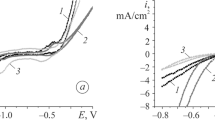
We study the electrochemical behavior of Fe80Si6B14, Co72Si18B10, and Fe84Nb2B14 amorphous metal alloys based on Fe and Сo in alkali solutions of different concentrations. The protective oxide layers on Fe80Si6B14 electrodes have lower degrees of corrosion protection than for the Co72Si18B10 electrodes. Niobium clusters in the Fe84Nb2B14 alloy are reactive, which results in the rapid initial interaction of the electrode with a 1 М aqueous solution of KОН, which is retarded under the conditions of long-term contact with ions of the electrolyte. The Fe80Si6B14 and Fe84Nb2B14 alloys are characterized by a similar electrochemical behavior in 1 M and 5 М aqueous solution of KOH under the conditions of cyclic polarization of samples in corrosive media. As a result of fivefold scanning of the potential, it is shown that the electrochemical properties of the electrodes made of amorphous metallic alloys depend on the electrolyte concentration.



Similar content being viewed by others
References
M. Zhu, S. Chen, L. Yao, Y. Li, Y. Wang, Z. Jian, and F. Chang, “The influence of Ni or Co substitution for Fe on glass forming ability and magnetic properties in the quaternary Fe−Nb−B−Ni and (Fe, Ni, Co)−Nb−B alloy systems,” J. Mater. Res., 30, No. 6, 811−817 (2015).
G. Vlasak, P. Svec, M. Kuzminski, A. Slawska-Waniewska, B. Butvinova, P. Butvin, and J. Hosko, “Evolution of physical properties of amorphous Fe−Ni−Nb−B alloys with different Ni/Fe ratio upon thermal treatment,” J. Alloy Comp., 509, No. 1, S64−S68 (2011).
J. C. Farmer, J. J. Haslam, S. D. Day, T. Lian, and R. Rebak, “Corrosion resistance of iron-based amorphous metal coatings,” in: Proc. of the ASME Pressure Vessels and Piping Division Conf. PVP2006-ICPVT-11 (Vancouver, July 23–27, 2006), ASME, Vancouver (2006), pp. 685−691.
O. M. Hertsyk, M. O. Kovbuz, and L. M. Bednarska, “Amorphization and elemental composition of metal alloys as anticorrosion factors,” Fiz.-Khim. Mekh. Mater., Special Issue, No. 2, 172−174 (2001).
L. M. Boichyshyn and O. M. Hertsyk, Amorphous Metal Alloys: Structure, Properties, and Methods of Investigations: A Textbook [in Ukrainian], I. Franko Lviv National University, Lviv (2016).
M.-O. Danylyak and L. M. Boichyshyn, “Specific features of the geometry of the surface of amorphous metallic alloys,” Pratsi NTSh. Khim. Nauky, LIII, 132−144 (2018).
M. O. Kovbuz, O. M. Hertsyk, T. H. Pereverzeva, S. E. Shurko, N. L. Pandyak, and Kh. V. Protsyshyn, “Influence of the compositions of corrosive media on the corrosion resistance of multicomponent amorphous alloys based on Fe,” Khim., Fiz. Tekhnol. Poverkh., 9, No. 1, 64−73 (2018).
L. M. Boichyshyn, O. M. Hertsyk, M. O. Kovbuz, Morphology, Structure, and Properties of Amorphous Alloys Doped with REM [in Ukrainian], I. Franko Lviv National University, Lviv (2019).
L. M. Boichyshyn, O. M. Hertsyk, M. O. Kovbuz, B. Ya. Kotur, and V. K. Nosenko, “Corrosion-electrochemical properties of Fe–Nb–B amorphous alloys doped with disprosium or terbium,” Fiz.-Khim. Mekh. Mater., 50, No. 5, 29–33 (2014); English translation: Mater. Sci., 50, No. 5, 653–658 (2015).
V. M. Artemenko, M. D. Sakhnenko, and V. V. Shtefan, Methodical Recommendations for the Laboratory Work on the “Kinetics of Electrolytic Hydrogen Release” for Full-Time and Extramural Students Specialized in “Technical Electrochemistry” and “Chemical Technology of Rare Dispersed Elements and Materials Based on These Elements [in Ukrainian], “KhPI” National Technical University, Kharkiv (2007).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Fizyko-Khimichna Mekhanika Materialiv, Vol. 55, No. 5, pp. 84–89, September–October, 2019.
Rights and permissions
About this article
Cite this article
Boichyshyn, L.M., Hertsyk, О.М., Lopachak, М.М. et al. Electrochemical Properties of Ternary Amorphous Alloys Based on Iron and Cobalt in Alkali Solutions. Mater Sci 55, 703–709 (2020). https://doi.org/10.1007/s11003-020-00361-w
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11003-020-00361-w