Abstract
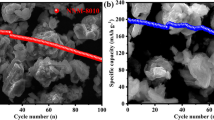
We report on the electrochemical properties of layered manganese oxides, with and without cobalt substituents, as cathodes in sodium ion batteries. We fabricated sub-micrometre-sized particles of Na0.7MnO2 + z and Na0.7Co0.11Mn0.89O2 + z via combustion synthesis. X-ray diffraction revealed the same layered hexagonal P2-type bronze structure with high crystallinity for both materials. Potentiostatic and galvanostatic charge/discharge cycles in the range 1.5–3.8 V vs. Na | Na+ were performed to identify potential-dependent phase transitions, capacity, and capacity retention. After charging to 3.8 V, both materials had an initial discharge capacity of 138 mA h g−1 at a rate of 0.3 C. For the 20th cycle, those values reduced to 75 and 92 mA h g−1 for Co-free and Co-doped samples, respectively. Our findings indicate that earlier works probably underestimated the potential of (doped) P2-type Na0.7MnO2 + z as cathode material for sodium ion batteries in terms of capacity and cycle stability. Apart from doping, a simple optimization parameter seems to be the particle size of the active material.






Similar content being viewed by others
References
Wu Y, Wan C, Jiang C, Fang S (2002) Introduction, principles and advances of lithium secondary batteries. Tsinghua University Press, Beijing
Scrosati B, Garche J (2010) J Power Sources 195:2419–2430
Liu C, Li F, Ma L, Cheng H (2010) Adv Mater 22:E28–E62
Ceder G, Hautier G, Jain A, Ong SP (2012) MRS Bull 37:185–191
Vetter J, Novák P, Wagner MR, Veit C, Möller K-C, Besenhard JO, Winter M, Wohlfahrt-Mehrens M, Vogler C, Hammouche A (2005) J Power Sources 147:269–281
Wu YP, Rahm E, Holze R (2002) Electrochim Acta 47:3491–3507
Liu L, Tian F, Zhou M, Guo H, Wang X (2012) Electrochim Acta 70:360–364
Qu Q, Fu L, Zhan X, Samuelis D, Maier J, Li L, Tian S, Li Z, Wu Y (2011) Energy Environ Sci 4:3985
Tang W, Liu LL, Tian S, Li L, Li LL, Yue YB, Bai Y, Wu YP, Zhu K, Holze R (2011) Electrochem Commun 13:1159–1162
Ellis BL, Nazar LF (2012) Curr Opin Solid State Mater Sci 16:168–177
Chevrier VL, Ceder G (2011) J Electrochem Soc 158:A1011–A1014
Ellis BL, Makahnouk WRM, Makimura Y, Toghill K, Nazar LF (2007) Nat Mater 6:749–753
Kim S-W, Seo D-H, Ma X, Ceder G, Kang K (2012) Adv Energy Mater 2:710–721
Palomares V, Serras P, Villaluenga I, Hueso KB, Carretero-González J, Rojo T (2012) Energy Environ Sci 5:5884–5901
Tarascon J-M, Armand M (2001) Nature 414:359–367
Parant J-P, Olazcuaga R, Devalette M, Fouassier C, Hagenmuller P (1971) J Solid State Chem 3:1–11
Delmas C, Fouassier C, Hagenmuller P (1980) Physica B + C 99:81–85
Doeff MM, Anapolsky A, Edman L, Richardson TJ, De Jonghe LC (2001) J Electrochem Soc 148:A230–A236
Eriksson TA, Lee YJ, Hollingsworth J, Reimer JA, Cairns EJ, Zhang X, Doeff MM (2003) Chem Mater 15:4456–4463
Mendiboure A, Delmas C, Hagenmuller P (1985) J Solid State Chem 57:323–331
Caballero A, Hernán L, Morales J, Sánchez L, Santos Peña J, Aranda MAG (2002) J Mater Chem 12:1142–1147
Shao-Horn Y (1999) J Electrochem Soc 146:2404–2412
Ma X, Chen H, Ceder G (2011) J Electrochem Soc 158:A1307–A1312
Kim H, Kim D, Seo D, Yeom M, Kang K, Kim DK, Jung Y (2012) Chem Mater 24:1205–1211
Tarascon J, Guyomard D, Wilkens B (1992) Solid State Ionics 57:113–120
Sauvage F, Laffont L, Tarascon J-M, Baudrin E (2007) Inorg Chem 46:3289–3294
Yang S, Wang X, Wang Y, Chen Q, Li J, Yang X (2010) T Nonferr Metal Soc 20:1892–1898
Chick LA, Pederson LR, Maupin GD, Bates JL, Thomas LE, Exarhos GJ (1990) Mater Lett 10:6–12
Berthelot R, Carlier D, Delmas C (2011) Nat Mater 10:74–80
Carlier D, Cheng JH, Berthelot R, Guignard M, Yoncheva M, Stoyanova R, Hwang BJ, Delmas C (2011) Dalton Trans 40:9306–9312
Dollé M, Hollingsworth J, Richardson TJ, Doeff MM (2004) Solid State Ionics 175:225–228
Dollé M, Patoux S, Doeff MM (2005) Chem Mater 17:1036–1043
Rietveld HM (1969) J Appl Crystallogr 2:65–71
Rietveld HM (1967) Acta Crystallogr 22:151–152
Cheary RW, Coelho A (1992) J Appl Crystallogr 25:109–121
Lu Z, Donaberger RA, Dahn JR (2000) Chem Mater 12:3583–3590
Shu G, Prodi A, Chu S, Lee Y, Sheu H, Chou F (2007) Phys Rev B Condens Matter 76:184115
Meng YS, Hinuma Y, Ceder G (2008) J Chem Phys 128:104708
Yabuuchi N, Kajiyama M, Iwatate J, Nishikawa H, Hitomi S, Okuyama R, Usui R, Yamada Y, Komaba S (2012) Nat Mater 11:512–517
Huang Q, Foo ML, Lynn JW, Zandbergen HW, Lawes G, Wang Y, Toby BH, Ramirez AP, Ong NP, Cava RJ (2004) J Phys Condens Matter 16:5803–5814
Zandbergen H, Foo M, Xu Q, Kumar V, Cava R (2004) Phys Rev B Condens Matter 70:024101
Zhang P, Capaz R, Cohen M, Louie S (2005) Phys Rev B Condens Matter 71:153102
Robertson AD, Armstrong AR, Bruce PG (2001) Chem Mater 13:2380–2386
Qu QT, Shi Y, Tian S, Chen YH, Wu YP, Holze R (2009) J Power Sources 194:1222–1225
Wohlfahrt-Mehrens M, Butz A, Oesten R, Arnold G, Hemmer RP, Huggins RA (1997) J Power Sources 68:582–585
Robertson AD, Armstrong AR, Paterson AJ, Duncan MJ, Bruce PG (2003) J Mater Chem 13:2367–2373
Acknowledgments
The authors would like to thank Miss Han-Yi Chen, Miss Yin Ting Teng, and Mr. Jan Geder for their valuable help in conducting SEM, XRD, and BET measurements. This work was financially supported by the Singapore National Research Foundation under its Campus for Research Excellence and Technological Enterprise (CREATE) programme.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Nicolas Bucher and Steffen Hartung contributed equally to this manuscript.
Dedicated to Prof. Wolf Vielstich on the occasion of his 90th birthday and in recognition of his contributions to electrochemistry.
Rights and permissions
About this article
Cite this article
Bucher, N., Hartung, S., Gocheva, I. et al. Combustion-synthesized sodium manganese (cobalt) oxides as cathodes for sodium ion batteries. J Solid State Electrochem 17, 1923–1929 (2013). https://doi.org/10.1007/s10008-013-2047-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-013-2047-x