period of 6 years. This amounts to an additional sink of 27 g C m−2 (0–15 cm) per year, but this FACE effect was not significant. At Oak Ridge, Jastrow and others (2005) found a significant FACE-induced additional sink of 220 g C m−2 in the top 5 cm of the mineral soil over a period of 5 years, which is on average 44 g C m−2 (0–5 cm) per year. However, for the 0–15 depth increment this sink was found to be 28 g C m−2 (0–15 cm) per year (based on the data from Figure 1 in Jastrow and others 2005).
These FACE-induced additional soil C sink rates cannot be related directly, because they apply to different depth intervals. Because the C content of these soils is far from constant with depth, a simple linear correction for depth is not feasible. For instance, Jastrow and others (2005) observed a FACE effect on soil C for the 0–5-cm depth increment, but not for the 0–15-cm depth increment. Still, a qualitative comparison of the vertical distribution of the FACE-induced additional C sinks in the forest floor—soil systems of the three sites is possible (Table 7).
C Sinks in the Forest Floor–Soil System and the Role of Bioturbation
The largest additional forest floor C sink is observed in the relative low NPP coniferous Duke Forest, which is underlain by an acidic, low base saturation (BS), soil that does not exhibit mixing of forest floor material into the mineral soil (bioturbation) (Table 7). The second largest additional forest floor C sink is found in the high NPP poplar system, which is underlain by an acidic, low BS, soil that does not exhibit bioturbation either. At Oak Ridge, the intermediate NPP deciduous site which is underlain by a slightly acidic soil with high base status, most aboveground litter is incorporated into the mineral soil by bioturbation. As a result, the litter input at Oak Ridge, both above- and belowground, is largely concentrated into the top centimeters of the mineral soil.
Jastrow and others (2005) raised the question of whether the lack of a FACE effect on soil C content in some other studies is a general response or a function of (1) the low statistical power of most forest FACE experiments, and/or (2) the magnitude of CO2-stimulated C inputs relative to the duration of the experiments. Suggestion (1) certainly applies to Pop-EuroFACE. Despite the fact that the increase in soil C under FACE was more than twice as much as under ambient CO2 between 2001 and 2004, the FACE effect was not significant probably in part due to the combination low statistical power and soil spatial variability. Suggestion (2) applies as well, that is, during the first half of the experiment, the FACE effect on soil C was opposite from what we had hypothesized due to an unexpected priming effect. Only during the second half of the 6-year experiment, FACE increased soil C which probably would have become a significant effect after several more years of FACE treatment. Jastrow and others (2005) also argued that the sampling increment used among several experiments may have affected the statistical outcome. For instance at Oak Ridge, a significant FACE effect on soil C was observed for the top 5 cm, whereas sampling a thicker soil increment, for example, 0–15 cm, would have “diluted” the increase in C which would have resulted in a non-significant effect.
The detection of a significant FACE effect may also depend on the vertical distribution of the FACE-induced C sink. In a forest floor–soil system without bioturbation the sink is distributed over two compartments, that is, the forest floor and the mineral soil (Duke Forest, POP-EuroFACE). In an ecosystem with bioturbation, the FACE-induced C sink is concentrated in the top of the mineral soil (Oak Ridge), which makes it more likely that a FACE effect on soil C is detected.
Physical Protection and Stabilization of Soil C: Macro-Aggregates
We observed an increase in the C and N contents of macro-aggregates due to FACE (Table 3), which means that the formation of macro-aggregates increased under FACE. The light iPOM fraction within these macro-aggregates also increased under FACE (Table 4). This fraction consists primarily of fresh plant remains incorporated in macro-aggregates. The presence of this extra labile C in the soil was also confirmed by chemical fractionation and by respiration measurements (Hoosbeek and others 2006a, 2007). According to the model of aggregate formation, these fresh plant remains become sites for microbial activity and nucleation centers for further aggregation. Lagomarsino and others (2008) observed an increase in the microbial biomass N (MBN/TN) under FACE, which in combination with an increase in the light iPOM C fraction inside macro-aggregates, is likely to explain the increased formation of the mineral-associated iPOM fraction under FACE. The association of iPOM with mineral surfaces increased under FACE. This mineral-associated fraction is thought to be less vulnerable to decomposition by extracellular enzymes.
The increase in soil C at the Duke Forest FACE experiment occurred entirely within the free LF, whereas the iPOM and mineral-associated fractions were not affected by FACE. Lichter and others (2005) estimated the FACE-induced C sink into protected SOM pools to be about 3 g C m−2 y−1. Because this amount is less than the uncertainty associated with obtaining the soil fractions, Lichter and others (2005) concluded that it was no surprise that physical fractionation failed to detect a treatment effect.
Physical Protection and Stabilization of Soil C: Micro-Aggregates
Eventually, the binding agents in macro-aggregates degrade, resulting in a breakdown of macro-aggregates and the release of microbially processed organic matter and micro-aggregates. The C and N contents of the light iPOM fraction of these “free” micro-aggregates increased due to FACE, which means that still more relatively fresh plant remains were present in these aggregates. However, C content of the stabilized and regarded older mineral-associated iPOM fraction was lower under FACE. This effect may have been caused by the priming effect during the first rotation by which more older organic matter was lost under FACE. P. euramericana increased C and N contents of the light iPOM and C content of the fine iPOM intra-micro-aggregate fractions, which is in agreement with earlier observations by (Hoosbeek and others 2006b).
Physical Protection and Stabilization of Soil C: Micro-Aggregates Isolated from Macro-Aggregates
Breaking up the macro-aggregates resulted in the release of micro-aggregates that were still present in the “nursery.” Although most micro-aggregates present in macro-aggregates are assumed to have been formed there, some of the micro-aggregates may have been included during formation of macro-aggregates. These micro-aggregates are then actually older than the macro-aggregates they are released from.
FACE increased C and N contents of the light iPOM fractions of micro-aggregates from within macro-aggregates, which means that more organic matter is available for further stabilization into the heavy iPOM and mineral-associated intra-micro-aggregate fractions. This in combination with the increased microbial biomass explains that under FACE the C content of the fine iPOM and C and N contents of the mineral-associated iPOM fractions increased, which proves that the amount of stabilized C and N increased under FACE treatment.
At Oak Ridge, the proportion of soil C found in ‘free’ micro-aggregates and micro-aggregates isolated from macro-aggregates averaged 58% in both FACE and ambient plots and did not change over time. Jastrow and others (2005) concluded that the extra FACE-induced C input into the soil was protected in the same manner as under ambient conditions.
Physical Protection and Stabilization of Soil C Compared Among Forest FACE Experiments
Taking the results of physical fractionation of the forest FACE experiments together, the following picture emerges: At Duke, only the forest regrowth effect was large enough to have an effect on the LF. FACE-induced C flows into the LF and subsequent protected and stabilized fractions were below the detection limit of the fractionation techniques. At Oak Ridge, the protection and stabilization processes in the soil kept up with the extra C input due to FACE, that is, the extra C input due to FACE is protected in the same manor as under ambient CO2. Whereas at POP-EuroFACE iPOM and mineral-associated C and N fractions increased in macro-aggregates and in newly formed micro-aggregates, which indicates that protection and stabilization processes increased due to FACE.
Conclusions
After 6 years of FACE treatment in a short-rotation poplar plantation, we observed an additional C sink of 32 g C m−2 y−1 in the forest floor litter layer. Despite decreased leaf litter N input under FACE, the C/N ratio of the total forest floor was not affected by FACE due to increased N immobilization under FACE. We infer that the forest floor as a whole, and in particular the humified fraction, will serve as an additional C sink under future increased atmospheric CO2 concentrations in poplar stands.
During the 6-year experiment, soil C content increased equally under ambient and increased CO2 treatment. However, during the first half of the experiment the increase in soil C was suppressed under FACE due to a priming effect, whereas during the second half of the experiment the increase in soil C was larger under FACE. An additional sink of 54 g C m−2 y−1 in the top 10 cm of the mineral soil was created under FACE during the second half of the experiment. However, this FACE effect was not significant due to a combination of soil spatial variability and the low number of replicates that are inherent to the present generation of forest stand FACE experiments. The soil C/N ratio increased during the first 5 years, but decreased during the sixth year due to the increased immobilization of ammonium-N into organic N under FACE.
In a qualitative comparison among three forest FACE experiments, we found that the type of vegetation and soil BS are important factors related to the size of the additional C sinks of the forest floor—soil system under FACE, where, soil BS is an indicator for the presence of soil bioturbation (BT). At the Duke Forest experiment (coniferous, low BS, no BT), the largest C sink occurs in the forest floor, whereas the relatively small, not significant, C sink in the mineral soil occurs in the unprotected free LF. At the Oak Ridge experiment (deciduous, high BS, with BT), the aboveground litter is incorporated into the mineral soil (A horizon) where, in combination with root turnover, a significant C sink occurs under FACE in the top 5 cm. This additional C is protected and stabilized in the same manor as under ambient C. Results of the POP-EuroFACE experiment (deciduous, low BS, negligible BT) show a modest additional C sink in the forest floor and a large, but not significant, C sink in the top 10 cm of the mineral soil under FACE between 2001 and 2004. Despite the fact that the increase in soil C under FACE during this period was not significant, physical fractionation revealed that the amount of stabilized SOM increased due to FACE.
References
Baldock JA, Skjemstad JO. 2000. Role of the soil matrix and minerals in protecting natural organic materials against biological attack. Organic Geochemistry 31: 697–710.
Calfapietra C, De Angelis P, Gielen B, Lukac M, Moscatelli MC, Avino G, Lagomarsino A, Polle A, Ceulemans R, Scarascia-Mugnozza G, Hoosbeek MR, Cotrufo MF. 2007. Increased nitrogen-use efficiency of a short-rotation polar plantation in elevated CO2 concentration. Tree Physiology 27: 1153–1163.
Calfapietra C, Gielen B, Galema ANJ, Lukac M, De Angelis P, Moscatelli MC, Ceulemans R, Scarascia-Mugnozza G. 2003. Free-air CO2 enrichment (FACE) enhances biomass production in a short-rotation poplar plantation. Tree Physiology 23: 805–814.
Chenu C, Stotzky G. 2002. Interactions between microorganisms and soil particles: An overview. In Interactions between microorganisms and soil particles. Eds. PM Huang, J-M Bollag and N Senesi. New York: John Wiley & Sons, Ltd.
Cotrufo MF, De Angelis P, Polle A. 2005. Leaf litter production and decomposition in a poplar short-rotation coppice exposed to free air CO2 enrichment (POPFACE). Global Change Biology 11: 971–982.
Davidson EA, Janssens IA. 2006. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature 440: 165–173.
DeLucia EH, Hamilton JG, Naidu SL, Thomas RB, Andrews JA, Finzi AC, Lavine M, Matamala R, Mohan JE, Hendrey GR, Schlesinger WH. 1999. Net primary production of a forest ecosystem with experimental CO2 enrichment. Nature 284: 1177–1179.
Elliott ET. 1986. Aggregate structure and carbon, nitrogen, and phosphorous in native and cultivated soils. Soil Sci. Soc. Am. J. 50: 627–633.
Finzi AC, Norby RJ, Calfapietra C, Gallet - Budynek A, Gielen B, Holmes WE, Hoosbeek MR, Iversen CM, Jackson RB, Kubiske ME, Ledford J, Liberloo M, Oren R, Polle A, Pritchard S, Zak DR, Schlesinger WH, Ceulemans R. 2007. Increases in nitrogen uptake rather than nitrogen-use efficiency support higher rates of temperate forest productivity under elevated CO2. Proc. Nat. Acad. Sci. USA 104: 14014–14019.
Gielen B, Calfapietra C, Lukac M, Wittig VE, De Angelis P, Janssens IA, Moscatelli MC, Grego S, Cotrufo MF, Godbold DL, Hoosbeek MR, Long SP, Miglietta F, Polle A, Bernacchi CJ, Davey PA, Ceulemans R, Scarascia-Mugnozza GE. 2005. Net carbon storage in a poplar plantation (POPFACE) after three years of free-air CO2 enrichment. Tree Physiology 25: 1399–1408.
Hamilton JG, DeLucia EH, George K, Naidu SL, Finzi AC, Schlesinger WH. 2002. Forest carbon balance under elevated CO2. Oecologia 131: 250–260.
Handa IT, Körner C, Hättenschwiler S. 2005. A test of the treeline carbon limitation hypothesis by in situ CO2 enrichment and defoliation. Ecology 86: 1288–1300.
Handa IT, Körner C, Hättenschwiler S. 2006. Conifer stem growth at the altitudinal treeline in response to four years of CO2 enrichment. Global Change Biology 12: 2417–2430.
Hättenschwiler S, Handa IT, Egli L, Asshoff R, Ammann W, Körner C. 2002. Atmospheric CO2 enrichment of alpine treeline conifers. New Phytologist 156: 363–375.
Hoosbeek MR, Li Y, Scarascia-Mugnozza G. 2006a. Free atmospheric CO2 enrichment (FACE) increased labile and total carbon in the mineral soil of a short rotation Poplar plantation. Plant and Soil 281: 247–254.
Hoosbeek MR, Lukac M, Van Dam D, Godbold DL, Velthorst EJ, Biondi FA, Peressotti A, Cotrufo MF, De Angelis P, Scarascia-Mugnozza G. 2004. More new carbon in the mineral soil of a poplar plantation under Free Air Carbon Enrichment (POPFACE): Cause of increased priming effect? Global Biogeochemical cycles 18: GB1040, doi:1010.1029/2003GB002127.
Hoosbeek MR, Vos JM, Bakker EJ, Scarascia-Mugnozza G. 2006b. Effects of free atmospheric CO2 enrichment (FACE), N fertilization and poplar genotype on the physical protection of carbon in the mineral soil of a polar plantation after five years. Biogeosciences 3: 479–487.
Hoosbeek MR, Vos JM, Meinders MBJ, Velthorst EJ, Scarascia-Mugnozza G. 2007. Free atmospheric CO2 enrichment (FACE) increased respiration and humification in the mineral soil of a Poplar plantation. Geoderma 138: 204–212.
Houghton RA. 2003. The contemporary carbon cycle. In Biogeochemistry. Ed. WH Schlesinger: Elsevier. pp 473–513.
Houghton RA, Davidson EA, Woodwell GM. 1998. Missing sinks, feedbacks, and understanding the role of terrestrial ecosystems in the global carbon balance. Global Biogeochemical cycles 12: 25–34.
Janssens IA, Freibauer A, Ciais P, Smith P, Nabuurs GJ, Folberth G, Schlamadinger B, Hutjes RWA, Ceulemans R, Schulze E-D, Valentini R, Dolman AJ. 2003. Europe’s terrestrial biosphere absorbs 7 to 12% of Europen anthropogenic CO2 emissions. Science 300: 1538–1542.
Jastrow JD. 1996. Soil aggregate formation and the accrual of particulate and mineral-associated organic matter. Soil Biology and Biochemistry 28: 665–676.
Jastrow JD, Miller RM, Matamala R, Norby RJ, Boutton TW, Rice CW, Owensby CE. 2005. Elevated atmospheric carbon dioxide increases soil carbon. Global Change Biology 11: 2057–2064.
Karnosky DF, Zak DR, Pregitzer KS, Awmack CS, Bockheim JG, Dickson RE, Hendrey GR, Host GE, King JS, Kopper BJ, Kruger EL, Kubiske ME, Lindroth RL, Mattson WJ, McDonald EP, Noormets A, Oksanen E, Parsons WFJ, Percy KE, Podila GK, Riemenschneider DE, Sharma P, Thakur R, Sôber A, Sôber J, Jones WS, Anttonen S, Vapaavuori E, Mankovska B, Heilman W, Isebrands JG. 2003. Troposheric O3 moderates responses of temperate hardwood forests to elevated CO2: a synthesis of molecular to ecosystem results from the Aspen FACE project. Functional Ecology 17: 289–304.
Kemper WD, Rosenau RC. 1986. Aggregate stability and size distribution. In Methods of soil analysis. Part I. Physical and mineralogical methods. Ed. A Klute. Madison, WI American Society of Agronomy.
King JS, Pregitzer KS, Zak DR, Sôber J, Isebrands JG, Dickson RE, Hendrey GR, Karnosky DF. 2001. Fine root biomass and fluxes of soil carbon in young stands of paper birch and trembling aspen as affected by elevated atmospheric CO2 and tropospheric O3. Oecologia 128: 237–250.
Körner C, Asshoff R, Bignucolo O, Hättenschwiler S, Keel SG, Peláez-Riedl S, Pepin S, Siegwolf RTW, Zotz G. 2005. Carbon flux and growth in mature decideous forest trees exposed to elevated CO2. Science 309: 1360–1362.
Krull ES, Baldock JA, Skjemstad JO. 2003. Importance of mechanisms and processes of the stabilisation of soil organic matter for modelling carbon turnover. Functional Plant Biology 30: 207–222.
Lagomarsino A, Moscatelli MC, Hoosbeek MR, De Angelis P, Grego S. 2008. Assessment of soil nitrogen and phosphorous availability under elevated CO2 and N-fertilization in a short rotation poplar plantation. Plant and Soil 308: 131–147.
Liberloo M, Calfapietra C, Lukac M, Godbold D, Luo ZB, Polle A, Hoosbeek MR, Kull O, Marek M, Raines C, Taylor G, Scarascia-Mugnozza G, Ceulemans R. 2006a. Woody biomass production during the second rotation of a bio-energy Populus plantation increases in a future high CO2 world. Global Change Biology 12: 1094–1106.
Liberloo M, Tulva I, Raïm O, Kull O, Ceulemans R. 2006b. Photosynthetic stimulation under long-term CO2 enrichment and fertilization is sustained across a closed Populus canopy profile (EuroFACE). New Phytology 173: 537–549.
Lichter J, Barron SH, Bevacqua CE, Finzi AC, Irving KF, Stemmler EA, Schlesinger WH. 2005. Soil carbon sequestration and turnover in a pine forest after six years of atmospheric CO2 enrichment. Ecology 86: 1835–1847.
Loya WM, Pregitzer KS, Karberg NJ, King JS, Giardina CP. 2003. Reduction of soil carbon formation by tropospheric ozone under increased carbon dioxide levels. Nature 425: 705–707.
Lukac M, Calfapietra C, Godbold DL. 2003. Production, turnover and mycorrhizal colonization of root systems of three Populus species grown under elevated CO2 (POPFACE). Global Change Biology 9: 838–848.
Miglietta F, Peressotti A, Vaccari FP, Zaldei A, De Angelis P, Scarascia-Mugnozza G. 2001. Free-air CO2 enrichment (FACE) of a poplar plantation: the POPFACE fumigation system. New Phytologist 150: 465–476.
Norby RJ, DeLucia EH, Gielen B, Calfapietra C, Giardina CP, King JS, Ledford J, McCarthy HR, Moore DJP, Ceulemans R, De Angelis P, Finzi AC, Karnosky DF, Kubiske ME, Lukac M, Pregitzer KS, Scarascia-Mugnozza G, Schlesinger WH, Oren R. 2005. Forest response to elevated CO2 is conserved across a broad range of productivity. Proc. Nat. Acad. Sci. USA 102: 18052–18056.
Norby RJ, Hanson PJ, O’Neill EG, Tschaplinski TJ, Weltzin JF, Hansen RA, Cheng W, Wullschleger SD, Gunderson CA, Edwards NT, Johnson DW. 2002. Net primary productivity of a CO2-enriched deciduous forest and the implications for carbon storage. Ecological Applications 12: 1261–1266.
Norby RJ, Wullschleger SD, Hanson PJ, Gunderson CA, Tschaplinski TJ, Jastrow JD. 2006. CO2 Enrichment of a deciduous forest: The Oak Ridge FACE Experiment. In Managed Ecosystems and CO2. Eds. J Nösberger, SP Long, RJ Norby, M Stitt, GR Hendrey and H Blum. Berlin Heidelberg: Springer-Verlag.
Oades JM. 1984. Soil organic matter and structural stability: mechanisms and implications for management. Plant and Soil 76: 319–337.
Oades JM. 1993. The role of biology in the formation, stabilization and degradation of soil structure. Geoderma 56: 377–400.
Prior SA, Runion GB, Torbert HA, Rogers HH. 2004. Elevated atmospheric CO2 in agroecosystems: Soil physical properties. Soil Science 169: 434–439.
Puget P, Chenu C, Balesdent J. 1995. Total and young organic matter distributions in aggregates of silty cultivated soils. European Journal of Soil Science 46: 449–459.
Pulleman MM, Jongmans AG, Marinissen JCY, Bouma J. 2003. Effects of organic versus conventional arable farming on soil structure and organic matter dynamics in a marine loam in the Netherlands. Soil Use and Management 19: 157 - 165.
Rillig MC, Wright SF, Allen MF, Field CB. 1999. Rise in carbon dioxide changes soil strcuture. Nature 400: 628.
Scarascia-Mugnozza GE, Calfapietra C, Ceulemans R, Gielen B, Cotrufo MF, De Angelis P, Godbold DL, Hoosbeek MR, Kull O, Lukac M, Marek M, Miglietta F, A. P, Raines C, Sabatti M, Anselmi N, Taylor G. 2006. Responses to elevated [CO2] of a short rotation, multispecies poplar plantation: the POPFACE/EUROFACE experiment. In Managed Ecosystems and CO2. Eds. J Nösberger, SP Long, RJ Norby, M Stitt, GR Hendrey and H Blum. Berlin, Heidelberg, New York: Springer Verlag. pp 173–195.
Schimel DS. 1995. Terrestrial ecosystems and the carbon cycle. Global Change Biology 1: 77–91.
Schlesinger WH, Bernhardt ES, DeLucia EH, Ellsworth DS, Finzi AC, Hendrey GR, Hofmockel KS, Lichter J, Matamala R, Moore D, Oren R, Pippen JS, Thomas RB. 2006. The Duke Forest FACE experiment: CO2 enrichment of a loblolly pine forest. In Managed Ecosystems and CO2. Eds. J Nösberger, SP Long, RJ Norby, M Stitt, GR Hendrey and H Blum. Berlin, Heidelberg: Springer-Verlag.
Schlesinger WH, Lichter J. 2001. Limited carbon storage in soil and litter of experimental forest plots under increased atmospheric CO2. Nature 411: 466–469.
Six J, Callewaert P, Lenders S, De Gryze S, Morris SJ, Gregorich EG, Paul EA, Paustian K. 2002. Measuring and understanding carbon storage in afforested soils by physical fractionation. Soil Sci. Soc. A. J. 66: 1981–1987.
Six J, Carpentier A, Van Kessel C, Merckx R, Harris D, Horwath WR, Lüscher A. 2001. Impact of elevated CO2 on soil organic matter dynamics as related to changes in aggregate turnover and residue quality. Plant and Soil 234: 27–36.
Six J, Elliott ET, Paustian K. 1999. Aggregate and soil organic matter dynamics under conventional and no-tillage systems. Soil Sci. Soc. A. J. 63: 1350–1358.
Six J, Elliott ET, Paustian K, Doran JW. 1998. Aggregation and soil organic matter accumulation in cultivated and native grassland soils. Soil Sci. Soc. Am. J. 62: 1367–1377.
Van Groenigen KJ, Harris D, Horwath WR, Hartwig UA, Van Kessel C. 2002. Linking sequestration of 13C and 15N in aggregates in a pasture soil following 8 years of elevated atmospheric CO2. Global Change Biology 11: 1094–1108.
Van Groenigen KJ, Six J, Hungate BA, De Graaff MA, Van Breemen N, Van Kessel C. 2006. Element interactions limit soil carbon storage. Proc Natl Acad Sci U S A 103:6571–4.
Van Lagen B. 1996. Soil Analyses. In Manual for soil and water analyses. Eds. P Buurman, B Van Lagen, EJ Velthorst. Leiden, The Netherlands: Backhuys Publishers.
Van Veen JA, Kuikman PJ. 1990. Soil structural aspects of decomposition of organic matter. Biogeochemistry 11: 213–233.
Acknowledgements
The authors thank Mr. Eef Velthorst and Mrs. Judith Vos for analyzing soil samples and for performing physical fractionations. Dr. Paolo De Angelis, Dr. Carlo Calfapietra and colleagues are gratefully acknowledged for managing and maintaining the FACE facility. Two anonymous reviewers are acknowledged for providing valuable and constructive comments. Funding was provided by the European Commission Fifth Framework Program, Environment and Climate RTD Program, research contract EVR1-CT-2002-40027 (EUROFACE) and by the Centre of Excellence “Forest and Climate” of the Italian Ministry of University and Research (MIUR).
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author Contribution: MRH conceived of and designed the study, performed research, analyzed data, and wrote the paper; GES conceived of and designed the study and performed research.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Hoosbeek, M.R., Scarascia-Mugnozza, G.E. Increased Litter Build Up and Soil Organic Matter Stabilization in a Poplar Plantation After 6 Years of Atmospheric CO2 Enrichment (FACE): Final Results of POP-EuroFACE Compared to Other Forest FACE Experiments. Ecosystems 12, 220–239 (2009). https://doi.org/10.1007/s10021-008-9219-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10021-008-9219-z