Abstract
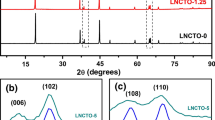
Li[Ni1/3Co1/3Mn1/3]O2 (NCM 111) is a promising alternative to LiCoO2, as it is less expensive, more structurally stable, and has better safety characteristics. However, its capacity of 155 mAh g−1 is quite low, and cycling at potentials above 4.5 V leads to rapid capacity deterioration. Here, we report a successful synthesis of lithium-rich layered oxides (LLOs) with a core of LiMO2 (R-3m, M = Ni, Co) and a shell of Li2MnO3 (C2/m) (the molar ratio of Ni, Co to Mn is the same as that in NCM 111). The core–shell structure of these LLOs was confirmed by XRD, TEM, and XPS. The Rietveld refinement data showed that these LLOs possess less Li+/Ni2+ cation disorder and stronger M*–O (M* = Mn, Co, Ni) bonds than NCM 111. The core–shell material Li1.15Na0.5(Ni1/3Co1/3)core(Mn1/3)shellO2 can be cycled to a high upper cutoff potential of 4.7 V, delivers a high discharge capacity of 218 mAh g−1 at 20 mA g−1, and retains 90 % of its discharge capacity at 100 mA g−1 after 90 cycles; thus, the use of this material in lithium ion batteries could substantially increase their energy density.

Average voltage vs. number of cycles for the core–shell and pristine materials at 20 mA g−1 for 10 cycles followed by 90 cycles at 100 mA g−1










Similar content being viewed by others
References
Santhanam R, Rambabu B (2010) Research progress in high voltage spinel LiNi0.5Mn1.5O4 material. J Power Sources 195(17):5442–5451
Han S, Xia Y, Wei Z, Qiu B, Pan L, Gu Q, Liu Z, Guo Z (2015) A comparative study on the oxidation state of lattice oxygen among Li1.14Ni0.136Co0.136Mn0.544O2, Li2MnO3, LiNi0.5Co0.2Mn0.3O2 and LiCoO2 for the initial charge–discharge. J Mater Chem A 3(22):11930–11939
Dou S (2013) Review and prospect of layered lithium nickel manganese oxide as cathode materials for Li-ion batteries. J Solid State Electrochem 17(4):911–926
Kim H-S, Kim K, Moon S-I, Kim I-J, Gu H-B (2008) A study on carbon-coated LiNi1/3Mn1/3Co1/3O2 cathode material for lithium secondary batteries. J Solid State Electrochem 12(7–8):867–872
Van der Ven A, Aydinol MK, Ceder G (1998) First-principles evidence for stage ordering in LixCoO2. J Electrochem Soc 145(6):2149–2155
Jiang X, Sha Y, Cai R, Shao Z (2015) The solid-state chelation synthesis of LiNi1/3Co1/3Mn1/3O2 as a cathode material for lithium-ion batteries. J Mater Chem A 3(19):10536–10544
Noh H-J, Youn S, Yoon CS, Sun Y-K (2013) Comparison of the structural and electrochemical properties of layered Li[Ni x Co y Mn z ]O2 (x = 1/3, 0.5, 0.6, 0.7, 0.8 and 0.85) cathode material for lithium-ion batteries. J Power Sources 233:121–130
Xia H, Lu L, Meng YS, Ceder G (2007) Phase transitions and high-voltage electrochemical behavior of LiCoO2 thin films grown by pulsed laser deposition. J Electrochem Soc 154(4):A337
Lin B, Wen Z, Wang X, Liu Y (2010) Preparation and characterization of carbon-coated Li[Ni1/3Co1/3Mn1/3]O2 cathode material for lithium-ion batteries. J Solid State Electrochem 14(10):1807–1811
Yang S, Wang X, Yang X, Liu L, Liu Z, Bai Y, Wang Y (2011) Influence of Li source on tap density and high rate cycling performance of spherical Li[Ni1/3Co1/3Mn1/3]O2 for advanced lithium-ion batteries. J Solid State Electrochem 16(3):1229–1237
Yano A, Aoyama S, Shikano M, Sakaebe H, Tatsumi K, Ogumi Z (2015) Surface structure and high-voltage charge/discharge characteristics of Al-oxide coated LiNi1/3Co1/3Mn1/3O2 cathodes. J Electrochem Soc 162(2):A3137–A3144
Thackeray MM, Kang S-H, Johnson CS, Vaughey JT, Benedek R, Hackney SA (2007) Li2MnO3-stabilized LiMO2 (M = Mn, Ni, Co) electrodes for lithium-ion batteries. J Mater Chem 17(30):3112–3125
Kang SH, Thackeray MM (2008) Stabilization of xLi2MnO3⋅(1 − x)LiMO2 electrode surfaces (M = Mn, Ni, Co) with mildly acidic, fluorinated solutions. J Electrochem Soc 155(4):A269
Kim S-M, Jin B-S, Lee S-M, Kim H-S (2015) Effects of the fluorine-substitution and acid treatment on the electrochemical performances of 0.3Li2MnO3 · 0.7LiMn0.60Ni0.25Co0.15O2 cathode material for Li-ion battery. Electrochim Acta 171:35–41
Mezaal MA, Qu L, Li G, Zhang R, Jiang X, Zhang K, Liu W, Lei L (2015) Promoting the cyclic and rate performance of lithium-rich ternary materials via surface modification and lattice expansion. RSC Adv 5(113):93048–93056
Li J, Xu Y, Li X, Zhang Z (2013) Li2MnO3 stabilized LiNi1/3Co1/3Mn1/3O2 cathode with improved performance for lithium ion batteries. Appl Surf Sci 285:235–240
Yu H, Zhou H (2013) High-energy cathode materials (Li2MnO3-LiMO2) for lithium-ion batteries. J Phys Chem Lett 4(8):1268–1280
Yang C, Zhang Q, Ding W, Zang J, Lei M, Zheng M, Dong Q (2015) Improving the electrochemical performance of layered lithium-rich cathode materials by fabricating a spinel outer layer with Ni3+. J Mater Chem A 3(14):7554–7559
Yan J, Liu X, Li B (2014) Recent progress in Li-rich layered oxides as cathode materials for Li-ion batteries. RSC Adv 4(108):63268–63284
Long BR, Croy JR, Dogan F, Suchomel MR, Key B, Wen J, Miller DJ, Thackeray MM, Balasubramanian M (2014) Effect of cooling rates on phase separation in 0.5Li2MnO3·0.5LiCoO2 electrode materials for Li-ion batteries. Chem Mater 26(11):3565–3572
Son MY, Lee JK, Kang YC (2014) Fabrication and electrochemical performance of 0.6Li2MnO3-0.4Li(Ni1/3Co1/3Mn1/3)O2 microspheres by two-step spray-drying process. Sci Rep 4:5752
Park MS (2014) First-principles study of native point defects in LiNi1/3Co1/3Mn1/3O2 and Li2MnO3. Phys Chem Chem Phys 16(31):16798–16804
Kumar PS, Sakunthala A, Reddy MV, Shanmugam S, Prabu M (2015) Correlation between the structural, electrical and electrochemical performance of layered Li(Ni0.33Co0.33Mn0.33)O2 for lithium ion battery. J Solid State Electrochem 20(7):1665–1876
Boultif A, Louër D (2004) Powder pattern indexing with the dichotomy method. J Appl Crystallogr 37(5):724–731
Liu W, Oh P, Liu X, Lee MJ, Cho W, Chae S, Kim Y, Cho J (2015) Nickel-rich layered lithium transition-metal oxide for high-energy lithium-ion batteries. Angew Chem Int Ed Engl 54(15):4440–4457
Oh P, Ko M, Myeong S, Kim Y, Cho J (2014) A novel surface treatment method and new insight into discharge voltage deterioration for high-performance 0.4Li2MnO3-0.6LiNi1/3Co1/3Mn1/3O2 cathode materials. Adv Energ Mater 4(16):1400631
Noh JK, Kim S, Kim H, Choi W, Chang W, Byun D, Cho BW, Chung KY (2014) Mechanochemical synthesis of Li2MnO3 shell/LiMO2 (M = Ni, Co, Mn) core-structured nanocomposites for lithium-ion batteries. Sci Rep 4:4847
Ryu W-H, Kim D-H, Kang S-H, Kwon H-S (2013) Electrochemical properties of nanosized Li-rich layered oxide as positive electrode materials for Li-ion batteries. RSC Adv 3(22):8527–8534
Yang J, Hou M, Haller S, Wang Y, Wang C, Xia Y (2016) Improving the cycling performance of the layered Ni-rich oxide cathode by introducing low-content Li2MnO3. Electrochim Acta 189:101–110
Li Q, Li G, Fu C, Luo D, Fan J, Xie D, Li L (2015) Balancing stability and specific energy in Li-rich cathodes for lithium ion batteries: a case study of a novel Li–Mn–Ni–Co oxide. J Mater Chem A 3(19):10592–10602
Fu F, Deng Y-P, Shen C-H, Xu G-L, Peng X-X, Wang Q, Xu Y-F, Fang J-C, Huang L, Sun S-G (2014) A hierarchical micro/nanostructured 0.5Li2MnO3·0.5LiMn0.4Ni0.3Co0.3O2 material synthesized by solvothermal route as high rate cathode of lithium ion battery. Electrochem Commun 44:54–58
Shen CH, Huang L, Lin Z, Shen SY, Wang Q, Su H, Fu F, Zheng XM (2014) Kinetics and structural changes of Li-rich layered oxide 0.5Li2MnO3.0.5LiNi0.292Co0.375Mn0.333O2 material investigated by a novel technique combining in situ XRD and a multipotential step. ACS Appl Mater Interfaces 6(15):13271–13279
Li W, Reimers J, Dahn J (1992) Crystal structure of Li x Ni2−x O2 and a lattice-gas model for the order–disorder transition. Phys Rev B 46(6):3236–3246
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mezaal, M.A., Qu, L., Li, G. et al. LiMO2@Li2MnO3 positive-electrode material for high energy density lithium ion batteries. J Solid State Electrochem 21, 145–152 (2017). https://doi.org/10.1007/s10008-016-3345-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-016-3345-x