Abstract
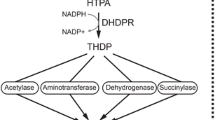
Dihydrodipicolinate synthase (DHDPS) catalyses the first reaction of the (S)-lysine biosynthesis pathway in bacteria and plants. The hypothetical gene for dihydrodipicolinate synthase (dapA) of Thermoanaerobacter tengcongensis was found in a cluster containing several genes of the diaminopimelate lysine–synthesis pathway. The dapA gene was cloned in Escherichia coli, DHDPS was subsequently produced and purified to homogeneity. The T. tengcongensis DHDPS was found to be thermostable (T 0.5 = 3 h at 90°C). The specific condensation of pyruvate and (S)-aspartate-β -semialdehyde was catalyzed optimally at 80°C at pH 8.0. Enzyme kinetics were determined at 60°C, as close as possible to in vivo conditions. The established kinetic parameters were in the same range as for example E. coli dihydrodipicolinate synthase. The specific activity of the T. tengcongensis DHDPS was relatively high even at 30°C. Like most dihydrodipicolinate synthases known at present, the DHDPS of T. tengcongensis seems to be a tetramer. A structural model reveals that the active site is well conserved. The binding site of the allosteric inhibitor lysine appears not to be conserved, which agrees with the fact that the DHDPS of T. tengcongensis is not inhibited by lysine under physiological conditions.






Similar content being viewed by others
Abbreviations
- DAP:
-
Diaminopimelate
- DIBAL:
-
Diisobutylaluminium hydride
- DHDP:
-
Dihydrodipicolinate
- DHDPR:
-
Dihydrodipicolinate reductase
- DHDPS:
-
Dihydrodipicolinate synthase
- HTPA:
-
(4S)-4–hydroxy-2,3,4,5-tetrahydro-(2S)-dipicolinic acid
- NAL:
-
N-acetylneuraminate lyase
- (S)-ASA:
-
(S)-aspartate-β-semialdehyde
- TCA:
-
Trichloroacetic acid
References
Blagova E, Levdikov V, Milioti N, Fogg MJ, Kalliomaa AK, Brannigan JA, Wilson KS, Wilkinson AJ (2006) Crystal structure of dihydrodipicolinate synthase (BA3935) from Bacillus anthracis at 1.94 A resolution. Proteins 62:297–301
Blickling S, Beisel HG, Bozic D, Knablein J, Laber B, Huber R (1997a) Structure of dihydrodipicolinate synthase of Nicotiana sylvestris reveals novel quaternary structure. J Mol Biol 274:608–621
Blickling S, Renner C, Laber B, Pohlenz HD, Holak TA, Huber R (1997b) Reaction mechanism of Escherichia coli dihydrodipicolinate synthase investigated by X-ray crystallography and NMR spectroscopy. Biochemistry 36:24–33
Cahyanto MN, Kawasaki H, Nagashio M, Fujiyama K, Seki T (2006) Regulation of aspartokinase, aspartate semialdehyde dehydrogenase, dihydrodipicolinate synthase and dihydrodipicolinate reductase in Lactobacillus plantarum. Microbiology 152:105–112
Choi KH, Lai V, Foster CE, Morris AJ, Tolan DR, Allen KN (2006) New superfamily members identified for Schiff-base enzymes based on verification of catalytically essential residues. Biochemistry 45:8546–8555
Coulter CV, Gerrard JA, Kraunsoe JAE, Moore DJ, Pratt AJ (1996) (S)-Aspartate semi-aldehyde: synthetic and structural studies. Tetrahedron 52:7127–7136
Cremer J, Treptow C, Eggeling L, Sahm H (1988) Regulation of enzymes of lysine biosynthesis in Corynebacterium glutamicum. J Gen Microbiol 134:3221–3229
Dereppe C, Bold G, Ghisalba O, Ebert E, Schar HP (1992) Purification and characterization of dihydrodipicolinate synthase from pea. Plant Physiol 98:813–821
Dobson RC, Gerrard JA, Pearce FG (2004a) Dihydrodipicolinate synthase is not inhibited by its substrate, (S)-aspartate beta-semialdehyde. Biochem J 377:757–762
Dobson RC, Valegard K, Gerrard JA (2004b) The crystal structure of three site-directed mutants of Escherichia coli dihydrodipicolinate synthase: further evidence for a catalytic triad. J Mol Biol 338:329–339
Dobson RC, Griffin MD, Jameson GB, Gerrard JA (2005) The crystal structures of native and (S)-lysine-bound dihydrodipicolinate synthase from Escherichia coli with improved resolution show new features of biological significance. Acta crystallogr Sect D 61:1116–1124
Frisch DA, Gengenbach BG, Tommey AM, Sellner JM, Somers DA, Myers DE (1991) Isolation and characterization of dihydrodipicolinate synthase from maize. Plant Physiol 96:444–452
Ghislain M, Frankard V, Jacobs M (1995) A dinucleotide mutation in dihydrodipicolinate synthase of Nicotiana sylvestris leads to lysine overproduction. Plant J 8:733–743
Grazi E, Rowley PT, Cheng T, Tchola O, Horecker BL (1962) The mechanism of action of aldolases. III. Schiff base formation with lysine. Biochem Biophys Res Commun 9:38–43
Hoganson DA, Stahly DP (1975) Regulation of dihydrodipicolinate synthase during growth and sporulation of Bacillus cereus. J Bacteriol 124:1344–1350
Horecker BL, Kornberg A (1948) The extinction coefficients of the reduced band of pyridin nucleotides. J Biol Chem 175:385–390
Karsten WE (1997) Dihydrodipicolinate synthase from Escherichia coli: pH dependent changes in the kinetic mechanism and kinetic mechanism of allosteric inhibition by l-lysine. Biochemistry 36:1730–1739
Kumpaisal R, Hashimoto T, Yamada Y (1987) Purification and characterization of dihydrodipicolinate synthase from wheat suspension cultures. Plant Physiol 85:145–151
Laber B, Gomis-Ruth FX, Romao MJ, Huber R (1992) Escherichia coli dihydrodipicolinate synthase. Identification of the active site and crystallization. Biochem J 288(Pt 2):691–695
Massant J, Verstreken P, Durbecq V, Kholti A, Legrain C, Beeckmans S, Cornelis P, Glansdorff N (2002) Metabolic channeling of carbamoyl phosphate, a thermolabile intermediate: evidence for physical interaction between carbamate kinase-like carbamoyl-phosphate synthetase and ornithine carbamoyltransferase from the hyperthermophile Pyrococcus furiosus. J Biol Chem 277:18517–18522
Mazelis M, Whatley FR, Whatley J (1977) The enzymology of lysine biosynthesis in higher plants. The occurrence, characterization and some regulatory properties of dihydrodipicolinate synthase. FEBS Lett 84:236–240
Mirwaldt C, Korndorfer I, Huber R (1995) The crystal structure of dihydrodipicolinate synthase from Escherichia coli at 2.5 A resolution. J Mol Biol 246:227–239
Pearce FG, Perugini MA, McKerchar HJ, Gerrard JA (2006) Dihydrodipicolinate synthase from Thermotoga maritima. Biochem J 400:359–366
Roberts SJ, Morris JC, Dobson RC, Gerrard JA (2003) The preparation of (S)-aspartate semi-aldehyde appropriate for use in biochemical studies. Bioorg Med Chem Lett 13:265–267
Scapin G, Reddy SG, Zheng R, Blanchard JS (1997) Three-dimensional structure of Escherichia coli dihydrodipicolinate reductase in complex with NADH and the inhibitor 2,6-pyridinedicarboxylate. Biochemistry 36:15081–15088
Shedlarski JG, Gilvarg C (1970) The pyruvate-aspartic semialdehyde condensing enzyme of Escherichia coli. J Biol Chem 245:1362–1373
Stahly DP (1969) Dihydrodipicolinic acid synthase of Bacillus licheniformis. Biochim Biophys Acta 191:439–451
Tam PH, Phenix CP, Palmer DR (2004) MosA, a protein implicated in rhizopine biosynthesis in Sinorhizobium meliloti L5-30, is a dihydrodipicolinate synthase. J Mol Biol 335:393–397
Tsujimoto N, Gunji Y, Ogawa-Miyata Y, Shimaoka M, Yasueda H (2006) l-Lysine biosynthetic pathway of Methylophilus methylotrophus and construction of an l-Lysine producer. J Biotechnol 124:327–337
Tudor DW, Lewis T, Robins DJ (1993) Synthesis of the Trifluoroacetate salt of aspartic-acid beta-semialdehyde, an intermediate in the biosynthesis of l-Lysine, l-Threonine, and l-Methionine. Synthesis-Stuttgart: 1061–1062
Vauterin M, Jacobs M (1994) Isolation of a poplar and an Arabidopsis thaliana dihydrodipicolinate synthase cDNA clone. Plant Mol Biol 25:545–550
Vieille C, Hess JM, Kelly RM, Zeikus JG (1995) xylA cloning and sequencing and biochemical characterization of xylose isomerase from Thermotoga neapolitana. Appl Environ Microbiol 61:1867–1875
Wang F, Blanchard JS, Tang XJ (1997) Hydrogen exchange/electrospray ionization mass spectrometry studies of substrate and inhibitor binding and conformational changes of Escherichia coli dihydrodipicolinate reductase. Biochemistry 36:3755–3759
Wernic D, Dimaio J, Adams J (1989) Enantiospecific synthesis of l-alpha-aminosuberic acid—synthetic applications in preparation of atrial natriuretic factor analogs. J Org Chem 54:4224–4228
Wolterink-van Loo S, van Eerde A, Siemerink MA, Akerboom J, Dijkstra BW, van der Oost J (2007) Biochemical and structural exploration of the catalytic capacity of sulfolobus KDG aldolases. Biochem J 403:421–430
Xue Y, Xu Y, Liu Y, Ma Y, Zhou P (2001) Thermoanaerobacter tengcongensis sp. nov., a novel anaerobic, saccharolytic, thermophilic bacterium isolated from a hot spring in Tengcong, China. Int J Syst Evol Microbiol 51:1335–1341
Yugari Y, Gilvarg C (1965) The condensation step in diaminopimelate synthesis. J Biol Chem 240:4710–4716
Ziegenhorn J, Senn M, Bucher T (1976) Molar absorptivities of beta-NADH and beta-NADPH. Clin Chem 22:151–160
Acknowledgments
We want to thank Dr. Gabriella M. Preda (West University of Timisoara) for the synthesis of a batch of (S)-ASA. This research is performed as part of the IBOS Programme (Integration of Biosynthesis & Organic Synthesis) of Advanced Chemical Technologies for Sustainability (ACTS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Antranikian.
Rights and permissions
About this article
Cite this article
Wolterink-van Loo, S., Levisson, M., Cabrières, M.C. et al. Characterization of a thermostable dihydrodipicolinate synthase from Thermoanaerobacter tengcongensis . Extremophiles 12, 461–469 (2008). https://doi.org/10.1007/s00792-008-0152-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-008-0152-z