Abstract
Objectives
IL-23p19/Ebi3 (IL-39) was described as a new IL-12 family member. The aim of this study is to evaluate the gingival crevicular fluid (GCF) IL-39 levels in periodontal diseases and health and to correlate them to GCF levels of IL-1β and periostin.
Materials and methods
Sixty-six adult patients were included in the study. The study design was comprised of three groups, each containing 22 individuals: the periodontally healthy (PH), gingivitis (G), and periodontitis (P) groups. The clinical periodontal parameters were recorded and GCF samples were collected from the participants. GCF interleukin (IL)-39, IL-1β, and periostin levels were examined using the enzyme-linked immunosorbent assay.
Results
GCF IL‑1β, periostin, and IL-39 levels were higher in the P and G groups than in the PH group (p < 0.001). Positive correlations were detected between all GCF biochemical parameters and clinical periodontal parameters (p < 0.05). In the multivariate generalized linear regression analysis, the P (β = 37.6, 95% CI = 22.9–52.4) and G (β = 28.4, 95% CI = 15.8–41) groups were associated with GCF IL-39 levels (p < 0.001).
Conclusion
IL-39 levels were elevated in the presence of periodontal disease paralleling the increase in IL‑1β and periostin levels. IL-39 may have a role in the periodontal inflammation process.
Statement of clinical relevance
IL-39, a new cytokine from the IL-12 family, can be a possible predictor marker of periodontal diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Periodontal diseases are multifactorial chronic inflammatory diseases. The main driver of periodontal disease in susceptible individuals is the interaction and development of dysbiotic communities and destructive inflammation which co-develop and are reciprocally reinforced [1]. The nature of microbial stimuli that trigger disruptions in periodontal homeostasis are determinants of host responses in the gingiva [2]. Host modulation therapies, such as anti-cytokine therapy and engineered exosomes derived from dendritic cells, can restore the balance between pro-inflammatory and anti-inflammatory mediators and create an environment that can reverse dysbiosis [3, 4]. For these reasons and to understand the role of cytokines in periodontitis, cytokine studies are currently being conducted.
The complex cytokine network involved in the immune system includes specific cytokine receptors, pro-inflammatory and anti-inflammatory cytokines. In this context, cytokines in inflamed periodontal tissues are thought to have a prominent effect on the host modulation and initiation and progression of periodontal disease [5]. One of the most studied is interleukin 1 (IL-1), which is an important proinflammatory mediator in the host inflammatory response. IL-1β has a crucial role in activating proteinase, collagenase, stimulating the production of prostaglandin E2, and enhancing bone resorption and degradation of the extracellular matrix in periodontal diseases [6, 7].
Periodontal tissue destruction is mediated by interactive relationships between proinflammatory and anti-inflammatory mediators in the host response [8]. Periostin, a secreted hematopoietic stem cell protein of 90 kDa containing glutamate, is expressed in the periosteum and periodontal ligaments [9]. It is thought to enhance cell survival and differentiation, promote cell adhesion and fibrogenesis, and to affect periodontal tissue remodeling and bone formation by affecting differentiation, adhesion, and proliferation of osteoblasts [10, 11]. This protein regulates cell functions to favor tissue regeneration by different signaling pathways such as the PI3K/Akt/mTOR, αvβ3 integrin/FAK/PI3K/Akt, αvβ3 integrin/extracellular-related kinase [10, 12,13,14].
Previous studies suggested that periostin expression is regulated by various factors such as interleukin-4, interleukin-13, and transforming growth factor-β (TGF-β) [15]. Periostin expression in the periodontal ligament is regulated by TGF-β, mechanical stress such as during mastication and tooth movement [11]. A study stated that periostin values increased in the presence of periodontitis in gingival crevicular fluid (GCF) [16].
The IL-12 family of cytokines regulates adaptive immune responses and endogenous responses by inducing natural killer and Th1 cells to produce interferon-gamma (IFN-γ) [17]. IL-1β has a major role in IL-12β b production and IL-12 αβ heterodimer secretion and can synergize with IFN-γ secretions of high amounts of IL-12 [18]. IL-12 is higher in GCF in the presence of periodontitis [19]. In recent years, IL-23p19/Ebi3 (IL-39) was described as a new IL-12 family member as an additional combination to the four known members of this cytokine by Wang et al. [20]. This cytokine has been proposed as pro-inflammatory and a potential predictor and prognostic marker in acute coronary syndrome (ACS) [21]. The association of IL-39 with inflammatory diseases, except for ACS and systemic lupus erythematosus, has not been clarified yet [22].
To our knowledge, there is no study in the literature investigating IL-39 cytokine levels in periodontal disease. We aimed to assess IL-39 levels in GCF in patients with different periodontal phenotypes and to correlate them with IL-1β and periostin levels. Our null hypothesis is that no differences exist in GCF levels of patients with periodontitis, gingivitis, and gingival health.
Materials and methods
The present case–control study was conducted in the Department of Periodontology in the Faculty of Dentistry at Hatay Mustafa Kemal University, Hatay, Turkey. The study protocol was approved by the Ethics Committee for the Use of Human Subjects in Research of Hatay Mustafa Kemal University (Protocol No: 2021/70) and the study was carried out in accordance with the tenets of the Declaration of Helsinki. Individuals were included in the study from July 2021 through November 2021. Written informed consent was obtained from each participant before the clinical periodontal examination.
Study groups
Sixty-six adult individuals were enrolled in the study. Participants were recruited into 3 sex, age-, and body mass index (BMI)-matched groups with 22 participants in each periodontally healthy (PH), gingivitis (G), and periodontitis (P) groups. In accordance with the demographic variables of the patients included in the P group, the patients in the G and PH groups were included.
The inclusion criteria included the following: (a) individuals who had more than 18 teeth; (b) individuals who had had no antibiotic therapy in the past 3 months; (c) individuals who had no history of periodontal treatment in the past 6 months; and (d) individuals who had never smoked.
The exclusion criteria included the following: (a) individuals who had any self-reported systemic condition or disease, which are confounding factors, e.g., AIDS, rheumatoid arthritis, cardiovascular diseases, diabetes; (b) individuals were current or former smokers; (c) individuals were pregnant, and (d) individuals with BMIs ≥ 25 kg/m2.
Clinical measurements
Periodontal parameters
Periodontal clinical parameters were recorded by a single calibrated examiner (k = 0.93) (author AS). Intra-examiner agreement was determined for CAL. The intra-examiner reproducibility was determined through repeated examinations of 10 subjects with a one-hour interval. Clinical periodontal measurements were assessed using the following periodontal measurements for periodontal diagnosis.
The measurements were performed using a Williams periodontal probe (Hu-Friedy, Chicago, IL, USA) and included probing pocket depth (PPD), clinical attachment level (CAL), plaque index (PI) [23], gingival index (GI) [24], and percentage bleeding on probing (BOP) [25] at six sites per tooth (mesio-buccal, buccal, disto-buccal, mesio-lingual, lingual and disto-lingual) on each tooth.
Diagnosis of periodontal diseases and conditions was made according to the radiographic and clinical diagnostic criteria proposed by the 2017 World Workshop on Classification of Periodontal and Peri-implant Diseases and Conditions [26]. Individuals with a BOP < 10% without attachment loss and radiographic bone loss were considered to have periodontal health [27]. Only generalized gingivitis patients were included in this study. Individuals presenting with a BOP ≥ 30%, and PPD ≤ 3 mm without radiographic bone loss and attachment loss were considered to have gingivitis [28]. The criteria for periodontitis included patients with CAL ≥ 5 mm in two or more interproximal sites and PPD ≥ 6 mm in one or more interproximal sites. Only stage III–IV (severe) periodontitis was included in the present study [29].
Collection of GCF samples
GCF samples were collected following 8 h of night fasting and one week after clinical periodontal measurements [30]. The samples were collected from the teeth side with BOP positive in patients with gingivitis and CAL ≥ 5 mm, PPDs ≥ 5 mm, and BOP positive in patients with periodontitis. Six GCF samples were taken per participant. Samples were collected from a mesio-buccal and a disto-palatinal site on each of three teeth (incisors, premolars, and molars). Saliva contamination was prevented by isolation with cotton rolls and gently air-drying the sampling area. Samples contaminated with saliva or blood were not included. Plaque was gently removed from the sampling area by the periodontal curette. The samples were collected within 30 s with standardized paper strips (Periopaper; Oraflow Inc., Plainview, NY) by the orifice method [31]. The volumes were measured on a pre-calibrated electronic gingival fluid measuring device (Oraflow Inc., Plainview, NY)|| [32]. The values of the electronic device were referenced to a standard curve and converted to an actual volume (µl). All of the Periopaper strips were pooled in plastic Eppendorf microcentrifuge tubes. They were stored at – 80 °C until the biochemical analysis.
At a later stage, samples were thawed, 750 μl phosphate buffer (Phosphate Buffered Saline, pH: 7.00, 137 mM NaCl, 10 mM Na2PO4, and 2.7 mM KCl) was added into the Eppendorf microcentrifuge tubes containing the sample strips and samples were eluted for 30 min at room temperature before the assay [33]. The tubes were centrifuged at 12,000 × g for 15 min. After removing the strips, the supernatants were used for the measurement of IL-1β, periostin, and IL-39.
Laboratory analyses
Measurement of IL‑1β levels in GCF samples
IL-1β levels were assayed with commercially available kits using the enzyme-linked immunosorbent assay (ELISA) method (Elabscience, catalog no: E-EL-H0149). The optical density was measured spectrophotometrically at a wavelength of 450 nm (Thermo Scientific MultiscanGo, Finland). The assay ranges for the IL-1β kit were 7.81–500 pg/mL, sensitivity 4.69 pg/mL, and the intra- and interassay coefficients of variance (CV%) were < 10%. The results were presented as pg.
Measurement of periostin levels in GCF samples
Periostin levels were determined with commercially available kits using the ELISA method (Elabscience, catalog no: E-EL-H6113). The optical density was measured spectrophotometrically at a wavelength of 450 nm (Thermo Scientific MultiscanGo, Finland). The assay ranges for the periostin kit were 3.13–200 ng/mL, sensitivity 1.88 ng/mL, and the intra- and interassay coefficients of variance (CV%) were < 10%. The results were presented as pg.
Measurement of IL‑39 levels in GCF samples
IL-39 levels were studied with commercially available kits using the ELISA method (MyBioSource, catalog no: MBS167915). The optical density was measured spectrophotometrically at a wavelength of 450 nm (Thermo Scientific MultiscanGo, Finland). The assay ranges for the IL-39 kit were 2–600 ng/L, sensitivity 1.07 ng/L, and the intra- and interassay coefficients of variance (CV%) were < 10%. The results were presented as ng.
Statistical analysis
The main study outcome was IL-39 (pg/30sn) GCF levels between different periodontal phenotypes (periodontitis, gingivitis, healthy). In the absence of previous data about IL-39 GCF levels, a convenience sample of 66 participants was chosen for this study. Post-hoc sample size calculation revealed that 22 patients per group would give 100% power for effect size f of 1.04 (standard deviation: 13.72) and α = 0.05. Gpower package version 3.1 was used for sample size calculations.
The normality of continuous variables was evaluated by Shapiro–Wilk’s test. Non-parametric statistical methods were performed for values with skewed distribution. Descriptive statistics were presented as median (interquartile range) for the non-normally distributed variables.
Kruskal–Wallis test was performed for comparison of more than two non-normally distributed variables and Dunn multiple comparison test was performed for post hoc pairwise multiple comparison analyses. One-way ANOVA test was performed for comparison of more than two normally distributed groups and Tukey test was performed for post hoc pairwise multiple comparison analyses. In addition, significance values have been adjusted by the Bonferroni correction for multiple tests. The chi-square test was used to analyze the associations between categorical variables. ROC analysis was performed in order to evaluate diagnostic performance of IL-39 levels (pg/30sn) for periodontal diseases. Area under curve value and also sensitivity and specificity values have been calculated according to presence of periodontal diseases and absence. The correlation between two non-normally distributed variables was evaluated by Spearman Rho correlation coefficient. Multivariate generalized linear model was performed to evaluate the association between the presence of periodontal disease and IL-39 levels. The variables were included in the multivariate model, provided that significance at the 5% level was obtained in the univariate generalized linear model analysis. Variance inflation factors were calculated to check multicollinearity. IL-39 levels (pg/30sn) were selected as dependent variable, while age, sex, BMI, IL-1β levels (pg/30sn), periostin levels (pg/30sn), and periodontal groups were set as independent variables in the multivariate model. The MedCalc Statistical Software (ver. 12.7.7; MedCalc Software bvba, Ostend, Belgium) was used for statistical analyses and p < 0.05 was considered statistically significant.
Results
Demographic findings
Table 1 shows the demographic characteristics of the groups. Sex, age, and BMI were not statistically significantly different among the groups (p = 0.999, p = 1, and p = 0.997 respectively).
Clinical findings
Table 2 shows the clinical periodontal parameters. PI, GI, BOP (%), and GCF volume were higher in the P and G groups than in the PH group (p < 0.001). As by definition, PPD was higher in the P and G groups than in the PH group and was higher in the P group than in the G group (p < 0.001). CAL were higher in the P group than in the G and PH groups (p < 0.001).
Laboratory findings
Table 3 shows the intergroup comparisons of biochemical markers. GCF IL‑1β, periostin, and IL-39 total amounts were higher in the P and G groups than in the PH group (p < 0.001). GCF IL‑1β and perostin concentrations were not significantly different among the groups (p = 0.571 and p = 0.071 respectively). GCF IL-39 concentrations were higher in the PH group than in the P and G groups (p < 0.001).
Correlations
Table 4 shows correlations between clinical periodontal parameters and GCF IL‑1β, periostin, and IL-39 total amounts. Positive correlations were detected between GCF IL‑1β, periostin, and GCF IL-39 total amounts and all clinical periodontal parameters (p < 0.05).
Table 5 shows univariate and multivariate generalized linear regression analyses of factors affecting GCF IL-39 total amounts. Age, sex, and BMI were not associated with GCF IL-39 total amounts (p > 0.05). P (β = 33.6, 95% CI = 42.6–24.6) and G (β = 25.3, 95% CI = 16.3–34.3) groups, Periostin total amounts (β = 0.29, 95% CI = 0.14–0.44), and IL‑1β total amounts (β = 0.29, 95% CI = 0.17–0.4) were associated with GCF IL-39 total amounts (p < 0.001) in the univariate generalized linear regression analysis. P (β = 37.6, 95% CI = − 22.9–52.4) and G (β = 28.4, 95% CI = 15.8–41) groups were associated with higher GCF IL-39 total amounts (p < 0.001 and 0.031, respectively) in the multivariate generalized linear regression analysis.
Roc analysis
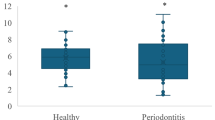
In ROC analysis, area under curve (AUC) value was calculated as 0.947 for IL-39 levels (pg/30sn). Sensitivity was calculated as 95.5%, whereas specificity values as 81.82%. The cut off value was found as > 114.7 (Fig. 1).
Discussion
This is the first study to evaluate GCF IL-39, IL-1β, and periostin levels in periodontitis, gingivitis and periodontal health to the best of the authors’ knowledge. The findings of the present study show that IL-39, IL-1β, and periostin levels were elevated in GCF in presence of periodontal disease.
We presented data for both total levels and concentrations and discussed both in the current study. It is interesting to notice that higher ‘concentrations’ of the 3 studied cytokines were detected in healthy vs. gingivitis and periodontitis patients. However, total amounts of cytokines in GCF samples per sampling time have been suggested as a better indicator of relative GCF constituent activity rather than concentration because concentrations are directly affected by the sample volume [34,35,36]. Several studies have indicated that the expression of GCF data as the total amount per standardized sampling time is a more sensitive way than reporting them as concentration, and should be used when estimating periodontal disease activity [34, 35, 37, 38]. This is particularly true in the present study, as GCF volume was 8 times higher in periodontitis vs. controls and 6 times higher in gingivitis vs. controls. Therefore, total amounts have been taken as the main results in the current study.
The main novel finding of the study relates to IL-39, which was discovered as a new inflammatory marker in recent years, is produced in vivo and in vitro by activated mouse B cells and upregulates neutrophils [20, 39]. IL-39 total levels were higher in GCF of periodontitis and gingivitis patients compared with healthy controls. Also, P and G groups were associated with higher GCF IL-39 total levels compared to PH group in generalized linear regression analysis. These results were independent confounding factors such as BMI, age, and sex. Therefore, IL-39 levels appeared to increase in the presence of periodontal infection, suggesting that IL-39 may be an inflammatory marker in humans, playing a role in the inflammatory processes of periodontal diseases as well as in different disease groups. This is in agreement with some previous literature [21, 39, 40]. In a preclinical study, Wang et al. suggested that IL-39 may have a role in lupus immunopathogenicity [20]. It has been demonstrated that the number of B cells involved in the production of IL-39 increases in mice with lupus, and the severity of the disease decreases as a result of suppression of IL-39 [39]. Also, it was noticed that anti-IL-39 antibodies have a therapeutic effect in a lupus-like pathology in mice, suggesting that IL-39 could be a potential marker for the treatment of systemic lupus erythematosus due to its proinflammatory nature [41]. Another study showed that IL-39 affects pancreatic cancer by pro-tumorigenic function [42]. In a clinical study, it has been reported that IL-39 levels in serum are increased in patients with ACS compared with healthy individuals and there were positive correlations between IL-39 levels and hs-CRP [21]. In addition, it has been shown that serum IL-39 levels were higher in patients with relapsing–remitting multiple sclerosis and neuromyelitis optica spectrum disorders than healthy controls [43]. In contrast, a few studies failed to show evidence of IL-39 production by human cells. Two in-vivo studies noticed that, although IL-39 is secreted in human cells, it may be below the detection limit or it has no functional response [40, 44]. In these studies, the choice of cells was made according to previous studies that were carried out on mice [40, 44]. The discrepancy between the results of these studies and those of the present study may be due to the production of IL-39 by other cells or targeting different cells in humans as opposed to mice. Another reason for this discrepancy can be that several inducing pathways interact in in-vivo studies as opposed to one-way interactions in in-vitro studies.
Further evidence of a potential pro-inflammatory nature of IL-39 derives from its correlation with IL-1β levels in the present study. IL-1β is a key cytokine in periodontal diseases because of its dual function in collagen degradation and effects on enhancing bone resorption and inhibiting bone formation [6, 45, 46]. GCF IL‑1β total amounts were higher in the P and G groups than in the PH group. Compatible with the current study results, previous studies have demonstrated that IL-1β is usually found in higher levels in patients with periodontitis than in the healthy controls in GCF and different tissue samples [47, 48] including saliva [49], although its potential use as a biomarker is yet unclear due to inconsistencies across studies [50]. The association between IL-39 and IL-1β levels in generalized linear regression analysis is supported by a study which demonstrated that IL-1 plays a role through upregulation of IL-12 secretion by dendritic cells in innate and adaptive immunity [18]. Another study noticed that IL-12 needs the induction of IL-1 beta to stimulate IFN-gamma production by natural killer cells and IL-1 plays a role in the IL-12-mediated inflammatory response to some bacteria [51]. The results of these previous studies are consistent with the fact that IL-39, a cytokine from the IL-12 family, correlates with IL-1 levels in the presence of periodontal inflammation.
Periostin, playing a role in the remodeling of periodontal tissues, is an important adhesion molecule and structural mediator [10]. This protein is localized among the cytoplasmic processes of periodontal cementoblasts and fibroblasts and the adjacent collagen fibrils [52]. Thus, it can be used as a marker of the periodontal regeneration process [53]. The results of the present study indicated that periostin total amounts were higher in the P and G groups than in the PH group. There are some contradictory results in the literature. It has been noticed that Porphyromonas gingivalis lipopolysaccharide and TNF-α decrease periostin levels in the periodontal ligament [54]. In contrast with the present study, GCF periostin levels were previously shown to decrease in relation to the severity of periodontitis [55, 56]. These results may support the idea that periodontal inflammation can cause of periostin downregulation. However, in agreement with the present study, Arslan and co-workers found that periostin GCF levels decreased from the periodontitis group to the gingivitis and to the healthy control group [16]. As in the present study, samples had been taken from bleeding sites (GI = 2 and BOP positive). It can be considered that the inconsistency with some of the previous studies may be related to the differences in the disease activity in the site of the GCF samples taken. Previous studies have also shown that periostin differentially expressed and upregulated contributes to the development and progression of various inflammatory diseases such as cancer, diabetes, and bowel disease [57, 58]. The results may suggest that in addition to playing a role in the regeneration processes, periostin can also be effective in regulating inflammatory responses [11, 59]. It can be thought that periostin increases as a protective mechanism and response in the periodontal inflammation process. In this process, periostin may increase to allow tissue repair and remodeling.
Lending further support to the association with the periodontal groups in this study are the correlations between the assessed biomarkers and continuous measures of disease severity. We detected positive correlations between GCF IL‑1β, periostin and IL-39 levels, and all periodontal clinical parameters, in keeping with some previous studies [21, 43, 60, 61]. In the Roc analysis, IL-39 showed strong performance in presence of periodontal disease. IL-39 showed a high specificity for periodontal disease and high sensitivity for periodontal health. Thus, GCF IL-39 levels could potentially be used as a biomarker for the presence of periodontal diseases, upon confirmation of the findings of the present study.
The strengths of this study are the novelty in assessing GCF levels of IL-39, the rigorous pre-sampling protocol, and the fact that smokers were excluded, while sex, age, and BMI were matched among the groups to reduce the risk of finding spurious associations [62, 63]. Biochemical findings are given as both concentration and total amount values in the present study.
The case–control design and relatively small sample size may be limitations of the present study.
Conclusions
The present study findings showed that GCF IL-39, IL‑1β, and periostin total amounts were higher in patients with periodontitis and gingivitis than in the healthy controls. These results suggest that the presence of periodontal diseases is associated with increased GCF IL-39, IL-1β, and periostin levels. IL-39, a new cytokine from the IL-12 family, may have a role in the periodontal inflammation process and it can be a possible marker of periodontal diseases. Further studies with larger sample size and longitudinal assessment post-treatment are needed to clarify the functions of IL-39 and its possible role in the pathogenesis of periodontal diseases.
References
Hajishengallis G, Lamont RJ (2021) Polymicrobial communities in periodontal disease: their quasi-organismal nature and dialogue with the host. Periodontol 2000 86:210–230. https://doi.org/10.1111/prd.12371
Abusleme L, Hoare A, Hong BY, Diaz PI (2021) Microbial signatures of health, gingivitis, and periodontitis. Periodontol 2000 86:57–78. https://doi.org/10.1111/prd.12362
Hajishengallis G, Chavakis T, Lambris JD (2020) Current understanding of periodontal disease pathogenesis and targets for host-modulation therapy. Periodontol 2000 84:14–34. https://doi.org/10.1111/prd.12331
El-Awady AR, Elashiry M, Morandini AC, Meghil MM, Cutler CW (2022) Dendritic cells a critical link to alveolar bone loss and systemic disease risk in periodontitis: Immunotherapeutic implications. Periodontol 2000 89:41–50. https://doi.org/10.1111/prd.12428
Cekici A, Kantarci A, Hasturk H, Van Dyke TE (2014) Inflammatory and immune pathways in the pathogenesis of periodontal disease. Periodontol 2000 64(1):57–80
Mundy GR (1991) Inflammatory mediators and the destruction of bone. J Periodontal Res 26(3 Pt 2):213–217. https://doi.org/10.1111/j.1600-0765.1991.tb01647.x
Schwartz Z, Goultschin J, Dean DD, Boyan BD (1997) Mechanisms of alveolar bone destruction in periodontitis. Periodontol 2000 14:158–172. https://doi.org/10.1111/j.1600-0757.1997.tb00196.x
Van Dyke TE (2017) Pro-resolving mediators in the regulation of periodontal disease. Mol Aspects Med 58:21–36. https://doi.org/10.1016/j.mam.2017.04.006
Horiuchi K, Amizuka N, Takeshita S, Takamatsu H, Katsuura M, Ozawa H, Toyama Y, Bonewald LF, Kudo A (1999) Identification and characterization of a novel protein, periostin, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor beta. J Bone Miner Res 14(7):1239–1249. https://doi.org/10.1359/jbmr.1999.14.7.1239
Du J, Li M (2017) Functions of Periostin in dental tissues and its role in periodontal tissues’ regeneration. Cell Mol Life Sci 74(23):4279–4286. https://doi.org/10.1007/s00018-017-2645-3
Duchamp de Lageneste O, Colnot C (2019) Periostin in bone regeneration. Adv Exp Med Biol 1132:49–61. https://doi.org/10.1007/978-981-13-6657-4_6
Padial-Molina M, Volk SL, Rios HF (2014) Periostin increases migration and proliferation of human periodontal ligament fibroblasts challenged by tumor necrosis factor -alpha and Porphyromonas gingivalis lipopolysaccharides. J Periodontal Res 49:405–414. https://doi.org/10.1111/jre.12120
Matsuzawa M, Arai C, Nomura Y, Murata T, Yamakoshi Y, Oida S, Hanada N, Nakamura Y (2015) Periostin of human periodontal ligament fibroblasts promotes migration of human mesenchymal stem cell through the alphavbeta3 integrin/FAK/PI3K/Akt pathway. J Periodontal Res 50:855–863. https://doi.org/10.1111/jre.12277
Watanabe T, Yasue A, Fujihara S, Tanaka E (2012) PERIOSTIN regulates MMP-2 expression via the alphavbeta3 integrin/ERK pathway in human periodontal ligament cells. Arch Oral Biol 57:52–59. https://doi.org/10.1016/j.archoralbio.2011.07.010
Izuhara K, Nunomura S, Nanri Y, Ogawa M, Ono J, Mitamura Y, Yoshihara T (2017) Periostin in inflammation and allergy. Cell Mol Life Sci 74(23):4293–4303. https://doi.org/10.1007/s00018-017-2648-0
Arslan R, Karsiyaka Hendek M, Kisa U, Olgun E (2021) The effect of non-surgical periodontal treatment on gingival crevicular fluid periostin levels in patients with gingivitis and periodontitis. Oral Dis 27(6):1478–1486. https://doi.org/10.1111/odi.13664
Zundler S, Neurath MF (2015) Interleukin-12: Functional activities and implications for disease. Cytokine Growth Factor Rev 26(5):559–568. https://doi.org/10.1016/j.cytogfr.2015.07.003
Wesa AK, Galy A (2001) IL-1 beta induces dendritic cells to produce IL-12. Int Immunol 13(8):1053–1061. https://doi.org/10.1093/intimm/13.8.1053
Tsai IS, Tsai CC, Ho YP, Ho KY, Wu YM, Hung CC (2005) Interleukin-12 and interleukin-16 in periodontal disease. Cytokine 31(1):34–40. https://doi.org/10.1016/j.cyto.2005.02.007
Wang X, Wei Y, Xiao H, Liu X, Zhang Y, Han G, Chen G, Hou C, Ma N, Shen B et al. A novel IL-23p19/Ebi3 (IL-39) cytokine mediates inflammation in Lupus-like mice. Eur J Immunol 46(6):1343–1350. https://doi.org/10.1002/eji.201546095
Luo Y, Liu F, Liu H, Chen H, Cheng W, Dong S, Xiong W (2017) Elevated serum IL-39 in patients with ST-segment elevation myocardial infarction was related with left ventricular systolic dysfunction. Biomark Med 11(6):419–426. https://doi.org/10.2217/bmm-2016-0361
Lu Z, Xu K, Wang X, Li Y, Li M (2020) Interleukin 39: a new member of interleukin 12 family. Cent Eur J Immunol 45(2):214–217. https://doi.org/10.5114/ceji.2020.97911
Silness J, Loe H (1964) Periodontal disease in pregnancy. Ii. Correlation between Oral Hygiene and Periodontal Condtion. Acta Odontol Scand 22:121–135. https://doi.org/10.3109/00016356408993968
Loe H, Silness J (1963) Periodontal Disease in pregnancy. I Prevalence and Severity. Acta Odontol Scand 21:533–551. https://doi.org/10.3109/00016356309011240
Ainamo J, Bay I (1975) Problems and proposals for recording gingivitis and plaque. Int Dent J 25(4):229–235
Caton JG, Armitage G, Berglundh T, Chapple ILC, Jepsen S, Kornman KS, Mealey BL, Papapanou PN, Sanz M, Tonetti MS (2018) A new classification scheme for periodontal and peri-implant diseases and conditions—introduction and key changes from the 1999 classification. J Periodontol 89(Suppl 1):S1–S8. https://doi.org/10.1002/JPER.18-0157
Tonetti MS, Sanz M (2019) Implementation of the new classification of periodontal diseases: decision-making algorithms for clinical practice and education. J Clin Periodontol 46(4):398–405. https://doi.org/10.1111/jcpe.13104
Trombelli L, Farina R, Silva CO, Tatakis DN (2018) Plaque-induced gingivitis: case definition and diagnostic considerations. J Clin Periodontol 45(Suppl 20):S44–S67. https://doi.org/10.1111/jcpe.12939
Tonetti MS, Greenwell H, Kornman KS (2018) Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J Clin Periodontol 45(Suppl 20):S149–S161. https://doi.org/10.1111/jcpe.12945
Akalin FA, Baltacioglu E, Alver A, Karabulut E (2007) Lipid peroxidation levels and total oxidant status in serum, saliva and gingival crevicular fluid in patients with chronic periodontitis. J Clin Periodontol 34(7):558–565. https://doi.org/10.1111/j.1600-051X.2007.01091.x
Rudin HJ, Overdiek HF, Rateitschak KH (1970) Correlation between sulcus fluid rate and clinical and histological inflammation of the marginal gingiva. Helv Odontol Acta 14(1):21–26
Chapple IL, Landini G, Griffiths GS, Patel NC, Ward RS (1999) Calibration of the Periotron 8000 and 6000 by polynomial regression. J Periodontal Res 34(2):79–86. https://doi.org/10.1111/j.1600-0765.1999.tb02226.x
Chapple IL, Mason GI, Garner I, Matthews JB, Thorpe GH, Maxwell SR, Whitehead TP (1997) Enhanced chemiluminescent assay for measuring the total antioxidant capacity of serum, saliva and crevicular fluid. Ann Clin Biochem 34(Pt 4):412–421. https://doi.org/10.1177/000456329703400413
Lamster IB, Oshrain RL, Gordon JM (1986) Enzyme activity in human gingival crevicular fluid: consideration in data reporting based on analysis of individual crevicular sites. J Clin Periodontol 13(8):799–804
Tsai CC, Ho YP, Chen CC (1995) Levels of interleukin-1 beta and interleukin-8 in gingival crevicular fluids in adult periodontitis. J Periodontol 66:852–859. https://doi.org/10.1902/jop.1995.66.10.852
Chapple IL, Matthews JB, Thorpe GH, Glenwright HD, Smith JM, Saxby MS (1993) A new ultrasensitive chemiluminescent assay for the site-specific quantification of alkaline phosphatase in gingival crevicular fluid. J Periodontal Res 28:266–273. https://doi.org/10.1111/j.1600-0765.1993.tb02093.x
Ikezawa I, Tai H, Shimada Y, Komatsu Y, Galicia JC, Yoshie H (2005) Imbalance between soluble tumour necrosis factor receptors type 1 and 2 in chronic periodontitis. J Clin Periodontol 32:1047–1054. https://doi.org/10.1111/j.1600-051X.2005.00832.x
Giannopoulou C, Cappuyns I, Mombelli A (2003) Effect of smoking on gingival crevicular fluid cytokine profile during experimental gingivitis. J Clin Periodontol 30:996–1002. https://doi.org/10.1034/j.1600-051x.2003.00416.x
Wang X, Liu X, Zhang Y, Wang Z, Zhu G, Han G, Chen G, Hou C, Wang T, Ma N, Shen B, Li Y, Xiao H, Wang R (2016) Interleukin (IL)-39 [IL-23p19/Epstein-Barr virus-induced 3 (Ebi3)] induces differentiation/expansion of neutrophils in lupus-prone mice. Clin Exp Immunol 186:144–156. https://doi.org/10.1111/cei.12840
Ecoeur F, Weiss J, Schleeger S, Guntermann C (2020) Lack of evidence for expression and function of IL-39 in human immune cells. PLoS One 15(12):e0242329. https://doi.org/10.1371/journal.pone.0242329
Wang X, Zhang Y, Wang Z, Liu X, Zhu G, Han G, Chen G, Hou C, Wang T, Shen B et al (2018) AntiIL39 (IL23p19/Ebi3) polyclonal antibodies ameliorate autoimmune symptoms in lupuslike mice. Mol Med Rep 17(1):1660–1666. https://doi.org/10.3892/mmr.2017.8048
Manning AA, Zhao L, Zhu Z, Xiao H, Redington CG, Ding VA, Stewart-Hester T, Bai Q, Dunlap J, Wakefield MR et al (2018) IL-39 acts as a friend to pancreatic cancer. Med Oncol 36(1):12. https://doi.org/10.1007/s12032-018-1236-y
Yang MG, Tian S, Zhang Q, Han J, Liu C, Zhou Y, Zhu J, Jin T (2020) Elevated serum interleukin-39 levels in patients with neuromyelitis optica spectrum disorders correlated with disease severity. Mult Scler Relat Disord 46:102430. https://doi.org/10.1016/j.msard.2020.102430
Bridgewood C, Alase A, Watad A, Wittmann M, Cuthbert R, McGonagle D (2019) The IL-23p19/EBI3 heterodimeric cytokine termed IL-39 remains a theoretical cytokine in man. Inflamm Res 68(6):423–426. https://doi.org/10.1007/s00011-019-01235-x
Ohshima M, Otsuka K, Suzuki K (1994) Interleukin-1 beta stimulates collagenase production by cultured human periodontal ligament fibroblasts. J Periodontal Res 29:421–429. https://doi.org/10.1111/j.1600-0765.1994.tb01244.x
Dinarello CA (1991) Interleukin-1 and interleukin-1 antagonism. Blood 77:1627–1652
Orozco A, Gemmell E, Bickel M, Seymour GJ (2006) Interleukin-1beta, interleukin-12 and interleukin-18 levels in gingival fluid and serum of patients with gingivitis and periodontitis. Oral Microbiol Immunol 21:256–260. https://doi.org/10.1111/j.1399-302X.2006.00292.x
Masada MP, Persson R, Kenney JS, Lee SW, Page RC, Allison AC (1990) Measurement of interleukin-1 alpha and -1 beta in gingival crevicular fluid: implications for the pathogenesis of periodontal disease. J Periodontal Res 25:156–163. https://doi.org/10.1111/j.1600-0765.1990.tb01038.x
Ebersole JL, Nagarajan R, Akers D, Miller CS (2015) Targeted salivary biomarkers for discrimination of periodontal health and disease(s). Front Cell Infect Microbiol 5:62. https://doi.org/10.3389/fcimb.2015.00062
Kc S, Wang XZ, Gallagher JE (2020) Diagnostic sensitivity and specificity of host-derived salivary biomarkers in periodontal disease amongst adults: systematic review. J Clin Periodontol 47:289–308. https://doi.org/10.1111/jcpe.13218
Hunter CA, Chizzonite R, Remington JS (1995) IL-1 beta is required for IL-12 to induce production of IFN-gamma by NK cells. A role for IL-1 beta in the T cell-independent mechanism of resistance against intracellular pathogens. J Immunol 155:4347–4354
Suzuki H, Amizuka N, Kii I, Kawano Y, Nozawa-Inoue K, Suzuki A, Yoshie H, Kudo A, Maeda T (2004) Immunohistochemical localization of periostin in tooth and its surrounding tissues in mouse mandibles during development. Anat Rec A Discov Mol Cell Evol Biol 281:1264–1275. https://doi.org/10.1002/ar.a.20080
Park CH, Rios HF, Taut AD, Padial-Molina M, Flanagan CL, Pilipchuk SP, Hollister SJ, Giannobile WV (2014) Image-based, fiber guiding scaffolds: a platform for regenerating tissue interfaces. Tissue Eng Part C Methods 20:533–542. https://doi.org/10.1089/ten.TEC.2013.0619
Padial-Molina M, Volk SL, Rodriguez JC, Marchesan JT, Galindo-Moreno P, Rios HF (2013) Tumor necrosis factor-alpha and Porphyromonas gingivalis lipopolysaccharides decrease periostin in human periodontal ligament fibroblasts. J Periodontol 84:694–703. https://doi.org/10.1902/jop.2012.120078
Kumaresan D, Balasundaram A, Naik VK, Appukuttan DP (2016) Gingival crevicular fluid periostin levels in chronic periodontitis patients following nonsurgical periodontal treatment with low-level laser therapy. Eur J Dent 10:546–550. https://doi.org/10.4103/1305-7456.195179
Aral CA, Koseoglu S, Saglam M, Pekbagriyanik T, Savran L (2016) Gingival crevicular fluid and salivary periostin levels in non-smoker subjects with chronic and aggressive periodontitis: periostin levels in chronic and aggressive periodontitis. Inflammation 39:986–993. https://doi.org/10.1007/s10753-016-0328-0
Prakoura N, Chatziantoniou C (2017) Periostin in kidney diseases. Cell Mol Life Sci 74:4315–4320. https://doi.org/10.1007/s00018-017-2650-6
Ding Y, Ge Q, Qu H, Feng Z, Long J, Wei Q, Zhou Q, Wu R, Yao L, Deng H (2018) Increased serum periostin concentrations are associated with the presence of diabetic retinopathy in patients with type 2 diabetes mellitus. J Endocrinol Invest 41:937–945. https://doi.org/10.1007/s40618-017-0820-x
Prakoura N, Kavvadas P, Kormann R, Dussaule JC, Chadjichristos CE, Chatziantoniou C (2017) NFkappaB-induced periostin activates integrin-beta3 signaling to promote renal injury in GN. J Am Soc Nephrol 28:1475–1490. https://doi.org/10.1681/ASN.2016070709
Baeza M, Garrido M, Hernandez-Rios P, Dezerega A, Garcia-Sesnich J, Strauss F, Aitken JP, Lesaffre E, Vanbelle S, Gamonal J, Brignardello-Petersen R, Tervahartiala T, Sorsa T, Hernandez M (2016) Diagnostic accuracy for apical and chronic periodontitis biomarkers in gingival crevicular fluid: an exploratory study. J Clin Periodontol 43:34–45. https://doi.org/10.1111/jcpe.12479
Engebretson SP, Grbic JT, Singer R, Lamster IB (2002) GCF IL-1beta profiles in periodontal disease. J Clin Periodontol 29:48–53. https://doi.org/10.1034/j.1600-051x.2002.290108.x
Nishida N, Tanaka M, Hayashi N, Nagata H, Takeshita T, Nakayama K, Morimoto K, Shizukuishi S (2005) Determination of smoking and obesity as periodontitis risks using the classification and regression tree method. J Periodontol 76:923–928. https://doi.org/10.1902/jop.2005.76.6.923
Brock GR, Butterworth CJ, Matthews JB, Chapple IL (2004) Local and systemic total antioxidant capacity in periodontitis and health. J Clin Periodontol 31:515–521. https://doi.org/10.1111/j.1600-051X.2004.00509.x
Acknowledgements
The authors would like to thank Arzu BAYGUL, Asistant Prof., from Koc University Medical Faculty Biostatistics Department for carrying out statistical analyses of the study.
Funding
The present study was self-funded by the authors.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Deciding the material and method were performed by Aysegul SARI, Serhat KOSEOGLU, and Serdar DOGAN. Data collection was performed by Ayşegul SARI. Carrying out biochemical analyses by Serdar DOGAN. The first draft of the manuscript was written by Aysegul SARI, Serhat KOSEOGLU, and Serdar DOGAN. Data interpretations and manuscript revisions were made by Luigi NIBALI, Aysegul SARI, and Serhat KOSEOGLU. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study protocol was approved by the Ethics Committee for the use of Human Subjects in Research, Hatay Mustafa Kemal University, Hatay, Turkey (Protocol No. 2021/70) and the study was carried out pursuant to the Declaration of Helsinki.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sari, A., Dogan, S., Nibali, L. et al. Evaluation of IL-23p19/Ebi3 (IL-39) gingival crevicular fluid levels in periodontal health, gingivitis, and periodontitis. Clin Oral Invest 26, 7209–7218 (2022). https://doi.org/10.1007/s00784-022-04681-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04681-w