Abstract
Background and objective
Agenesis of the maxillary lateral incisor occurs in up to 4% of all individuals and requires multidisciplinary treatment. Its developmental origins, however, are not fully understood. Earlier studies documented genetic factors contributing to agenesis but also an association with craniofacial morphology. In this study, we assessed the association between maxillary morphology and lateral incisor agenesis by a geometric morphometric approach to disentangle the roles of developmental plasticity and genetic factors.
Materials and methods
We quantified the maxillary alveolar ridge by 19 two-dimensional landmarks on cross-sectional images of 101 computed tomography scans. We compared the shape and size of the alveolar ridge across patients with unilateral or bilateral agenesis of maxillary lateral incisors and patients with extracted or in situ incisors.
Results
The maxillary alveolar ridge was clearly narrower in patients with agenesis or an extracted incisor compared to the control group, whereas the contralateral side of the unilateral agenesis had an intermediate width. Despite massive individual variation, the ventral curvature of the alveolar ridge was, on average, more pronounced in the bilateral agenesis group compared to unilateral agenesis and tooth extraction.
Conclusions
This suggests that pleiotropic genetic and epigenetic factors influence both tooth development and cranial growth, but an inappropriately sized or shaped alveolar process may also inhibit normal formation or development of the tooth bud, thus leading to dental agenesis.
Clinical relevance
Our results indicate that bilateral agenesis of the lateral incisor tends to be associated with a higher need of bone augmentation prior to implant placement than unilateral agenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
After third molars and lower second premolars, maxillary lateral incisors are the third most common congenitally missing teeth [1, 2] with a reported prevalence of about 4% [3]. Maxillary bone is a complex structure, surrounded by muscular, dental, and skeletal components, which contribute to mastication, speech, and breathing [4]. Missing maxillary lateral incisors often pose a challenge to dentists and patients. There are four main treatment options for maxillary lateral incisor agenesis that typically require a multidisciplinary treatment approach: an implant-supported crown, a tooth-supported restoration, orthodontic space closure, or auto-transplantation [5,6,7,8,9,10]. An implant or transplanted root surface requires full bony coverage and, hence, a sufficiently sized alveolar process in this region. A sound understanding of maxillary form and the required alveolar characteristics are pivotal in the decision for the best treatment choice.
It has been shown that the anterior maxilla is prone to bone loss after tooth extractions in this area [11]. Resorptive activity during postnatal ontogeny in the anterior part of the maxilla has been described by various authors [12, 13]. Previous studies have suggested that the resorptive field within the anterior maxilla increases in size during ontogeny but also tooth extractions can lead to resorptive alveolar ridge remodeling. In an earlier radiographic study on patients with congenitally missing maxillary lateral incisors, it has been shown that mesio-distal gap width negatively correlates with edentulous alveolar ridge dimensions, i.e., an increased gap width was associated with a reduced alveolar ridge dimension [14].
While these studies clearly document an influence of tooth extraction or agenesis on maxillary form, the influence of maxillary size and shape on dental agenesis is less obvious. Various studies indicate that dental agenesis may not be purely genetically determined [15,16,17] because it is also associated with craniofacial morphology [18,19,20,21]. As tooth development proceeds concomitantly with alveolar growth, alveolar dimensions may influence odontogenesis [20, 22]: a too small or inappropriately shaped alveolar process may inhibit normal formation or development of the tooth bud, leading to agenesis of a tooth in this anatomical region. Beyond correlation studies, such an effect of skeletal dimensions on human tooth development cannot be directly assessed, but comparative and experimental studies in animals are in line with this explanation. Evolutionary trends of tooth loss in primates and other mammals show particular patterns and tend to occur in the inverse order as they develop ontogenetically (i.e., the first tooth to be lost is the one that develops last in a certain class of teeth, e.g., lateral incisor, third molar) [23,24,25]. An evolutionary decrease in the number of teeth in primates is also associated with reduced demands on mastication and the size of the jaw [26, 27]. Similarly, it has been suggested that the reduction of the number of teeth in modern humans has resulted from the shortening of the jaws [28,29,30]. Also among modern humans, craniofacial dimensions have been reported to correlate with third molar agenesis [31, 32].
However, correlations between human cranial dimensions and dental agenesis not necessarily result from unidirectional pathways only, i.e., from the effects of dental agenesis on bone remodeling and the effects of bone dimensions on tooth development. Certain genetic and epigenetic factors may additionally influence the development of both the cranium and the teeth. The fact that the major genetic drivers of tooth development, such as Shh and members of the Fgf, Wnt, and Bmp gene families, also determine craniofacial growth seems to support this “pleiotropy hypothesis” [33,34,35]. Such pleiotropic factors may also be reflected by the common co-occurrence of dental agenesis with other dental and craniofacial anomalies [36,37,38]. A morphometric study on mandibular form and second premolar agenesis also concluded that (epi) genetic factors with influences on both skeletal and dental development most likely account for the observed skeleto-dental associations [20].
Geometric morphometric analyses, which potentially allow disentangling the roles of developmental plasticity and genetic factors, are rarely used in dentistry in general and in the study of dental agenesis specifically. Altogether, to the best of our knowledge, developmental origins of lateral incisor agenesis are still not fully understood and need to be further investigated. Hence, in the present study, we assessed the association between maxillary morphology and lateral incisor agenesis by comparing the shape and size of maxillary cross-sections across adult patients with unilateral or bilateral agenesis of maxillary lateral incisors and patients with extracted or in situ incisors. Two-dimensional landmarks and semilandmarks were collected on cross-sections of CT scans and analyzed by geometric morphometric methods [39,40,41]. Based on these data, we aimed at testing three hypotheses [21]:
H1: Agenesis causes a change in maxillary morphology due to inadequate alveolar ridge development in the area of the missing tooth (maxillary plasticity).
H2: Agenesis is caused by spatial limitations within the maxilla (dental plasticity).
H3: Common genetic/epigenetic factors cause agenesis and affect maxillary form (pleiotropy).
Material and methods
Imaging
The retrospective study sample consisted of 101 CT scans of patients treated at the University Clinic of Dentistry, Medical University of Vienna (see [14] for more details). All CT scans were indicated by the treating dentist at that time due to medical reasons following the ALARA principle and not due to study purpose. CT scans were recorded following a standard dental CT investigation protocol [42] and a high-resolution bone algorithm with a Tomoscan SR 6000 (Philips Medical Systems, Eindhoven, the Netherlands; 75 mA, 120 kV, 2 s scanning time, 1 mm slice thickness, field of view [FoV]: 90 mm) or with a Somatom Sensation 4 (Siemens, Forchheim, Germany; 80 mA, 120 kV, 1 s scanning time, 0.5 mm slice thickness, FoV: 90 mm). Ortho-radial multiplanar reconstructions were calculated orthogonally to a manually drawn central line of the jaw arch in the axial plane.
Patients were divided into three groups: agenesis of a lateral incisor, edentulous alveolar ridge at least three months after extraction of a lateral incisor, and a control group with lateral incisors in situ. If lateral incisors were missing bilaterally, one side was chosen by coin toss, and the other side was considered the contralateral side. CT scans were screened for the following inclusion criteria in order to reduce the effects of potentially confounding factors: an age of 18 years or older at the time of the scan, presence of regularly erupted adjacent teeth, and a minimum mesio-distal gap dimension of 4 mm at the marginal bone level. Exclusion criteria were cleft lip and/or cleft palate, previous augmentation procedure, and a cystic lesion exceeding 5 mm in diameter at the time of tooth loss in the extraction group. All available CT scans between 2004 and 2014 were screened for theses eligibility criteria. Patients fulfilling the criteria were included consecutively until the intended sample size was reached. Demographic data such as patient age and sex as well as data on orthodontic treatment prior to CT scanning were acquired from the patients’ dental records.
Measurements
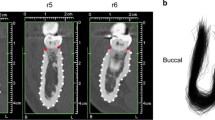
All images were arranged with the buccal side to the left and the palatal side to the right and presented in a random order to a single investigator (L.S.). For each individual, one cross-sectional picture was selected at either the mesio-distal midpoint of the lateral incisor tooth crown (control group) or at the center of the gap between central incisor and canine (agenesis and extraction). All images were randomized, and the following landmarks were digitized (see Fig. 1): (landmark 1) the most ventral point of the buccal curvature, (lm. 2 and 3) two points on the buccal and palatal alveolar crest, (lm. 4) the deepest point on the contour of the palatal curvature, and (lm. 5) one central point on the nasal floor. Landmarks 2 and 3 were placed at the same location on the alveolar ridge when no tooth was in situ. Additionally, 14 semi-landmarks were placed approximately equidistantly along the buccal contour of the maxilla and the contour of the palate (lm. 6–19). The landmark configurations were scaled based on a measured scale bar, and oriented along the central axis of the alveolar ridge by projecting the landmarks of each configuration onto the two principal components of landmarks 1–5. Randomization of images and location of landmarks were performed using tpsUtil (version 1.68) and tpsDIG2 (version 2.22; James Rohlf). To assess intra-rater reliability, 33 images of the agenesis group were measured twice in a random order.
Landmark scheme used in this study, shown once on a cross-section with a lateral incisor in situ (left) and once in a case of agenesis. Landmarks 1, 2, and 3 are anatomical landmarks; the remaining 16 landmarks (red dots) are semilandmarks. Landmarks 2 and 3 (buccal and palatal alveolar crest) are placed at the same location on the alveolar ridge when no tooth was in situ
Morphometric analysis
In total, the sample comprised 161 configurations (including the contralateral sides) of 19 landmarks each. The sample consists of seven subsets: unilateral agenesis (Auni, N = 12), bilateral agenesis (Abi, N = 25), tooth extraction (Ex, N = 24), control group with the incisors in place (CG, N = 40), contralateral side of Auni (AuniCont, N = 13), contralateral side of Abi (AbiCont, N = 25), and contralateral side of Ex (ExCont, N = 20). The deviating sample sizes of the contralateral sides result from cases that had to be excluded because of image quality. Only landmarks 1, 2, and 3 were treated as fixed anatomical landmarks; all other 16 landmarks were treated as semilandmarks and their positions along the bone outline were estimated using the sliding landmark algorithm [43, 44] (Fig. 1). This involves an iterative sliding of the semilandmarks along their outline curves so as to minimize the Procrustes distance, a measure of overall shape difference, between each configuration and the sample mean shape. The more common approach to minimize bending energy was not applicable here because the large-scale component of shape variation was not sufficiently constrained by the three anatomical landmarks. Thereafter, all configurations were superimposed by generalized Procrustes analysis [45, 46] in order to standardize for variation in overall location, scale, and orientation. Based on the resulting sets of Procrustes shape coordinates, group mean shapes were computed and analyzed by principal component analysis (PCA). Shape differences were visualized by series of reconstructed shapes [39, 47]. Additionally, a between-group PCA of all individuals was performed [48]. Type I errors for multivariate differences in group mean shapes were estimated using permutation tests with 5000 random permutations and the Procrustes distance between the mean shapes as test statistic [41, 49].
In addition to shape, we also studied the size of the maxillary cross-sections. But because the length and width of the alveolar ridge differed considerable in their statistical behavior and also in their functional relevance, the variation in the vertical and horizontal dimensions of the maxillary cross-sections was studied separately. To this end, the centroid size (square root of the summed squared differences from the mean value) was computed separately for the x and y coordinates of each configuration. This allowed us to quantify variation in the overall length and width of the alveolar ridge, without referring to any single distance measurement. Type I errors for differences in the vertical and horizontal dimensions were estimated by one-way ANOVA (histograms indicated no major deviations from normality). All morphometric and statistical analyses were performed in Mathematica 12 (Wolfram Research Inc.).
Intra-rater reliability was estimated by the intraclass correlation coefficients (ICC) applied to the first two principal components as well as to the vertical and horizontal dimensions of the maxillary cross-sections.
Results
We plotted the vertical and horizontal dimensions for the following seven subsets of the sample: unilateral agenesis (Auni, N = 12), bilateral agenesis (Abi, N = 25), tooth extraction (Ex, N = 24), control group with the incisors in place (CG, N = 40), contralateral side of Auni (AuniCont, N = 13), contralateral side of Abi (AbiCont, N = 25), and contralateral side of Ex (ExCont, N = 20). The overall width of the alveolar ridge differed significantly among the seven groups (one-way ANOVA, F = 8.87, p < 0.001), where the three agenesis groups (Auni, Abi, AbiCont) and the group with the extracted incisor had the narrowest alveolar ridge. The control group and the contralateral side of the extracted tooth had the widest alveolar dimensions, and the contralateral side of the unilateral agenesis had an intermediate width (Fig. 2A). In fact, the alveolar ridge was significantly narrower in AuniCont than in the control group (p = 0.042 despite the small size of the AuniCont sample), even though in both groups the incisor was in situ. The height of the alveolar ridge did not significantly differ among the seven groups (ANOVA, F = 1.61, p = 0.15) and showed no systematic relationship with agenesis (Fig. 2B).
Box plots showing the distribution of overall width (A) and length (B) of the alveolar process at the upper lateral incisor in the seven groups. (Abi = bilateral agenesis; AbiCont = contralateral side of Abi; Auni = unilateral agenesis; Ex = tooth extraction; AuniCont = contralateral side of Auni; ExCont = contralateral side of Ex; CG = control group). The black lines represent the group mean for each group and the white lines the median
Figure 3 shows the first two principal components (PCs) of the group mean shapes of the seven groups, accounting for 93.6% of total shape variation. PC 1 corresponds to the relative width of the alveolar ridge and separates the groups with an incisor in situ (high PC scores) from those with a missing incisor (low scores). The ordination of the groups along PC1 closely resembles that of the average horizontal dimensions in Fig. 2A. PC 2 reflects the ventral curvature of the maxillary alveolar ridge (Fig. 3, Fig. 4A) and primarily separates the two bilateral agenesis groups from those with unilateral agenesis and unilateral tooth extraction. In the bilateral agenesis group, this curvature was more pronounced than in the unilateral agenesis and tooth extraction groups. Among the groups with an incisor in situ, the contralateral side of unilateral agenesis (AuniCont) had the lowest PC 2 scores, i.e., the most pronounced ventral curvature.
The first two principal components (PCs) of the group mean shapes (group names as in Fig. 2). The maxillary shapes corresponding to the extreme values along PC 1 and PC 2 are visualized by the gray polygons
A Twofold extrapolation of the shape variation along PC 2 as depicted in Fig. 3, which reflects the ventral curvature of the maxillary alveolar ridge. These two shapes correspond to PC 2 scores of − 0.07 and 0.07, respectively. B Smooth histograms of the PC 2 scores for the unilateral agenesis group (red) and the two bilateral agenesis groups (black, gray)
Individual variation within groups was relatively large (Suppl. Figure 1), but the unilateral and bilateral agenesis groups differed clearly in their mean score along PC 2 (Fig. 4B). Because of the small unilateral agenesis sample (N = 12), the mean differences between bilateral and unilateral agenesis groups were at the border of statistical significance (Auni vs. Abi p = 0.08, Auni vs. AbiCont p = 0.12). However, the mean shapes of both bilateral agenesis groups differed significantly from the unilateral extraction group (p = 0.008, p = 0.023).
Intraclass correlation coefficients were 0.96 and 0.88 for PC1 and PC 2, respectively. The vertical and horizontal dimensions both showed intraclass correlations of 0.93. This indicates a relatively small measurement error and high intra-rater reliability. But as these coefficients were calculated from individuals of the agenesis group only, intraclass correlations among groups is likely even higher than within group.
Discussion
In this retrospective study, we compared the size and shape of the maxillary alveolar ridge in patients with bilateral or unilateral agenesis of the lateral incisor to patients with an extracted lateral incisor and patients with all lateral incisors in situ. We found that both the absolute dimensions of the alveolar ridge as well as its cross-sectional shape vary strongly among individuals. The corresponding statistical distributions overlapped among all groups. However, the average alveolar width (PC1) was clearly larger in the dentulous groups compared to patients with agenesis or extracted lateral incisors. Among edentulous groups, the shape of the ventral curvature of the alveolar ridge (PC 2) differed on average between bilateral agenesis and unilateral agenesis as well as post tooth extraction: in the bilateral agenesis group the buccal curvature of the alveolar ridge was most pronounced.
Alveolar ridge formation is strongly affected by tooth development and eruption [50]. In patients with agenesis of the upper lateral incisors, a labial concavity of the alveolar ridge is frequently present [51, 52]. Our results show a more pronounced ventral curvature of the alveolar ridge in individuals with bilateral lateral incisor agenesis in comparison to those with unilateral agenesis. This fits with the well-documented association of alveolar cross-sectional form and tooth agenesis. It also appears to indicate a positive correlation between the extents of these anomalies. Similar findings were reported for other teeth [53,54,55,56]. For instance, a previous study on lower second premolar agenesis revealed that cross-sectional mandibular size and shape significantly differs among individuals with and without agenesis of the lower second premolars [20].
Among our edentulous groups, the ventral curvature appeared least pronounced in patients, where the lateral incisor had been present but extracted. It is well documented that the residual alveolar ridge undergoes marked dimensional alterations after tooth extraction [57]: up to 35% bucco-oral width reduction within the first three months has been reported [58]. However, this reduction primarily occurs at the edge of the alveolar ridge, with a bucco-palatal width reduction resulting in a knife-edged shape rather than an accentuation of the ventral curvature.
Our first hypothesis, namely that the developing tooth affects maxillary growth, is clearly verified. Interestingly, we found that alveolar ridge shape differs on average between bilateral agenesis and the affected side of unilateral agenesis, even though in both cases an incisor is absent. Similarly, we found that the alveolar width of the unaffected side of unilateral agenesis tends to be smaller than in the control group (p = 0.042 despite the small size of the AuniCont group), even though in both cases the incisor is in situ. Both findings cannot be explained by H1. Instead, they suggest that maxillary shape has an effect on dental agenesis (H2) or that both are influenced by a shared common factor (H3), e.g., some pleiotropic genes. Potentially, even all three causal pathways (H1–H3) could be present.
Without an experimental design, it is difficult to distinguish between H2 and H3 based on our morphometric data, but the overlap of the groups in their size and shape distributions does not support a universal threshold of alveolar width necessary for dental development. However, such a threshold could depend on dental and cranial sizes, among other factors, and thus vary among individuals. From a statistical perspective, the relationship appears like a probabilistic one: The narrower the alveolar ridge and the more concavely shaped its ventral curvature, the more likely is the agenesis of the lateral incisor.
Clinically, the treatment of a missing lateral incisor is dictated by the width of the alveolar ridge. It has been shown that straightforward implant placement in this region has a tendency to be less likely if the tooth is missing due to agenesis rather than extraction, i.e., the more pronounced buccal curvature of the alveolar ridge might result in a higher need of bone augmentation prior to or in combination with implant placement than the crestal width. The present results suggest that this effect may be aggravated in cases of bilateral agenesis.
Some limitations — primarily due to the retrospective design — should be considered. Several parameters that potentially affect post-extraction bone remodeling were not completely available for all patients and thus not considered in the analysis, for example, presence of systemic diseases and medication, smoking status, and the exact timepoint of tooth extraction. Nevertheless, the presence of any augmentation material was defined as exclusion criterion and a minimum of 3 months passed between tooth extraction and CT recording. Another potential bias in the agenesis group is orthodontic treatment prior to CT scanning. This information was available for 19 patients, of which 12 patients received treatment, including space opening in the region of the missing lateral incisor. However — as reported previously [14] — none of the parameters defining the alveolar ridge dimension (i.e., bucco-palatal width, area, and height) presented any significant differences between these two groups (i.e., space opening vs. no space opening).
Conclusions
In summary, we found an association of the maxillary alveolar ridge and agenesis of the lateral incisor, but this association is not only a simple, unidirectional result of the absent permanent tooth. Instead, a too small or inappropriately shaped alveolar ridge may also inhibit normal formation or development of the tooth bud, thus leading to dental agenesis in this anatomical region. It is also possible that pleiotropic genetic and developmental factors influence both maxillary growth and dental development. Presumably, all three causal pathways contribute to the observed association.
Data availability
The data supporting the findings of this article are openly available in the Open Science Framework (OSF) repository at: https://osf.io/72kxc/.
References
Rakhshan V (2015) Meta-analysis of observational studies on the most commonly missing permanent dentition (excluding the third molars) in non-syndromic dental patients or randomly-selected subjects, and the factors affecting the observed rates. J Clin Pediatr Dent 39(3):199–207
Polder BJ et al (2004) A meta-analysis of the prevalence of dental agenesis of permanent teeth. Community Dent Oral Epidemiol 32(3):217–226
Behr M et al (2011) Survey of congenitally missing teeth in orthodontic patients in Eastern Bavaria. Eur J Orthod 33(1):32–36
Moss ML, Young RW (1960) A functional approach to craniology. Am J Phys Anthropol 18(4):281–292
Nordquist GG, McNeill RW (1975) Orthodontic vs restorative treatment of the congenitally absent lateral incisor–long term periodontal and occlusal evaluation. J Periodontol 46(3):139–43
Robertsson S, Mohlin B (2000) The congenitally missing upper lateral incisor A retrospective study of orthodontic space closure versus restorative treatment. Eur J Orthod 22(6):697–710
Kinzer GA, Kokich VO Jr (2005) Managing congenitally missing lateral incisors Part III: single-tooth implants. J Esthet Restor Dent 17(4):202–10
Kinzer GA, Kokich VO Jr (2005) Managing congenitally missing lateral incisors Part II: tooth-supported restorations. J Esthet Restor Dent 17(2):76–84
Kokich VO Jr, Kinzer GA (2005) Managing congenitally missing lateral incisors Part I: Canine substitution. J Esthet Restor Dent 17(1):5–10
Strbac GD et al (2017) Survival rate of autotransplanted teeth after 5 years - a retrospective cohort study. J Craniomaxillofac Surg 45(8):1143–1149
Cosyn J, Cleymaet R, De Bruyn H (2016) Predictors of alveolar process remodeling following ridge preservation in high-risk patients. Clin Implant Dent Relat Res 18(2):226–233
Kurihara S, Enlow DH, Rangel RD (1980) Remodeling reversals in anterior parts of the human mandible and maxilla. Angle Orthod 50(2):98–106
Schuh A et al (2019) Ontogeny of the human maxilla: a study of intra-population variability combining surface bone histology and geometric morphometrics. J Anat 235(2):233–245
Bertl K et al (2017) A wide mesio-distal gap in sites of congenitally missing maxillary lateral incisors is related to a thin alveolar ridge. Clin Oral Implants Res 28(9):1038–1045
Sarkar L, Sharpe PT (1999) Expression of Wnt signalling pathway genes during tooth development. Mech Dev 85(1–2):197–200
Satokata I, Maa R (1994) Msx1 deficient mice exhibit cleft palate and abnormalities of cra. Nat Genet 6(4):348–56
D’Souza RN et al (1999) Cbfa1 is required for epithelial-mesenchymal interactions regulating tooth development in mice. Development 126(13):2911–2920
Rodrigues, A.S., et al. (2019) Is dental agenesis associated with craniofacial morphology pattern? A systematic review and meta-analysis. Eur J Orthod
Takahashi Y et al (2018) Examination of craniofacial morphology in Japanese patients with congenitally missing teeth: a cross-sectional study. Prog Orthod 19(1):38
Bertl MH et al (2016) Second premolar agenesis is associated with mandibular form: a geometric morphometric analysis of mandibular cross-sections. Int J Oral Sci 8(4):254–260
Huang Y et al (2017) Obeservations on association between third molar agenesis and craniofacial morphology. J Orofac Orthop 78(6):504–510
Takeshima T et al (2004) Relationship between formation and eruption of permanent mandibular buccal dentition and vertical height of the mandibular body: longitudinal study of Japanese boys from 4 to 9 years of age. World J Orthod 5(4):332–338
De Coster PJ et al (2009) Dental agenesis: genetic and clinical perspectives. J Oral Pathol Med 38(1):1–17
Kangas AT et al (2004) Nonindependence of mammalian dental characters. Nature 432(7014):211–214
Kavanagh KD, Evans AR, Jernvall J (2007) Predicting evolutionary patterns of mammalian teeth from development. Nature 449(7161):427–432
Kim YH (2011) Investigation of hypodontia as clinically related dental anomaly: prevalence and characteristics. ISRN Dent 2011:246135
Schwindler, D.R., Primate dentition: an introduction to the teeth of non-human primates 2008: Cambridge University Press.
Altug-Atac AT, Erdem D (2007) Prevalence and distribution of dental anomalies in orthodontic patients. Am J Orthod Dentofacial Orthop 131(4):510–514
Jonke E et al (2007) Secular trends in the facial skull from the 19th century to the present, analyzed with geometric morphometrics. Am J Orthod Dentofacial Orthop 132(1):63–70
Ungar, P.S., Evolution’s bite: a story of teeth, diet, and human origins. 2017: Princeton University Press.
Kajii TS et al (2004) Agenesis of third molar germs depends on sagittal maxillary jaw dimensions in orthodontic patients in Japan. Angle Orthod 74(3):337–342
Moghadam MG, Etemadi F, Akbari N (2018) The relationship between agenesis of third molar and craniofacial morphology in orthodontic patients. J Int Soc Prev Community Dent 4:304–308
Dassule HR et al (2000) Sonic hedgehog regulates growth and morphogenesis of the tooth. Development 127(22):4775–4785
Jernvall J, Thesleff I (2012) Tooth shape formation and tooth renewal: evolving with the same signals. Development 139(19):3487–3497
Seppala, M., et al. (2017) Sonic hedgehog signaling and development of the dentition. J Dev Biol. 5(2)
Celikoglu M, Bayram M, Nur M (2011) Patterns of third-molar agenesis and associated dental anomalies in an orthodontic population. Am J Orthod Dentofacial Orthop 140(6):856–860
Garib, D.G., et al. (2010) Agenesis of maxillary lateral incisors and associated dental anomalies. Am J Orthod Dentofacial Orthop. 137(6):732.e1–6; discussion 732–3
Baccetti T (1998) A controlled study of associated dental anomalies. Angle Orthod 68(3):267–274
Mitteroecker P, Gunz P (2009) Advances in geometric morphometrics. Evol Biol 36(2):235–247
Halazonetis DJ (2004) Morphometrics for cephalometric diagnosis. Am J Orthod Dentofacial Orthop 125(5):571–581
Bookstein, F.L. (2018) A course in morphometrics for biologists: geometry and statistics for studies of organismal form., ed. C.U. Press.,Cambridge, UK.
Gahleitner A, Watzek G, Imhof H (2003) Dental CT: imaging technique, anatomy, and pathologic conditions of the jaws. Eur Radiol 13(2):366–376
Gunz P, Mitteroecker P (2013) Semilandmarks: a method for quantifying curves and surfaces. Hystrix, the Italian journal of mammalogy 24(1):103–109
Bookstein FL (1997) Landmark methods for forms without landmarks: morphometrics of group differences in outline shape. Med Image Anal 1(3):225–243
Rohlf FJ, Slice D (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Biol 39(1):40–59
Mitteroecker P, Gunz P, Windhager S, Schaefer K (2013) A brief review of shape, form, and allometry in geometric morphometrics, with applications to human facial morphology. Hystrix 24(1):59–66
Bookstein, F. (1991) Morphometric tools for landmark data: geometry and biology., ed. C.U. Press. Cambridge (UK).
Mitteroecker P, Bookstein F (2011) Linear discrimination, ordination, and the visualization of selection gradients in modern morphometrics. Evol Biol 38:100–114
Good, P., Permutation tests: a practical guide to resampling methods for testing hypotheses. 2nd edition. ed, ed. S.S.i.S. Springer. 2000, New York.
Schroeder HE (1986) (1986) The periodontium. Handbook of microscopic anatomy. Springer, Berlin, Germany, pp 233–246
Uribe F et al (2013) Cone-beam computed tomography evaluation of alveolar ridge width and height changes after orthodontic space opening in patients with congenitally missing maxillary lateral incisors. Am J Orthod Dentofacial Orthop 144(6):848–859
Uribe F et al (2013) Alveolar ridge width and height changes after orthodontic space opening in patients congenitally missing maxillary lateral incisors. Eur J Orthod 35(1):87–92
Graber LW (1978) Congenital absence of teeth: a review with emphasis on inheritance patterns. J Am Dent Assoc 96(2):266–275
Brook AH (1984) A unifying aetiological explanation for anomalies of human tooth number and size. Arch Oral Biol 29(5):373–378
Al-Nimri K, Gharaibeh T (2005) Space conditions and dental and occlusal features in patients with palatally impacted maxillary canines: an aetiological study. Eur J Orthod 27(5):461–465
Alves-Ferreira M et al (2014) Identification of genetic risk factors for maxillary lateral incisor agenesis. J Dent Res 93(5):452–458
Morjaria KR, Wilson R, Palmer RM (2014) Bone healing after tooth extraction with or without an intervention: a systematic review of randomized controlled trials. Clin Implant Dent Relat Res 16(1):1–20
Schropp L et al (2003) Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent 23(4):313–323
Funding
Open access funding provided by University of Vienna.
Author information
Authors and Affiliations
Contributions
Conceptualization M.N., P.M., and E.J. methodology M.N. and P.M.; validation M.N. and P.M.; formal analysis M.N., M.H.B., L.S., and P.M.; investigation M.N., L.S., and M.H.B.; resources A.G., P.M., and E.J.; data curation M.N.; writing original draft preparation M.N., P.M., and K.B.; writing, review and editing K.B., M.H.B., and E.J.; supervision P.M. and E.J.; project administration E.J.; All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics and consent to participate
The study was approved by the ethics committee of the Medical University of Vienna, Austria (2005/2012). The ethics approval of the present retrospective study did not require collecting any type of consent of the participants.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nemec, M., Schwarz, L., Bertl, M.H. et al. Maxillary lateral incisor agenesis is associated with maxillary form: a geometric morphometric analysis. Clin Oral Invest 27, 1063–1070 (2023). https://doi.org/10.1007/s00784-022-04690-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04690-9