Abstract
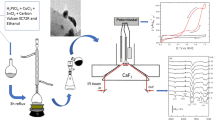
The role of surfactants, cetyl trimethyl ammonium bromide (CTAB), sodium dodecyl sulphate (SDS) and Triton X-100 (in the catalyst), on methanol oxidation at commercial 50:50 Pt–Ru/C catalyst-coated glassy carbon has been studied using cyclic voltammetry, scanning electron microscopy (SEM) and Fourier transform infrared spectroscopy (FT-IR). Surfactant containing catalysts showed a considerable reduction in the methanol oxidation potential. In terms of oxidation potential, better results (lower methanol oxidation potential) were observed in the order SDS > Triton X-100 > CTAB > no surfactant. SEM studies on the catalyst ink showed better homogeneity in the sample prepared using surfactant. This indicates better Pt Pt contact, which is likely to favour methanol adsorption and its oxidation. Hence, lowering of oxidation potentials for methanol oxidation could be seen with use of surfactants. Results of FT-IR on the catalyst ink showed definite changes in the frequencies in the case of Pt–Ru/C containing surfactants indicating definite interaction between catalyst and surfactant. Catalysts, with and without surfactants, yielded linear plots of concentration vs peak currents for methanol oxidation (0–2 M). With surfactant containing catalysts, reduction in methanol oxidation current was observed, and the order followed was the reverse of the above.




Similar content being viewed by others
References
Li X, Hsing IM (2006) Electrochim Acta 52:1358. doi:10.1016/j.electacta.2006.07.037
Xiong L, Manthiram A (2005) Solid State Ion 176:385. doi:10.1016/j.ssi.2004.08.005
Sen F, Gokagac G (2007) J Phys Chem C 111:1467. doi:10.1021/jp065809y
Gullon JS, Iglesias FJV, Montiel V, Aldaz A (2004) Electrochim Acta 49:5079. doi:10.1016/j.electacta.2004.06.017
Rojas S, Garcıa FJG, Jaras S, Huerta MVM, Fierro JLG, Boutonnet M (2005) Appl Catal A 285:24. doi:10.1016/j.apcata.2005.02.005
Liu Z, Lee JY, Han M, Chenc W, Gana LM (2002) J Mater Chem 12:2453. doi:10.1039/b200875k
Bonnmann H, Brijoux W, Brinkmann R, Dinjus E, Fretzen R, Joussen T, Koppler B, Korall B, Neiteler P, Richter J (1994) J Mol Catal 86:129. doi:10.1016/0304-5102(93)E0148-A
Bonnemann H, Braun G, Brijoux W, Brinkmann R, Tilling S, Seevogel K, Siepen K (1996) J Organomet Chem 520:143. doi:10.1016/0022-328X(96)06273-0
Bonnemann H, Britz P (1998) Langmuir 14:6654. doi:10.1021/la980459v
Eklund SE, Cliffel DE (2004) Langmuir 20:6012. doi:10.1021/la049787n
Zhou JH, He JP, Ji YJ, Dang WJ, Liu XL, Zhao GW, Zhang CX, Zhao JS, Fu QB, Hu HP (2007) Electrochim Acta 52:4691
Bauer A, Gyenge EL, Oloman CW (2006) Electrochim Acta 51:5356. doi:10.1016/j.electacta.2006.02.006
Su Y (2006) Electrochim Acta 51:4316. doi:10.1016/j.electacta.2005.12.032
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karthikeyan, N., Giridhar, V.V. & Vasudevan, D. Surfactant effects on methanol oxidation at Pt–Ru/C coated glassy carbon electrode. J Solid State Electrochem 14, 877–881 (2010). https://doi.org/10.1007/s10008-009-0867-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-009-0867-5