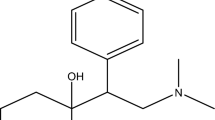
Abstract
We report a new type of solid-state electrode (type I) of a simple design with polyvinyl chloride membranes based on Mn(III) tetraphenylporphyrin and with graphite as the electronically conducting substrate. Enlargement of the membrane/graphite contact area by soaking graphite in the plasticizer with subsequent conditioning of the electrode at 30 °C allowed us to shorten the time required to achieve steady potential values of the sensors to just 3 days. These electrodes do not require a specially added RedOx system in the transducer layer. Stabilization of the EMF response of type I electrodes is compared to type II electrodes which contain a Cu0/Cu2+ RedOx couple in the transducer layer. Type I sensors are suitable for measuring the salicylate ion concentration in the clinically important concentration range down to 2.5 × 10−4 M with a sensitivity to salicylate ion of −59.0 mV decade−1 in solutions with a high constant background of chloride ions of 0.12 M at pH = 5.3, making this a promising technique for an effective design of solid-contact ion-selective electrodes with polymeric sensing membranes.

ᅟ







Similar content being viewed by others
References
Gao W-Y, Chrzanowski M, Ma S (2014) Metal-metalloporphyrin frameworks: a resurging class of functional materials. Chem Soc Rev 43(16):5841–5866
Biesaga M, Pyrzyńska K, Trojanowicz M (2000) Porphyrins in analytical chemistry. A review Talanta 51(2):209–224
Koposova E, Liu X, Pendin A, Thiele B, Shumilova G, Ermolenko Y, Offenhausser A, Mourzina Y (2016) Influence of Meso-substitution of the Porphyrin ring on enhanced hydrogen evolution in a photochemical system. J Phys Chem C 120(26):13873–13890
Koposova E, Shumilova G, Ermolenko Y, Kisner A, Offenhausser A, Mourzina Y (2015) Direct electrochemistry of cyt c and hydrogen peroxide biosensing on oleylamine- and citrate-stabilized gold nanostructures. Sensors and Actuators B-Chemical 207:1045–1052
Kimmel DW, LeBlanc G, Meschievitz ME, Cliffel DE (2012) Electrochemical sensors and biosensors. Anal Chem 84(2):685–707
Zhang X-B, Guo C-C, Jian L-X, G-L SHEN, Yu R-Q (2000) Bismetalloporphyrin complexes as ionic carriers for a salicylate-sensitive electrode. Anal Sci 16(12):1285–1289
Paolesse R, Nardis S, Monti D, Stefanelli M, Di Natale C (2017) Porphyrinoids for chemical sensor applications. Chem Rev 117(4):2517–2583
Ermolenko Y, Yoshinobu T, Mourzina Y, Levichev S, Furuichi K, Vlasov Y, Schoning MJ, Iwasaki H (2002) Photocurable membranes for ion-selective light-addressable potentiometric sensor. Sensors and Actuators B-Chemical 85(1–2):79–85
Santos EMG, Araújo AN, Couto CMCM, Montenegro MCBSM, Kejzlarová A, Solich P (2004) Ion selective electrodes for penicillin-G based on Mn(III)TPP-Cl and their application in pharmaceutical formulations control by sequential injection analysis. J Pharm Biomed Anal 36(4):701–709
Bühlmann P, Pretsch E, Bakker E (1998) Carrier-based ion-selective electrodes and bulk Optodes. 2. Ionophores for potentiometric and optical sensors. Chem Rev 98(4):1593–1688
Chen LD, Zou XU, Bühlmann P (2012) Cyanide-selective electrode based on Zn (II) Tetraphenylporphyrin as Ionophore. Anal Chem 84(21):9192–9198
Johnson RD, Bachas LG (2003) Ionophore-based ion-selective potentiometric and optical sensors. Anal Bioanal Chem 376(3):328–341
Lvova L, Di Natale C, Paolesse R (2013) Porphyrin-based chemical sensors and multisensor arrays operating in the liquid phase. Sensors Actuators B Chem 179:21–31
Shahrokhian S (2001) Lead Phthalocyanine as a selective carrier for preparation of a cysteine-selective electrode. Anal Chem 73(24):5972–5978
Shirmardi-Dezaki A, Shamsipur M, Akhond M, Sharghi H (2013) Cyanide selective electrodes based on a porphyrinatoiron (III) chloride derivative. J Electroanal Chem 689:63–68
Vlascici DPI, Chiriac VA, Fagadar-Cosma G, Popovici H, Fagadar-Cosma E (2013) Chemistry Central Journal 7:1–7
Farhadi K, Maleki R, Hosseinzadeh Yamchi R, Sharghi H, Shamsipur M (2004) [Tetrakis (4-N, N-dimethylaminobenzene) porphyrinato]-manganese (III) acetate as a novel carrier for a selective iodide PVC membrane electrode. Anal Sci 20(5):805–809
Santos EMG, Araújo AN, Couto CMCM, Montenegro MCBSM (2006) Construction and evaluation of PVC and sol–gel sensor membranes based on Mn(III)TPP-Cl. Application to valproate determination in pharmaceutical preparations. Anal Bioanal Chem 384(4):867–875
Starikova TA, Shumilova GI, Valiotti AB (2013) Electrochemical characteristics of membranes based on Mn(III) tetraphenylporphyrin. In English. Russ J Electrochem 49(9):856–862
Trinder P (1954) Rapid determination of salicylate in biological fluids. Biochem J 57(2):301–303
Zhu Y, Guan X, Ji H (2009) Electrochemical solid phase micro-extraction and determination of salicylic acid from blood samples by cyclic voltammetry and differential pulse voltammetry. J Solid State Electrochem 13(9):1417–1423
Zhang W-D, Xu B, Hong Y-X, Yu Y-X, Ye J-S, Zhang J-Q (2010) Electrochemical oxidation of salicylic acid at well-aligned multiwalled carbon nanotube electrode and its detection. J Solid State Electrochem 14(9):1713–1718
do Prado TM, SAS M (2016) Spectroelectrochemical study of salicylate in alkaline medium. J Solid State Electrochem 20(9):2569–2574
Batista EA, Temperini MLA (2007) An in situ SERS and FTIRAS study of salicylate interaction with copper electrode. J Solid State Electrochem 11(11):1559–1565
Shishkanova TV, Videnská K, Antonova SG, Kronďák M, Fitl P, Kopecký D, Vrňata M, Král V (2014) Application of polyaniline for potentiometric recognition of salicylate and its analogues. Electrochim Acta 115:553–558
Shahrokhian S, Hamzehloei A, Bagherzadeh M (2002) Chromium (III) Porphyrin as a selective Ionophore in a salicylate-selective membrane electrode. Anal Chem 74(14):3312–3320
Shahrokhian S, Amini MK, Kia R, Tangestaninejad S (2000) Salicylate-selective electrodes based on al (III) and Sn (IV) Salophens. Anal Chem 72(5):956–962
Poursaberi T, Hassanisadi M (2012) Application of metalloporphyrin grafted-graphene oxide for the construction of a novel salicylate-selective electrode. J Porphyrins Phthalocyanines 16(10):1140–1147
Messick MS, Krishnan SK, Hulvey MK, Steinle ED (2005) Development of anion selective polymer membrane electrodes based on lutetium (III) porphyrins. Anal Chim Acta 539(1–2):223–228
Malinowska E, Niedziółka J, Roźniecka E, Meyerhoff ME (2001) Salicylate-selective membrane electrodes based on Sn (IV)- and O Mo (V)-porphyrins: differences in response mechanism and analytical performance. J Electroanal Chem 514(1–2):109–117
Luo E-P, Chai Y-Q, Yuan R, Dai J-Y, Xu L (2009) Highly salicylate-selective membrane electrode based on a new thiomacrocyclic Schiff base complex of binuclear copper (II) as neutral carrier. Desalination 249(2):615–620
Ganjali MR, Norouzi P, Faridbod F, Rezapour M, Ahmadi A (2007) Application of tetra cyclohexyl tin (IV) as an anionic carrier for the construction of a new salicylate membrane sensor. J Chin Chem Soc 54(4):969–976
Yousry MI, Hosny I, Ola RS (2012) New copper (II)-selective chemically modified carbon paste electrode based on etioporphyrin I dihydrobromide. J Electroanal Chem 666:11–18
Vamvakaki M, Chaniotakis NA (1996) Solid-contact ion-selective electrode with stable internal electrode. Anal Chim Acta 320(1):53–61
Szűcs J, Lindfors T, Bobacka J, Gyurcsányi RE (2016) Ion-selective electrodes with 3D nanostructured conducting polymer solid contact. Electroanalysis 28(4):778–786
Ruzicka J, Rald K (1971) The liquid-state, iodide-selective electrode. Anal Chim Acta 53(1):1–12
Michalska A (2012) All-solid-state ion selective and all-solid-state reference electrodes. Electroanalysis 24(6):1253–1265
Kakhki S, Shams E, Barsan MM (2013) Fabrication of carbon paste electrode containing a new inorganic–organic hybrid based on [SiW12O40]4− polyoxoanion and Nile blue and its electrocatalytic activity toward nitrite reduction. J Electroanal Chem 704:80–85
Ivanova NM, Levin MB, Mikhelson KN (2012) Problems and prospects of solid contact ion-selective electrodes with ionophore-based membranes. Russ Chem Bull 61(5):926–936
Hu J, Stein A, Bühlmann P (2016) Rational design of all-solid-state ion-selective electrodes and reference electrodes. Trends Anal Chem 76:102–114
Brinić S, Buzuk M, Bralić M, Generalić E (2012) Solid-contact Cu (II) ion-selective electrode based on 1, 2-di-(o-salicylaldiminophenylthio)ethane. J Solid State Electrochem 16(4):1333–1341
Fierke MA, Lai C-Z, Bühlmann P, Stein A (2010) Effects of architecture and surface chemistry of three-dimensionally ordered Macroporous carbon solid contacts on performance of ion-selective electrodes. Anal Chem 82(2):680–688
Ivanova NM, Podeshvo IV, Goikhman MY, Yakimanskii AV, Mikhelson KN (2013) Potassium-selective solid contact electrodes with poly(amidoacid) Cu(I) complex, electron-ion exchanging resin and different sorts of carbon black in the transducer layer. Sensors Actuators B Chem 186:589–596
Paczosa-Bator B (2012) All-solid-state selective electrodes using carbon black. Talanta 93:424–427
Saravia LPH, Anandhakumar S, Parussulo ALA, Matias TA, Caldeira da Silva CC, Kowaltowski AJ, Araki K, Bertotti M (2016) Development of a tetraphenylporphyrin cobalt (II) modified glassy carbon electrode to monitor oxygen consumption in biological samples. J Electroanal Chem 775:72–76
Jarvis JM, Guzinski M, Pendley BD, Lindner E (2016) Poly(3-octylthiophene) as solid contact for ion-selective electrodes: contradictions and possibilities. J Solid State Electrochem 20(11):3033–3041
Mikhelson K (2013) Ion-selective Electrodes. In: Lecture notes in chemistry, vol 81. Springer, Heidelberg-New York-Dordrecht-London
Bobacka J, Ivaska A, Lewenstam A (2008) Potentiometric ion sensors. Chem Rev 108(2):329–351
Khripoun GA, Volkova EA, Liseenkov AV, Mikhelson KN (2006) Nitrate-selective solid contact electrodes with poly(3-octylthiophene) and poly(aniline) as ion-to-electron transducers buffered with electron-ion-exchanging resin. Electroanalysis 18(13–14):1322–1328
Dean J (1974) Lange's Handbook of. chemistry. 15 edn.,
Kulapin AI, Chernova RK, Kulapina EG (2005) Some regularities of interface formation in solid-contact potentiometric surfactant-selective sensors. In English. J Anal Chem 60(3):282–288
Kulapin AI, Mikhailova AM, Kulapina EG (2003) Stabilizing potential of solid-contact sensors selective towards surface-active substances. In English. Russ J Electrochem 39(5):585–590
Kulapin AI, Chernova RK, Kulapina EG (2002) New modified electrodes for the separate determination of polyoxyethylated nonylphenols. In English. J Anal Chem 57(7):638–643
Rezaei B, Meghdadi S, Nafisi V (2007) Fast response and selective perchlorate polymeric membrane electrode based on bis (dibenzoylmethanato) nickel (II) complex as a neutral carrier. Sensors Actuators B Chem 121(2):600–605
Valiotti AB, Vasil’eva OE, Starikova TA, Semeikin AS, Shumilova GI (2011) Electrochemical properties and spectrophotometry of membranes based on holmium tetraphenylporphyrinchloride. In English. Russ J Gen Chem 81(4):762
Moreno-Castilla C (2004) Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 42(1):83–94
Wang J, Wang F, Yao J, Guo H, Blake RE, Choi MMF, Song C (2013) Effect of pH and temperature on adsorption of dimethyl phthalate on carbon nanotubes in aqueous phase. Anal Lett 46(2):379–393
Starikova TASG, Pendin AA (2012) Russ J Gen Chem 82(11):1909–1914
Chaniotakis N, Chasser A, Meyerhoff M, Groves J (1988) Influence of porphyrin structure on anion selectivities of manganese (III) porphyrin based membrane electrodes. Anal Chem 60(2):185–188
Malinski T (2000) Porphyrin-based electrochemical sensors. In: Kadish KM, Smith KM, Guilard M (eds) The porphyrin handbook, vol vol 6. Academic Press, USA, pp 232–256
Chou J-H, Kosal ME, Nalwa HS, Rakow NA, Suslick KS (2000) Applications of porphyrins and metalloporphyrins to materials chemistry. In: Kadish K, Smith KM, Guilard R (eds) The porphyrin handbook, vol vol 6. Academic Press, USA, pp 44–131
Zhai J, Xie X, Bakker E (2015) Ion-selective optode nanospheres as heterogeneous indicator reagents in complexometric titrations. Anal Chem 87(5):2827–2831
Hill CL, Williamson MM (1985) Structural and electronic properties of six-coordinate manganese (III) porphyrin cations. Crystal and molecular structure of bis (N,N-dimethylformamide)(tetraphenylporphinato) manganese (III) perchlorate, [MnIIITPP(DMF)2]+ ClO4 -. Inorg Chem 24(18):2836–2841
Boucher LJ (1972) Manganese porphyrin complexes. Coord Chem Rev 7(3):289–329
Boucher LJ (1968) Manganese porphyrin complexes. I. Synthesis and spectroscopy of manganese (III) protoporphyrin IX dimethyl ester halides. J Am Chem Soc 90(24):6640–6645
Great Medical Encyclopedia (1981), vol 15. M. Soviet Encyclopedia,
Burns DT, Danzer K, Townshend A (2002) Use of the term "Recovery" and "Apparent Recovery" in analytical procedures (IUPAC recommendations 2002). Pure Appl Chem 74:2201–2205
Koch-Weser J (1972) Serum drug concentrations as therapeutic guides. N Engl J Med 287:227–231
Pau CP, Rechnitz GA (1984) Bound coefactor/dual enzyme electrode system for l-alanine. Anal Chim Acta 160:141–147
Ganjali MR, Kiani-Anbouhi R, Pourjavid MR, Salavati-Niasari M (2003) Bis (trans-cinnamaldehyde) ethylene diimine dibromonickel (II) complex as a neutral carrier for salicylate-selective liquid membrane and coated graphite sensors. Talanta 61(3):277–284
Chernyshov DV, Egorov VM, Shvedene NV, Pletnev IV (2009) Low-melting ionic solids: versatile materials for ion-sensing devices. ACS Appl Mater Interfaces 1(9):2055–2059
Acknowledgements
The authors acknowledge Saint-Petersburg State University for a research grant 12.38.218.2015. The Resource Educational Center of the St. Petersburg State University in chemistry is acknowledged for the scanning electron microscopy investigations.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Skripnikova, T.A., Starikova, A.A., Shumilova, G.I. et al. Towards stabilization of the potential response of Mn(III) tetraphenylporphyrin-based solid-state electrodes with selectivity for salicylate ions. J Solid State Electrochem 21, 2269–2279 (2017). https://doi.org/10.1007/s10008-017-3575-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-017-3575-6