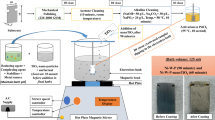
Abstract
The aim of this paper is electrodeposition of Zn/CeO2 and Zn-Mn/CeO2 nanocomposite coatings. Four plating solutions were used (R1–R4). Solution R1 was without manganese ions, whereas solutions R2, R3, and R4 had Mn2+/Zn2+ ratios of 1:1, 1:2, and 2:1, respectively. Electrodeposition is performed galvanostatically at 1 A dm−2, 2 A dm−2, and 4 A dm−2 current densities with ultrasonic bath assisted mixing. The morphology of the coatings was observed using optical and scanning electron microscopy, the chemical compositions were determined using energy dispersive x-ray spectroscopy, whereas electrochemical impedance spectroscopy and potentiodynamic polarization method were used to determine the corrosion resistance of the coatings. All the solutions yield compact coatings with a small grain size and surface roughness ranging from 0.6 to 1.47 μm at all current densities. The results suggest that with the increase in current density, the content of manganese in the coating decrease, whereas the content of CeO2 particles increases in Zn-Mn/CeO2 coatings. Corrosion resistance of the coatings obtained from R2, R3, and R4 solutions deposited at lower current densities is higher compared to the coatings deposited from solution R1. The coating with the highest corrosion resistance was obtained from solution R2 at 1 A dm−2 current density.
Graphical Abstract






Similar content being viewed by others
References
Tuaweri TJ, Jombo PP, Okpala AN (2014) Corrosion resistance characteristics of Zn-Ni/SiO2 composite coatings. IJAMSE 3(2):1–12
Ghaziof S, Gao W (2015) The effect of pulse electroplating on Zn–Ni alloy and Zn–Ni–Al2O3 composite coatings. J Alloys Compd 622:918–924. https://doi.org/10.1016/j.jallcom.2014.11.025
Bajat J, Mišković-Stanković V, Maksimović M, Dražić D, Zec S (2002) Electrochemical deposition and characterization of Zn-Co alloys and corrosion protection by electrodeposited epoxy coating on Zn-Co alloy. Electrochim Acta 47:4101–4112. https://doi.org/10.1016/S0013-4686(02)00418-8
Shourgeshty M, Aliofkhazraei M, Karimzadeh A (2019) Study on functionally graded Zn–Ni–Al2O3 coatings fabricated by pulse-electrodeposition. Surf Eng 35(2):167–176. https://doi.org/10.1080/02670844.2018.1432172
Ghaziof S, Kilmartin PA, Gao W (2015) Electrochemical studies of sol-enhanced Zn–Ni–Al2O3 composite and Zn–Ni alloy coatings. J Electroanal 755:63–70. https://doi.org/10.1016/j.jelechem.2015.07.041
Claudel F, Stein N, Allain N, Tidu A, Hajczak N, Lallement R, Close D (2019) Pulse electrodeposition and characterization of Zn–Mn coatings deposited from additive-free chloride electrolytes. J Appl Electrochem 49:399–411. https://doi.org/10.1007/s10800-019-01295-1
Tomić MV, Riđošić MG, Pavlović MG, Jokić M, Bajat J (2015) Impact of roughness of Zn-Mn coatings on corrosive stability. Material protection 56(1):75–80. https://doi.org/10.5937/ZasMat1501075T
Duszczyk J, Siuzdak K, Klimczuk T, Strychalska-Nowak J, Zaleska-Medynska A (2018) Manganese phosphatizing coatings: the effects of preparation conditions on surface properties. Materials 11(12):2585. https://doi.org/10.3390/ma11122585
Tomić MV, Bučko MM, Pavlović MG, Bajat JB (2010) Corrosion stability of electrochemically deposited Zn–Mn alloy coatings. Contemp Mater I–1: 87–93. https://doi.org/10.5767/anurs.cmat.100101.en.087T
Bučko MM, Stevanović S, Tomić MV, Pavlović M, Bajat J (2011) The peculiarities of electrochemical deposition and morphology of Zn-Mn alloy coatings obtained from pyrophosphate electrolyte. HEM IND 65(3):295–303. https://doi.org/10.2298/HEMIND101214003B
Bucko M, Riđošić M, Kovačina J, Tomić M, Bajat J (2022) Hardness and corrosion resistance of Zn− Mn/Al2O3 composite coatings produced by electrochemical deposition. IJEMS 29(4):540–549. https://doi.org/10.56042/ijems.v29i4.47785
Tomić MV, Petrović MM, Stanković S, Stevanović S, Bajat J (2015) Ternary Zn-Ni-Co alloy: anomalous codeposition and corrosion stability. J Serb Chem Soc 80(1):73–86. https://doi.org/10.2298/JSC260814113B
Wilcox GD, Petersen B (1996) Zinc manganese alloy electrodeposition. Transaction of IMF 74(4):115–118. https://doi.org/10.1080/00202967.1996.11871109
Close D, Stein N, Allain N, Tidu A, Drynski E, Merklein M, Lallement R (2016) Electrodeposition, microstructural characterization and anticorrosive properties of Zn-Mn alloy coatings from acidic chloride electrolyte containing 4-hydroxybenzaldehyde and ammonium thiocyanate. Surf Coat Technol 298:73–82. https://doi.org/10.1016/j.surfcoat.2016.04.043
Stein N, Claudel F, Allain N, Tidu A, Close D (2022) Anticorrosive performance and corrosion mechanisms of Zn–Mn-coated steel. Mater Corros. https://doi.org/10.1002/maco.202213473
Close D, Stein N, Allain N, Tidu A, Drynski E, Merklein M, Lallement R (2016) Electrodeposition, microstructural characterization and anticorrosive properties of Zn-Mn alloy coatings. In Electrochemical Society Meeting Abstracts 229 (No. 15, pp. 981–981). The Electrochemical Society, Inc. https://doi.org/10.1149/MA2016-01/15/981/meta
Boshkov N, Petrov K, Vitkova S, Raichevsky G (2005) Galvanic alloys Zn–Mn composition of the corrosion products and their protective ability in sulfate containing medium. Surf Coat Technol 194 (2–3): 276–282.https://doi.org/10.1016/j.surfcoat.2004.09.016
Loukil N, Feki M (2021) Chronopotentiometric study and morphological-structural characterization of environmentally friendly protective Zn-Mn coatings. J Electrochem Soc 168:073501. https://doi.org/10.1149/1945-7111/ac0aa5
Ganesan S, Prabhu G, Popov BN (2014) Electrodeposition and characterization of Zn-Mn coatings for corrosion protection. Surf Coat Technol 238:143–151. https://doi.org/10.1016/j.surfcoat.2013.10.062
Nishida Y (2001) Introduction to metal matrix composites: fabrication and recycling, AIST, Nagoya, Japan
Donnet C, Erdemir A (2004) Historical developments and new trends in tribological and solid lubricant coatings. Surf Coat Technol 180–181:76–84. https://doi.org/10.1016/j.surfcoat.2003.10.022
Riđošić M, Bučko M, Salicio-Paz A, Živković G-L, LS, Bajat JB, (2021) Ceria particles as efficient dopant in the electrodeposition of Zn-Co-CeO2 composite coatings with enhanced corrosion resistance: the effect of current density and particle concentration. Molecules 26(15):4578. https://doi.org/10.3390/molecules26154578
Veprek S (2004) Superhard nanocomposites: design concept, properties, present and future industrial applications. Eur Phys J Appl Phys 28:313–317. https://doi.org/10.1051/epjap:2004169
Riđošić M, Nikolić ND, Salicio-Paz A, García-Lecina E, Živković LS, Bajat J (2021) The effect of the ultrasound agitation and source of ceria particles on the morphology and structure of the Zn–Co–CeO2 composite coatings. Metals 11(5):704. https://doi.org/10.3390/met11050704
Riđošić M, Salicio-Paz A, Garcia-Lecina E, Zabinski P, Živković LS (2021) The effect of the ultrasound agitation and source of ceria particles on the morphology and structure of the Zn–Co–CeO2 composite coatings. JMRT 13:1336–1349. https://doi.org/10.1016/j.jmrt.2021.05.064
Walsh F, Ponce de Leon C (2014) A review of the electrodeposition of metal matrix composite coatings by inclusion of particles in a metal layer: an established and diversifying technology. Transactions of the IMF 92(2):83–98. https://doi.org/10.1179/0020296713Z.000000000161
Camargo MK, Tudela I, Schmidt U, Cobley AJ, Bund A (2016) Ultrasound assisted electrodeposition of Zn and Zn-TiO2 coatings. Electrochim Acta 198:287–295. https://doi.org/10.1016/j.electacta.2016.03.078
Montemor MF, Pinto R, Ferreira MGS (2009) Chemical composition and corrosion protection of saline films modified with CeO2 nanoparticles. Electrochim Acta 54(22):5179–5189. https://doi.org/10.1016/j.electacta.2009.01.053
Calado LM, Taryba MG, Carmezim MJ, Montemor MF (2018) Selfhealing ceria-modified coating for corrosion protection of AZ31 magnesium alloy. Corr Sci 142:12–21. https://doi.org/10.1016/j.corsci.2018.06.013
Ma Y, Yhang Y, Liu J, Ge Y, Yan X, Sun Y et al (2020) GO-modified double-walled polyurea microcapsules/epoxy composites for marine anticorrosive self-healing coating. Mater Des 189:108547. https://doi.org/10.1016/j.matdes.2020.108547
Cambon JB, Ansart F, Bonino JP, Turq V, Wu J, Zhang P (2012) Effect of cerium concentration on corrosion resistance and polymerization of hybrid sol-gel coating on martensitic stainless steel. Prog Org Coating 75(4):486–493. https://doi.org/10.1016/j.porgcoat.2012.06.005
Wu LK, Zhang JT, Hu JM, Zhang JQ (2012) Improved corrosion performance of electrophoretic coatings by silane addition. Corr Sci 56:58–66. https://doi.org/10.1016/j.corsci.2011.11.018
Ferraris S, Vitale A, Bertone E, Guastella S, Cassinelli C, Pan J, Spriano S (2016) Multifunctional commercially pure titanium for the improvement of bone integration: multiscale topography, wettability, corrosion resistance and biological functionalization. Mater Sci Eng C 60:384–393. https://doi.org/10.1016/j.msec.2015.11.049
Ma Y, Zhang Y, Liu J, Ge Y, Yan X, Sun Y, Wu J, Zhang P (2020) GO-modified double-walled polyurea microcapsules/epoxy composites for marine anticorrosive self-healing coating. Mater Des 189:108547. https://doi.org/10.1016/j.matdes.2020.108547
Jyotheender KS, Srivastava C (2019) Ni-graphene oxide composite coatings: Optimum graphene oxide for enhanced corrosion resistance. COMPOS PART B-ENG 175:107145. https://doi.org/10.1016/j.compositesb.2019.107145
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tomić, M.V., Mitrović, M.G. Electrodeposition of Zn-Mn/CeO2 composite coatings: evaluation of corrosion properties. J Solid State Electrochem 27, 1901–1910 (2023). https://doi.org/10.1007/s10008-023-05441-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-023-05441-w