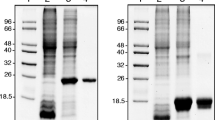
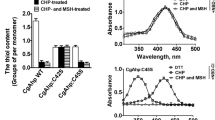
Abstract
Hydrogen peroxide (H2O2) produces hydroxyl radicals that directly attack a variety of biomolecules and cause severe cellular dysfunction. An extremely thermophilic bacterium, Thermus thermophilus HB8, possesses at least three enzymes that can scavenge H2O2: manganese-containing catalase (TTHA0122, MnCAT), a possible peroxiredoxin homologue (TTHA1300), and a possible heme peroxidase (HPX) homologue (TTHA1714). To investigate the roles of these proteins, we attempted to disrupt each of these genes in T. thermophilus HB8. Although we were able to completely disrupt ttha1300, we were unable to completely delete ttha0122 and ttha1714 because of polyploidy. Quantitative real-time PCR showed that, compared to the wild type, 31 % of ttha0122 and 11 % of ttha1714 remained in the ∆ttha0122 and ∆ttha1714 disruption mutants, respectively. Mutants with reduced levels of ttha0122 or ttha1714 exhibited a significant increase in spontaneous mutation frequency. ∆ttha1714 grew slower than the wild type under normal conditions. ∆ttha0122 grew very poorly after exposure to H2O2. Moreover, ∆ttha0122 did not show H2O2-scavenging activity, whereas ∆ttha1300 and ∆ttha1714 scavenged H2O2, a property similar to that exhibited by the wild type. MnCAT purified from T. thermophilus HB8 cells scavenged H2O2 in vitro. The recombinant form of the possible HPX homologue, reconstituted with hemin, showed peroxidase activity with H2O2 as an oxidant substrate. Based on these results, we propose that not only MnCAT but also the possible HPX homologue is involved in protecting the cell from oxidative stress in T. thermophilus.






Similar content being viewed by others
Abbreviations
- ROS:
-
Reactive oxygen species
- MnCAT:
-
Manganese-containing catalase
- HPX:
-
Heme peroxidase
- HTK :
-
Thermostable kanamycin-resistance gene
- HRP:
-
Horseradish peroxidase
- ABTS:
-
2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid)
- MES:
-
2-morpholinoethanesulfonic acid
- MOPS:
-
3-(N-morpholino)propanesulfonic acid
- SDS-PAGE:
-
SDS-polyacrylamide gel electrophoresis
- MALDI-TOF MS:
-
Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry
References
Agari Y, Kashihara A, Yokoyama S, Kuramitsu S, Shinkai A (2008) Global gene expression mediated by Thermus thermophilus SdrP, a CRP/FNR family transcriptional regulator. Mol Microbiol 70:60–75. doi:10.1111/j.1365-2958.2008.06388.x
Agari Y, Kuramitsu S, Shinkai A (2010) Identification of novel genes regulated by the oxidative stress-responsive transcriptional activator SdrP in Thermus thermophilus HB8. FEMS Microbiol Lett 313:127–134. doi:10.1111/j.1574-6968.2010.02133.x
Agari Y, Agari K, Sakamoto K, Kuramitsu S, Shinkai A (2011) TetR-family transcriptional repressor Thermus thermophilus FadR controls fatty acid degradation. Microbiology 157:1589–1601. doi:10.1099/mic.0.048017-0
Beers RF Jr, Sizer IW (1952) A spectrophotometric method for measuring the breakdown of hydrogen peroxide by catalase. J Biol Chem 195:133–140
Berry EA, Trumpower BL (1987) Simultaneous determination of hemes a, b, and c from pyridine hemochrome spectra. Anal Biochem 161:1–15
Bonny C, Montandon PE, Marc-Martin S, Stutz E (1991) Analysis of streptomycin-resistance of Escherichia coli mutants. Biochim Biophys Acta 1089:213–219
Cao Z, Mueller CW, Julin DA (2010) Analysis of the recJ gene and protein from Deinococcus radiodurans. DNA Repair (Amst) 9:66–75. doi:10.1016/j.dnarep.2009.10.009
Chelikani P, Fita I, Loewen PC (2004) Diversity of structures and properties among catalases. Cell Mol Life Sci 61:192–208. doi:10.1007/s00018-003-3206-5
Ebihara A et al (2005) Structure-based functional identification of a novel heme-binding protein from Thermus thermophilus HB8. J Struct Funct Genom 6:21–32. doi:10.1007/s10969-005-1103-x
Fukui K, Wakamatsu T, Agari Y, Masui R, Kuramitsu S (2011) Inactivation of the DNA repair genes mutS, mutL or the anti-recombination gene mutS2 leads to activation of vitamin B1 biosynthesis genes. PLoS ONE 6:e19053. doi:10.1371/journal.pone.0019053
Gay C, Collins J, Gebicki JM (1999) Hydroperoxide assay with the ferric-xylenol orange complex. Anal Biochem 273:149–155. doi:10.1006/abio.1999.4208
Giorgio M, Trinei M, Migliaccio E, Pelicci PG (2007) Hydrogen peroxide: a metabolic by-product or a common mediator of ageing signals? Nat Rev Mol Cell Biol 8:722–728
Hansen MT (1978) Multiplicity of genome equivalents in the radiation-resistant bacterium Micrococcus radiodurans. J Bacteriol 134:71–75
Hashimoto Y, Yano T, Kuramitsu S, Kagamiyama H (2001) Disruption of Thermus thermophilus genes by homologous recombination using a thermostable kanamycin-resistant marker. FEBS Lett 506:231–234
Hoseki J, Yano T, Koyama Y, Kuramitsu S, Kagamiyama H (1999) Directed evolution of thermostable kanamycin-resistance gene: a convenient selection marker for Thermus thermophilus. J Biochem 126:951–956
Imlay JA (2003) Pathways of oxidative damage. Annu Rev Microbiol 57:395–418
Imlay JA (2008) Cellular defenses against superoxide and hydrogen peroxide. Annu Rev Biochem 77:755–776. doi:10.1146/annurev.biochem.77.061606.161055
Kuramitsu S, Hiromi K, Hayashi H, Morino Y, Kagamiyama H (1990) Pre-steady-state kinetics of Escherichia coli aspartate aminotransferase catalyzed reactions and thermodynamic aspects of its substrate specificity. Biochemistry 29:5469–5476
Luria SE, Delbruck M (1943) Mutations of bacteria from virus sensitivity to virus resistance. Genetics 28:491–511
Matsui T, Ozaki S, Liong E, Phillips GN Jr, Watanabe Y (1999) Effects of the location of distal histidine in the reaction of myoglobin with hydrogen peroxide. J Biol Chem 274:2838–2844
Mishra S, Imlay J (2012) Why do bacteria use so many enzymes to scavenge hydrogen peroxide? Arch Biochem Biophys 525:145–160. doi:10.1016/j.abb.2012.04.014
Moreno R, Hidalgo A, Cava F, Fernandez-Lafuente R, Guisan JM, Berenguer J (2004) Use of an antisense RNA strategy to investigate the functional significance of Mn-catalase in the extreme thermophile Thermus thermophilus. J Bacteriol 186:7804–7806. doi:10.1128/JB.186.22.7804-7806.2004
Morita R et al (2010) Molecular mechanisms of the whole DNA repair system: a comparison of bacterial and eukaryotic systems. J Nucleic Acids 2010:179594. doi:10.4061/2010/179594
Nakane S, Wakamatsu T, Masui R, Kuramitsu S, Fukui K (2011) In vivo, in vitro, and X-ray crystallographic analyses suggest the involvement of an uncharacterized triose-phosphate isomerase (TIM) barrel protein in protection against oxidative stress. J Biol Chem 286:41636–41646. doi:10.1074/jbc.M111.293886
Ohtani N, Tomita M, Itaya M (2010) An extreme thermophile, Thermus thermophilus, is a polyploid bacterium. J Bacteriol 192:5499–5505. doi:10.1128/JB.00662-10
Omelchenko MV et al (2005) Comparative genomics of Thermus thermophilus and Deinococcus radiodurans: divergent routes of adaptation to thermophily and radiation resistance. BMC Evol Biol 5:57. doi:10.1186/1471-2148-5-57
Oshima T, Imahori K (1974) Description of Thermus thermophilus (Yoshida and Oshima) comb. nov., a nonsporulating thermophilic bacterium from a Japanese thermal spa. Int J Syst Bacteriol 24:102–112
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Seaver LC, Imlay JA (2001a) Alkyl hydroperoxide reductase is the primary scavenger of endogenous hydrogen peroxide in Escherichia coli. J Bacteriol 183:7173–7181. doi:10.1128/JB.183.24.7173-7181.2001
Seaver LC, Imlay JA (2001b) Hydrogen peroxide fluxes and compartmentalization inside growing Escherichia coli. J Bacteriol 183:7182–7189. doi:10.1128/JB.183.24.7182-7189.2001
Shank M, Barynin V, Dismukes GC (1994) Protein coordination to manganese determines the high catalytic rate of dimanganese catalases. Comparison to functional catalase mimics. Biochemistry 33:15433–15436
Shimada A, Masui R, Nakagawa N, Takahata Y, Kim K, Kuramitsu S, Fukui K (2010) A novel single-stranded DNA-specific 3ʹ-5ʹ exonuclease, Thermus thermophilus exonuclease I, is involved in several DNA repair pathways. Nucleic Acids Res 38:5692–5705. doi:10.1093/nar/gkq350
Smith PK et al (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150:76–85
Sugano Y (2009) DyP-type peroxidases comprise a novel heme peroxidase family. Cell Mol Life Sci 66:1387–1403. doi:10.1007/s00018-008-8651-8
Wallace SS (2002) Biological consequences of free radical-damaged DNA bases. Free Radic Biol Med 33:1–14
Whittaker JW (2012) Non-heme manganese catalase–the ‘other’ catalase. Arch Biochem Biophys 525:111–120. doi:10.1016/j.abb.2011.12.008
Wood ZA, Schroder E, Robin Harris J, Poole LB (2003) Structure, mechanism and regulation of peroxiredoxins. Trends Biochem Sci 28:32–40
Yokoyama S et al (2000) Structural genomics projects in Japan. Nat Struct Biol 7(Suppl):943–945
Zubieta C et al (2007a) Identification and structural characterization of heme binding in a novel dye-decolorizing peroxidase, TyrA. Proteins 69:234–243. doi:10.1002/prot.21673
Zubieta C et al (2007b) Crystal structures of two novel dye-decolorizing peroxidases reveal a beta-barrel fold with a conserved heme-binding motif. Proteins 69:223–233. doi:10.1002/prot.21550
Acknowledgments
We thank all the members of SR System Biology Research Group, RIKEN SPring-8, Harima Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by S. Albers.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ebihara, A., Manzoku, M., Fukui, K. et al. Roles of Mn-catalase and a possible heme peroxidase homologue in protection from oxidative stress in Thermus thermophilus . Extremophiles 19, 775–785 (2015). https://doi.org/10.1007/s00792-015-0753-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-015-0753-2