Abstract
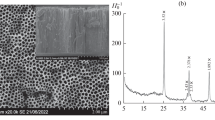
Titanium oxide nanotube electrodes were successfully prepared by anodic oxidation on pure Ti sheets in 0.5 wt.% NH4F + 1 wt.% (NH4)2SO4 + 90 wt.% glycerol mixed solutions. Nanotubes with diameter 40–60 nm and length 7.4 μm were observed by field emission scanning electron microscope. The electrochemical and photoelectrochemical characteristics of TiO2 nanotube electrode were investigated using linear polarization and electrochemical impedance spectroscopy techniques. The open-circuit potential dropped markedly under irradiation and with the increase of Cl− concentrations. A saturated photocurrent of approximately 1.3 mA cm−2 was observed under 10-W low-mercury lamp irradiation in 0.1 M NaCl solution, which was much higher than film electrode. Meanwhile, the highest photocurrent in NaCl solution implied that the photogenerated holes preferred to combine with Cl−. Thus, a significant synergetic effect on active chlorine production was observed in photoelectrocatalytic processes. Furthermore, the generation efficiency for active chlorine was about two times that using TiO2/Ti film electrode by sol–gel method. Finally, the effects of initial pH and Cl− concentration were also discussed.








Similar content being viewed by others
References
Fujishima A, Honda K (1972) Nature 238:37. doi:10.1038/238037a0
Zhao X, Zhu Y (2006) Environ Sci Technol 40:3367. doi:10.1021/es052029e
Li XZ, Liu HL, Yue PT (2000) Environ Sci Technol 34:4401. doi:10.1021/es000939k
Osugi ME, Umbuzeiro GA, Anderson MA, Zanoni MVB (2005) Electrochim Acta 50:5261. doi:10.1016/j.electacta.2005.01.058
Carneiro PA, Osugi ME, Sene JJ, Anderson MA, Zanoni MVB (2004) Electrochim Acta 49:3807. doi:10.1016/j.electacta.2003.12.057
Egerton TA, Janus M, Morawski AW (2006) Chemosphere 63:1203. doi:10.1016/j.chemosphere.2005.08.074
Li J, Li L, Zheng L, Xian Y, Jin L (2006) Electrochim Acta 51:4942. doi:10.1016/j.electacta.2006.01.037
Li G, Qu J, Zhang X, Ge J (2006) Water Res 40:213. doi:10.1016/j.watres.2005.10.039
He C, Li XZ, Graham N, Wang Y (2006) Appl Catal A 305:54. doi:10.1016/j.apcata.2006.02.051
Zwilling V, Aucouturier M, Darque-Ceretti E (1999) Electrochim Acta 45:921. doi:10.1016/S0013-4686(99)00283-2
Macák JM, Tsuchiya H, Schmuki P (2005) Angew Chem Int Ed 44:2100. doi:10.1002/anie.200462459
Xie Y (2006) Electrochim Acta 51:3399. doi:10.1016/j.electacta.2005.10.003
Macak JM, Sirotna K, Schmuki P (2005) Electrochim Acta 50:3679. doi:10.1016/j.electacta.2005.01.014
Macak JM, Tsuchiya H, Taveira L, Aldabergerova S, Schmuki P (2005) Angew Chem Int Ed 44:7463. doi:10.1002/anie.200502781
Bauer S, Kleber S, Schmuki P (2006) Electrochem Commun 8:1321. doi:10.1016/j.elecom.2006.05.030
Mor GK, Varghese OK, Paulose M, Grimes CA (2005) Adv Funct Mater 15:1291. doi:10.1002/adfm.200500096
Quan X, Yang S, Ruan X, Zhao H (2005) Environ Sci Technol 39:3370. doi:10.1021/es048684o
APHA AWWA, WEF (2005) Standard methods for the examination of water and wastewater, 21st edn. American Public Health Association, American Water Works Association, Water Environment Federation, Washington, DC
Pankove LI (1970) Optical processes in semiconductors. Dover, New York
Li MC, Shen JN (2006) J Solid State Electrochem 10:980. doi:10.1007/s10008-005-0043-5
Qu J, Zhao X (2008) Environ Sci Technol 42:4934. doi:10.1021/es702769p
Liu H, Cheng S, Wu M, Wu H, Zhang J, Li W, Cao C (2000) J Phys Chem A 104:7016. doi:10.1021/jp000171q
Wu X, Ma H, Chen S, Xu Z, Sui A (1999) J Electrochem Soc 146:1847. doi:10.1149/1.1391854
Ohtsuka T, Otsuki T (1999) J Electroanal Chem 473:272–278. doi:10.1016/S0022-0728(99)00238-7
Selcuk H, Anderson MA (2005) Desalination 176:219. doi:10.1016/j.desal.2004.10.016
Zhao X, Liu M, Zhu Y (2007) Thin Solid Films 515:7127. doi:10.1016/j.tsf.2007.03.025
Acknowledgments
This work was supported by the Funds for Creative Research Groups of People’s Republic of China (no. 50621804) and the National Natural Science Foundation of China (no. 50538090).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiao, S., Qu, J., Liu, H. et al. Fabrication of TiO2/Ti nanotube electrode and the photoelectrochemical behaviors in NaCl solutions. J Solid State Electrochem 13, 1959–1964 (2009). https://doi.org/10.1007/s10008-008-0734-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-008-0734-9