Abstract
Context
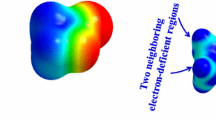
In guest–host interactions, guest molecules used for hydrogen bonding potential of clathrate hydrate cages are involved as “promoters” for hydrogen or methane storage. Among the promoter molecules, there are pinacolone and tert-butylmethyl ether containing oxygen atom, and tert-butyl amine among those containing nitrogen atom. The dimer and trimer interactions of these molecules with the water clusters were investigated. The cooperativity effect, which is a measure of the hydrogen bonding of these oxygen and nitrogen-containing complexes, was calculated and these complexes were analyzed. Moreover, molecular electrostatic potential (MEP) analysis was performed to determine intermolecular interactions. Clusters of several molecules may seem like a non-significant state of matter; however, in systematic studies with small clusters, important results can be obtained. Therefore, it is very useful to work with clusters to understand the properties of the dense phase, and this study aimed to examine the properties of structures, energies, infrared vibration frequencies, and topological parameters, which develop as a result of the interaction of structures in different clusters with water molecules.
Methods
The MP2 level was performed using aug-cc-pVDZ basis set calculations for this study. Tight converting criteria were used in the optimization step. Harmonic vibrational modes were calculated at the MP2/aug-cc-pVDZ level. Each minima obtained from the MP2 level was subjected to single-point energy calculations using CCSD(T) levels with the aug-cc-pVDZ basis set by the Gaussian16 package program suite. The topological analyses were performed with non-covalent interaction (NCI). MEP surface of the complex was also composed by Gaussian program using the optimized geometry at a MP2/aug-cc-pvdz level.









Similar content being viewed by others
Data availability
All generated or analyzed data during this study are included in the article.
References
Hawkins R, Davidson D (1966) Dielectric relaxation in the clathrate hydrates of some cyclic ethers. J Phys Chem 70:1889–1894. https://doi.org/10.1021/j100878a033
Buch V, Devlin JP, Monreal IA, Jagoda-Cwiklik B, Uras-Aytemiz N, Cwiklik L (2009) Clathrate hydrates with hydrogen-bonding guests. Phys Chem Chem Phys 11:10245–10265. https://doi.org/10.1039/B911600C
Alavi S, Udachin K, Ripmeester JA (2010) Effect of guest–host hydrogen bonding on the structures and properties of clathrate hydrates. Chem Eur J 16:1017–1025. https://doi.org/10.1002/chem.200902351
Scheiner S (1997) Hydrogen bonding: a theoretical perspective. Oxford University Press, New York
Hobza P, Havlas Z (2000) Blue-Shifting hydrogen bonds. Chem Rev 100:4253–4264. https://doi.org/10.1021/cr990050q
Grabowski SJ (2011) What is the covalency of hydrogen bonding? Chem Rev 111:2597–2625. https://doi.org/10.1021/cr800346f
Steiner T (2002) The hydrogen bond in the solid state. Angew Chem Int Ed Engl 41:48–76. https://doi.org/10.1002/1521-3773(20020104)41:1%3c48::AID-ANIE48%3e3.0.CO;2-U
Mahadevi AS, Sastry GN (2016) Cooperativity in noncovalent interactions. Chem Rev 116:2775–2825. https://doi.org/10.1021/cr500344e
Susilo R, Alavi S, Moudrakovski IL, Englezos P, Ripmeester JA (2009) Guest-Host hydrogen bonding in structure H clathrate hydrates. Chem Phys Chem 10:824–829. https://doi.org/10.1002/cphc.200900024
Makiya T, Murakami T, Takeya S, Sum AK, Alavi S, Ohmura R (2010) Synthesis and characterization of clathrate hydrates containing carbon dioxide and ethanol. Phys Chem Chem Phys 12:9927–9932. https://doi.org/10.1039/C002187C
Alavi S, Susilo R, Ripmeester JA (2009) Linking microscopic guest properties to macroscopic observables in clathrate hydrates: guest-host hydrogen bonding. J Chem Phys 130:174501. https://doi.org/10.1063/1.3124187
Alavi S, Takeya S, Ohmura R, Woo TK, Ripmeester JA (2010) Hydrogen-bonding alcohol-water interactions in binary ethanol, 1-propanol, and 2-propanol+methane structure II clathrate hydrates. J Chem Phys 133:074505. https://doi.org/10.1063/1.3469776
Allen PS, Khanzada AWK, McDowell CA (1973) Nuclear magnetic resonance absorption and relaxation study of tert -butylamine and tert -butylamine clathrate deuterate. J Chem Phys 59:470. https://doi.org/10.1063/1.1679831
Bjerrum N (1952) Structure and properties of ice. Sci 115:385–390. https://doi.org/10.1126/science.115.2989.385
Li Z, Singh S (2008) FTIR and ab initio investigations of the MTBE-water complex. J Phys Chem A 112:8593–8599. https://doi.org/10.1021/jp804246b
Di Palma TM, Bende A (2013) Vacuum ultraviolet photoionization and ab initio investigations of methyl tert-butyl ether (MTBE) clusters and MTBE–water clusters. Chem Phys Lett 561–562:18–23. https://doi.org/10.1016/j.cplett.2013.01.025
Johnson ER, Keinan S, Mori-Sánchez P, Contreras-García J, Cohen AJ, Yang W (2010) Revealing noncovalent interactions. J Am Chem Soc 132:6498–6506. https://doi.org/10.1021/ja100936w
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, et al. (2016) Gaussian 16, Revision C.01, Gaussian Inc. Wallingford CT
Boys SF, Bernardi F (1970) The calculation of small molecular interactions by the differences of separate total energies. Some procedures with reduced errors. Mol Phys 19:553–566. https://doi.org/10.1080/00268977000101561
Contreras-García J, Johnson ER, Keinan S, Chaudret R, Piquemal JP, Beratan DN, Yang W (2011) NCIPLOT: a program for plotting non-covalent interaction regions. J Chem Theory Comput 7:625–632. https://doi.org/10.1021/ct100641a
Andrezejewska A, Sadlej J (2004) Ab initio study on mixed methanol–hydrogen chloride dimer and trimers. Chem Phys Lett 393:228–235. https://doi.org/10.1016/j.cplett.2004.06.035
Cabaleiro-Lago EM, Rodriguez-Otero J (2002) Methanethiol dimer and trimer. An ab initio and DFT study of the interaction. J Phys Chem A 106:7440. https://doi.org/10.1021/jp021001b
Murray JS, Sen K (1996) Molecular electrostatic potentials: concepts and applications. Elsevier, Amsterdam, pp 105–538
Luque FJ, López JM, Orozco M (2000) Perspective on “Electrostatic interactions of a solute with a continuum. A direct utilization of ab initio molecular potentials for the prevision of solvent effects.” Theor Chem Acc 103:343–345. https://doi.org/10.1007/s002149900013
Acknowledgements
The numerical calculations in this article were partially performed at TUBITAK ULAKBIM, High Performance and Grid Computing Center (TRUBA resources).
Author information
Authors and Affiliations
Contributions
Zafer MAŞLAKCI has prepared every stage of the article himself, including data collection and analysis, review, methodology, verification, visualization, formal analysis, and writing—review and editing.
Corresponding author
Ethics declarations
Competing interests
The author declares no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maşlakcı, Z. Hydrogen bonding guest-water interactions in pinacolone, tert-butyl amine, and tert-butylmethyl ether: a theoretical study on energetics, structure, and topological + . J Mol Model 29, 110 (2023). https://doi.org/10.1007/s00894-023-05519-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-023-05519-2