Abstract
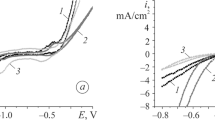
The dissolution processes of amorphous G15 (Ni58Co20B12Si10) and G16 (Ni25Co50B10Si15) alloys in carbonate-bicarbonate buffers (8.9≤pH≤10.5) have been studied using voltammetry and stationary polarization techniques combined with electrochemical impedance spectroscopy and X-ray photoelectron spectroscopy (XPS) measurements. Results indicate that the electrochemical processes are dependent on the applied potential, the alloy composition, and the pH and ionic strength of the electrolyte. An enhancement of the corrosion processes is observed when the pH and the ionic strength are increased and when the Co content of the alloy diminishes. Furthermore, XPS provided information about the composition of the surface layer.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Barbosa, M., Gassa, L. & Ruiz, E. Electrochemical behaviour of Ni-Co amorphous alloys in alkaline solutions. J Solid State Electrochem 6, 1–7 (2001). https://doi.org/10.1007/s100080000189
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s100080000189