Abstract
Objectives
The aim was to investigate color, gloss, or roughness of splint materials after storage in liquids and toothbrush simulation.
Materials and methods
A total of 58 × 8 (n = 10 per material and group) specimens were fabricated (hand-cast, thermoforming, CAD/CAM-milled, 3D-printed materials); stored in air, water, coffee, red wine, and cleaning tablets; and investigated after fabrication, 24 h, two-, and four-week storage or toothbrushing. Color values (L*, a*, b*; ISO 11664–4:2008; CM–3500d, Konica-Minolta), gloss (ISO 2813:2014), and roughness values were determined (3D laser-scanning-microscope, KJ 3D, Keyence) before and after simulation or storage. Statistics: Levene-test, one-way ANOVA, Bonferroni post hoc test, between-subjects effects, Pearson correlation (α = 0.05).
Results
Color, gloss, and roughness altered due to contact with staining solutions/toothbrush simulation. Highest impact on color, gloss, and roughness presented the material followed by storage time (ΔE material (η2 = 0.239/p < 0.001), storage time (η2 = 0.179/p < 0.001); gloss (η2 = 0.751/p < 0.001) (η2 = 0.401/p < 0.001); Ra/Rz (η2 ≥ 0.801/p < 0.001) (η2 ≥ 0.416/p < 0.001)). Correlations were found between Rz and Ra (Pearson 0.887/p ≤ 0.001) or Rz and ΔE (0.517/p ≤ 0.001) or Ra and ΔE (0.460/p ≤ 0.001).
Conclusions
Storage and toothbrushing were accompanied by a change in color, gloss, and roughness. Almost all materials showed visible discoloration after 4 weeks of storage. Gloss values decreased as storage time increased. The initial roughness and polishability were better with harder materials.
Clinical relevance.
Milled and 3D printed splints show good color, gloss, and roughness resistance after 4-week storage or toothbrush application.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Splints are an effective therapeutic treatment of temporomandibular disorders (TMD) [1]. The appliances improve individual symptoms such as pain and functional limitations [2,3,4]. It is state of the art to fabricate splints on gypsum models either by applying methacrylate in the sprinkle-on technique or by vacuum thermoforming [5]. Both techniques can be combined by adjusting the occlusal surface of a thermoformed splint with acrylic resin [6, 7]. The computer-aided design/computer-aided manufacturing (CAD/CAM) opened up new possibilities for splint production [8]. The clinical situation is either recorded directly with an intraoral scanner or impressions/plaster models are scanned [9]. Based on this digital impression, the occlusal devices can be designed with a CAD software [10]. In the subtractive process, the splints are milled from a prefabricated resin-based blank using a computerized numerical control (CNC) machine [11]. A more recent approach is 3D printing with stereolithography (SLA) or digital light processing (DLP) technology [12, 13]. Here, the splints are built up and cured layer by layer by a liquid photopolymer. The mechanical properties are affected by the type of material and the processing. Post-polymerization has an important role to play in ensuring the properties of the material [14, 15]. The success of a splint treatment depends to a large extent on the patient’s compliance. Therefore, in addition to the mechanical requirements, splints should also meet esthetic [16, 17], phonetic, and functional [18] demands. Basic esthetic requirements include color stability and surface gloss [19]. Color changes can be caused by intrinsic and extrinsic factors [20] and are therefore influenced by the chemical structure and the surface of the splint. Many studies have shown that the contact of resins with various staining liquids such as coffee, red wine, and mouthwashes leads to color changes [21,22,23]. Color changes of ΔE < 3.3 were generally considered as a threshold and classified as clinical acceptable [24]. The gloss of the splint depends on the surface roughness and the polish. The American Dental Association (ADA) considers gloss values between 40 and 60 to be desirable [25]. Alternative literature sources define gloss values \(\le\) 60 as poor finish and values between 70 and 80 as good [26,27,28]. Thus, the surface roughness plays an important role in the color behavior and gloss of the resins [29, 30], as well as in the accumulation of plaque and discoloring particles [31, 32]. Roughening caused by toothbrush can alter gloss and affect color stability [31, 33, 34].
The aim of the study was to investigate the effects of storage in different coloring liquids (water, coffee, red wine, and denture cleaner solution) and toothbrush simulation on color, gloss, or surface roughness of splint materials. It is expected that color, gloss, and surface roughness would change due to enduring contact with the staining solutions and toothbrush simulation. The null hypothesis was that the changes would depend on the material/fabrication, type of storage, and on the duration.
Materials and methods
A total of 58 × 8 (n = 10 per material and group) specimens (diameter 10 mm, thickness 2 mm) were fabricated from hand-cast, thermoforming, CAD/CAM-milled, and 3D-printed materials (Tables 1 and 2). Hand-cast specimens (Palapress vario transparent, Kulzer, Hanau, Germany, mixing ratio 10 g powder, 7 ml liquid) were poured in silicon (VPS Hydro Putty, Henry Schein, Langen, Germany) mold and polymerized in a pressure pot (55 °C and 2 bar). Thermoforming of clear foils (Erkodur, 2.00 mm, ∅ 120 mm; Erkodent, Pfalzgrafenweiler, Germany) was performed with Erkoform-3D Motion (Erkodent, Pfalzgrafenweiler, Germany). Specimens were milled from PMMA blanks (Optimill crystal clear; Dentona, Dortmund, Germany) with Zenotec select ion (Wieland Dental + Technik, Pforzheim, Germany). 3D printing job was created with the slicing software (Netfabb, Autodesk, San Rafael, USA; print direction: 90° to the building platform; support structures were used; layer thickness 50 µm). The materials LuxaPrint Ortho Plus (DMG, Hamburg, Germany) and KeySplint Soft (Keystone Industries, Gibbstown, NY, USA) were processed with the printer “P30 + ” (Straumann, Cares P series, Basel, Switzerland). Post-processing consisted of an automated wash cycle (P Wash, Straumann, Cares P series Basel, Switzerland) and LED post-polymerization (P Cure, Straumann, Cares P series Basel, Switzerland). The materials V-Print splint and Splint Flex (Voco, Cuxhaven, Germany) were printed with Solflex 650 (Voco, Cuxhaven, Germany). Specimens were manually cleaned (2 min isopropanol bath and ultrasonic) and post-polymerized with xenon light (Otoflash G171: 2000 flashes, 2 min cooling, 2000 flashes; NK Optik, Baierbrunn, Germany). All supports and protrusions were removed with burrs and sandpaper. Polishing was performed with a finishing buff and polishing paste (Polishing unit: WP-Ex 2000 II; Wassermann, Hamburg, Germany). Finally, the discs were cleaned in an ultrasonic bath (35 °C, 10 min, Sonorex super RK 102 H, Bandelin electronic, Berlin, Germany).
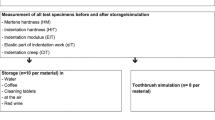
Specimens were stored in water (demineralized water), coffee (Cafet, Netto, Germany – instant coffee mild), red wine (Red wine sweet, Vino d’Italia, Italy), and cleaning tablets (Kukident – active plus, Kukident, Germany) in microwell plates. Solvents were renewed during the immersion test every 4 days. One disc was stored in 1 ml of test liquid. After storage, specimens were rinsed with water and carefully cleaned with a microfiber cloth. Specimens were investigated straight after fabrication (baseline), after 24 h, two-, and four-week storage. Specimens that were stored in air served as reference. Tooth brushing was performed with a toothbrush simulator (ZM-3; SD Mechatronik, Feldkirchen-Westerham, Germany; brush, Oral-B 1–2-3 indicator medium (35 mm), Oral B, Germany; slurry, 250 g toothpaste in 1 l demineralized water; load, 250 g, circular 10 mm movement, v = 40 mm/s, 72,000 cycles) on 8 specimens per material. Color, gloss, and roughness values were determined before and after the simulation (Fig. 1).
Color measurements were carried out using a spectrophotometer (CM–3500d, Konica-Minolta, Chiyoda, Japan) with black background. The measured color values (L*, a*, b*) were evaluated using the CIELAB system according to ISO 11664–4:2008 [35]. Color changes (ΔE) were calculated. A gloss meter (ZGM, Zehntner Testing, Sissach, Switzerland) was used to measure gloss (G) before and after immersion (angle 60°) according to ISO 2813:2014 [36]. Non-contact optical roughness values ((ISO 4287); Ra, Rz) were determined using a confocal 3D laser-scanning-microscope (KJ 3D, Keyence, Osaka, Japan; scanning area, 2400 × 1800 µm, λC = 0.8 mm). The arithmetic roughness Ra is the average of the absolute values along the single measuring section. The maximum height of the profile (maximum roughness; Rz) describes the absolute vertical distance between the maximum profile peak height and the maximum profile.
Calculations and statistical analysis were performed using SPSS 25.0 for Windows (IBM, Armonk, NY, USA). Homogeneity of the data was controlled with Shapiro–Wilk test. Means and standard deviations were calculated and analyzed using one-way analysis of variance and the Bonferroni test post hoc analysis. Between-subjects effects were investigated. The level of significance was set to α = 0.05. Pearson correlation between the individual parameters was determined.
Results
ΔE changes
Highest impact on ΔE presented the material (η2 = 0.239/p < 0.001), followed by storage time (η2 = 0.179/p < 0.001) and storage conditions and all other combinations (0.059 ≤ η2 ≤ 0.074/p < 0.001) (Fig. 2).
LuxaPrint Ortho Plus showed ΔE values between 0.69 (4 weeks air) and 2.82 (4 weeks coffee). No significant differences were found after different storage times (Anova, p ≥ 0.190; Bonferroni, p ≥ 0.329). Optimill crystal clear provided significant general differences (Anova p ≤ 0.028) and individual differences (p ≤ 0.031) after 4-week storage for coffee, mouth rinse, and air storage. ΔE varied between 0.88 (24 h mouth rinse) and 5.31 (4 weeks coffee). KeySplint Soft showed ΔE results between 2.11 (24 h coffee) and 7.06 (4 weeks coffee). Significant general differences (Anova p ≤ 0.011) and individual differences (p ≤ 0.032) after 2 weeks storage for coffee, mouth rinse, and red wine storage could be determined. Palapress vario provided significant general differences (Anova p ≤ 0.004) and individual differences (p ≤ 0.043) after 2 weeks storage for coffee and red wine as well as 4 weeks in water (p = 0.003). ΔE values between 0.87 (24 h red wine) and 4.45 (4 weeks coffee) were found. Erkodur showed ΔE results between 0.57 (24 h air) and 6.49 (4 weeks coffee). Significant general differences (Anova p ≤ 0.001) and individual differences (p < 0.001) after 4 weeks storage for coffee and in water (p < 0.001) could be determined. ΔE values for Splint Flex were between 0.97 (24 h water) and 6.87 (4 weeks coffee). Significant general differences (Anova p ≤ 0.014) and individual differences (p ≤ 0.038) were found after 4 weeks storage for coffee and red wine. V-Print splint provided no significant general differences (Anova p = 0.117) and individual differences (p < 0.001) only in air. Changes were significant after 2 weeks for storage in coffee and in water (p ≤ 0.019) and after 4 weeks (p ≤ 0.006). ΔE results between 1.04 (24 h red wine) and 3.60 (4 weeks coffee) were found.
Toothbrush abrasion provided ΔE values between 3.26 (KeySplint soft) and 20.95 (Erkodur) with significant differences between the groups (Anova p ≤ 0.001). Individual differences after toothbrush abrasion were found for LuxaPrint Ortho Plus and KeySplint Soft or Erkodur (p ≤ 0.001); Optimill crystal clear and KeySplint Soft, Erkodur, Splint Flex, or V-Print splint (p ≤ 0.010); KeySplint Soft and Palapress vario or Erkodur (p ≤ 0.004); Palapress vario and Erkodur (p = 0.000); and Erkodur and Splint Flex or V-Print splint (p ≤ 0.001).
Gloss
Highest impact on gloss presented the material (η2 = 0.751/p < 0.001), followed by storage time (η2 = 0.401/p < 0.001). Storage conditions and all other combinations besides material*storage time (η2 = 0.043/p < 0.001) provided moderate impact (0.253 ≤ η2 ≤ 0.077/p < 0.001). The materials showed significantly (Anova p < 0.001) different baseline gloss values (Fig. 3).
LuxaPrint Ortho Plus showed gloss units (GU) between 62 (4 weeks water) and 114 (baseline). Significant differences were found after 2 weeks in coffee, mouth rinse, red wine, or air and after 4 weeks in water (Anova p ≤ 0.005, Bonferroni p ≤ 0.039). Optimill crystal clear provided significant general differences (Anova p ≤ 0.004) and individual differences (p ≤ 0.024) after 2 weeks storage in coffee, water, or red wine and after 2 h in mouth rinse and air. Gloss varied between 75 GU (4 weeks coffee) and 117 GU (baseline). KeySplint Soft showed gloss values between 12 GU (24 h red wine) and 51 GU (baseline). Significant general differences (Anova p ≤ 0.0.01) and individual differences (p ≤ 0.031) after 24 h storage for coffee, mouth rinse, red wine storage, or air and 2 weeks in water could be determined. Palapress vario provided significant general differences (Anova p ≤ 0.026) and individual differences (p ≤ 0.023) after 2 weeks storage in red wine and 4 weeks in coffee or mouth rinse. Gloss values between 64 GU (4 weeks red wine) and 86 GU (baseline) were found. Erkodur provided gloss results between 52 GU (4 weeks coffee) and 116 GU (baseline). Erkodur showed significant general differences (Anova p ≤ 0.001) and individual differences (p ≤ 0.039) after 24 h storage in coffee or water and 2 weeks in mouth rinse, red wine, or air. Gloss for Splint Flex varied between 59 GU (2 weeks coffee) and 92 GU (24 h red wine). Only general significant differences (Anova p = 0.007) were found for coffee storage. V-Print splint provided no significant general differences (Anova p ≥ 0.435) nor individual differences (p < 0.001) only in air. Changes were significant after 2 weeks for storage for coffee and in water (p ≤ 0.019) and after 4 weeks (p ≤ 0.006). Gloss varied between 78 GU (4 weeks red wine) and 101 GU (baseline) were found.
Gloss changes due to toothbrush abrasion: Highest gloss was found for Optimill crystal clear (144 baseline) and lowest for Erkodur (12 after toothbrush abrasion) with significant differences between the groups (Anova p ≤ 0.001). Toothbrush treatment significantly (p ≤ 0.024) influenced gloss for all materials, but KeySplint Soft (p = 0.061). Clearest influence of toothbrush abrasion was found for Erkodur with a gloss change of 85 units. Smallest influence was provided by KeySplint Soft with a gloss change of 9 units.
Roughness
Highest impact on Ra and Rz (η2 ≥ 0.801/p < 0.001) presented the material, followed by storage time (η2 ≥ 0.416/p < 0.001) and their combinations (η2 ≥ 0.317/p < 0.001). The other combinations showed lower, but significant impact (0.072 ≤ η2 ≤ 0.271/p < 0.001). Both Rz and Ra provided significant (p ≤ 0.001) differences for the materials already in the baseline measurement (Fig. 4).
Significant Rz differences were found for LuxaPrint Ortho Plus after 4 weeks in coffee (p = 0.006). Optimill crystal clear provided significant differences (p = 0.001) after two 2 weeks coffee and 4 weeks mouth rinse, water, or red wine (p ≤ 0.001). KeySplint Soft showed significant (p = 0.000) differences after 4 weeks coffee and after 2 weeks red wine (p = 0.009). Significant (p ≤ 0.006) differences were found for Palapress vario after 2 weeks coffee or red wine and after 4 weeks water (p = 0.005). Erkodur provided significant (p = 0.008) differences after 4 weeks coffee and 24 h mouth rinse (P = 0.001). For Splint Flex, significant (p ≤ 0.021) differences could be determined after 2 weeks coffee, water, red wine, or air and after weeks mouth rinse (p = 0.004). V-Print splint showed significant (p ≤ 0.013) differences after 2 weeks coffee, mouth rinse, water, or red wine.
LuxaPrint Ortho Plus provided significant (p ≤ 0.035) Ra changes after 2 weeks coffee or mouth rinse. Significant changes were found for Optimill crystal clear after 24 h coffee (p = 0.014) and 2 weeks mouth rinse, water, or red wine (p ≤ 0.001). KeySplint Soft showed significant (p ≤ 0.046) changes after 2 weeks coffee, mouth rinse, or water and 24 h in red wine or air. Palapress vario provide significant (p ≤ 0.013) differences after 24 h in coffee, water, or red wine and 2 weeks mouth rinse (p = 0.020). For Erkodur, significant (p ≤ 0.018) changes were found after 24 h mouth rinse, water, or red wine and 4 weeks air (p = 0.048). Splint Flex provided significant (p ≤ 0.001) changes after 24 h water, red wine, or air and 2 weeks coffee. For V-Print splint, the changes were significant (p ≤ 0.007) after 24 h coffee, mouth rinse, water, or red wine.
Toothbrush treatment significantly influenced Rz (p ≤ 0.002) for all materials. Ra results were significantly (p ≤ 0.018) reduced for all materials but Optimill crystal clear (p = 0.327).
Significant correlations were found between Rz and Ra (Pearson 0.887/p ≤ 0.001) or Rz and ΔE (0.517/p ≤ 0.001) or Ra and ΔE (0.460/p ≤ 0.001). Significant negative correlations could be determined for gloss and Rz (− 0.714/p ≤ 0.001), Ra (− 0.712/p ≤ 0.001), and ΔE (− 0.558/p ≤ 0.001). p values of the Shapiro–Wilk normal distribution varied between 0.052 and 0.970 (color), 0.059 and 0.991 (gloss), 0.50 and 0.935 (roughness Ra), and 0.055 and 1.000 (roughness Rz) (Fig. 5).
Discussion
The hypothesis of this in vitro study could be confirmed. Color, gloss, and surface roughness altered due to contact with staining solutions/toothbrush simulation and the changes were dependent on the individual material, the type, and duration of storage. There were no clear differences between the material groups. Influences on other properties such as hardness or polymerization [37,38,39], which were found for other printed materials, should be investigated in further tests and compared with the current data.
Color
Different materials responded differently to the individual storage solutions. After 4 weeks at the latest, the ΔE values of all materials except LuxaPrint Ortho Plus reached or exceeded the acceptable ΔE value of 3.3 [24] in at least one of the storage media. The highest discoloration was obtained for specimens stored in coffee, followed by red wine, cleaning tablets, and demineralized water. Previous studies showing that coffee causes the most color changes in resins compared to other staining solutions could be confirmed [30, 40]. This was explained by the fact that coffee contains yellow colorants with different polarities. Yellow dyes could be absorbed into the organic phase through their compatibility with the polymer phase of the resin [30, 41]. Some materials might also show stronger discoloration as a result of a higher water absorption capacity [42]. The fact that the discoloration of most materials in air is much lower than in systems with moisture storage may confirm this assumption. The hypothesis that water is crucial in comparison to staining is also supported by the fact that the discoloration in water was comparable to storage in the other media—except coffee. However, since changes can also be observed for the soft printing systems during storage at air, incomplete polymerization [43, 44], and chemical reactivity could be reasons for the discoloration [23, 23, 23, 45, 46] of these materials. It is not only the resin composition, but also the conversion that seems important. Therefore, not only the fabrication but also the post-processing (cleaning, polymerization) may influence the color stability, as well as other properties such as the flexural strength. The results showed that the best color stability can be achieved with both milled and printed materials. The comparison with flexible soft materials indicated that they do not necessarily perform worse than conventional, firmer printing, thermoforming, or hand-cast materials. Significant correlations can be found between color change and roughness values, indicating that a decisive factor for color stability is a smooth surface and thus an excellent polish. The rougher surfaces were therefore probably more susceptible to discoloration [47,48,49,50]. Also, toothbrush abrasion demonstrated the influence of surface roughness on color behavior. After toothbrush simulation, all materials except KeySplint Soft achieved significantly higher color values. An explanation for this effect is the increasing surface roughness, influencing the superficial reflection [51].
Gloss
Considering that good polishing resulted in gloss values between 70 and 80 GU [26,27,28], all the examined materials, except for KeySplint Soft, had good baseline gloss values. The reduced polishability may be due to the lower hardness of the material. For the materials Luxaprint, Optimill, and Erkodur, even excellent gloss values of over 100 could be achieved. Due to the variety of each material, it was difficult to standardize the surface of the test specimens under laboratory conditions [52]. Gloss values are inversely proportional to color changes: Gloss values of all investigated resins decreased by immersion in the test solutions and with increasing storage time. There was a moderate impact from the type of test solution, while no changes were observed when stored at air. The reason for the reduction of gloss due to storage in the test solutions could be an increase in the surface roughness and thus a larger exposed surface area or an effect of discoloration. More likely, however, is a loss of gloss caused by water absorption on the material [2, 53]. This is supported by the fact that the gloss values in humid ambient conditions differed only slightly despite the different storage liquids, and that storage in air had a more pronounced effect on the gloss values. Toothbrush abrasion resulted in a significant reduction in gloss for all materials. Significant negative correlations can be found for gloss, roughness, and color values. KeySplint Soft is an exception here and both Voco materials (Splint Flex, V-Print splint) also show less decrease of gloss. This might be attributed to identical monomer or filler components. The reduction in gloss after toothbrushing is in most cases accompanied by increased surface roughness. The abrasion of the resin matrix and loss of surface particles could have caused changes in the surface topography [28, 54]. Murakami et al. and Heintze et al. concluded that tooth brushing also leads to microscopic and macroscopic roughness. The result is a diffuse reflection of the incident light, accompanied by a reduction in gloss [55, 56]. It can be assumed that the composition of the toothpaste also has an influence on the loss of the surface gloss; e.g., with a more abrasive toothpaste, it is easier to create cavities or particles from the paste can be rubbed into the material surface [57].
Roughness
The variety of materials studied had the greatest influence on roughness: both roughness values already showed significant differences for the materials in the initial measurement. Softer materials were rougher. One possible explanation could be that materials with lower surface hardness are more susceptible to scratches [27] and worse polishable with conventional means. After storage in the various test solutions, the materials can be divided into three groups: The soft materials (KeySplint Soft and Splint Flex) showed highest roughness, followed by Optimill crystal clear and Erkodur, as a third group with LuxaPrint Ortho Plus, Palapress vario, and V-Print splint achieved lowest roughness values.
The storage time also influenced the surface roughness. It can be assumed that water absorption reduces the hardness, which in turn increases the surface roughness [58, 59].
After toothbrush abrasion, the surface roughness increased significantly for all materials. An exception is the material KeySplint Soft, where the Rz-value even decreased after abrasion and the Ra-value only slightly increased. This is certainly due to the low hardness of the material. It is noticeable that the roughness values in this study are far above the threshold of clinical relevance (Ra < 0.2 µm) described earlier [60, 61]. Reference measurements with manual scanning prove that the values measured with a 3D laser-scanning microscope are higher by a power of ten. The different measurement setup and, for example, the different reflection of the specimen surface may be noticeable [62]. As the splint resins examined in the present study are typically transparent, surface reflections or absorption could have affected the measurements. Since the discs were measured at different storage times, the advantage that the surface remained undamaged by the optical measurement prevails. It can be assumed that the material hardness has an effect on roughness. Light-curing occlusal splint resins are expected to have comparable hardness as auto-polymerizing systems [63, 64], but hardness of 3D-printed occlusal splint materials is influenced by the print angle [17]. Martens hardness and indentation modulus depend on post-polymerization and are expected to decrease after water storage [39] [62].
The individual layers in transparent materials may affect neither the optical color nor gloss measurements, nor the roughness measurements. The present specimens were printed with a layer thickness of 50 µm in order to match surface quality and printing time [37, 65, 66]. Samples, which were printed with a lower (e.g., 25 µm) or adaptive layer thickness (inside 50 µm and outside 25 µm), might perform better, because an influence of the print parameters on the surface quality has been confirmed earlier.
The storage of the test specimens in various solutions and brushing with a toothbrush is accompanied by a change in color, gloss, and roughness. Discoloration and reduction of gloss usually leads to a loss of esthetic properties and thus limit the acceptance of the splint. Storage conditions such as pH in solution appear to be related to the hydrophilicity of the matrix and the chemical composition of the filler, which in turn affect sorption and solubility [67]. Greater surface roughness can cause bacteria and microorganisms to adhere more easily to the materials, leading to inflammation [60, 61]. If splints are to be used over a longer period of time, color- and gloss-resistant materials should therefore be preferred. A clinical consequence could be that occlusal splints are polished at regular intervals, which reduces the surface roughness [68, 69], removes extrinsic discoloration [70], and improves the gloss [68, 71]. In this in vitro study, the parameters were observed after 4 weeks of storage; however, it can be assumed that the values increase with increasing storage time [72, 73]. Photo-polymerization variables influence the structure and subsequent thermal response of dental resin matrices [74], and therefore, the degree of polymer polymerization or monomer release may be a key to the decrease in gloss and the increase in roughness and should be investigated in further studies. It is known that a combined heat and light-post-curing unit can improve the degree of conversion of 3D printed occlusal splints [75] and, e.g., hardness [38] or the in vitro performance [14, 19].
Conclusion
Almost all the materials tested showed visible discoloration after only 4 weeks of storage, especially in coffee. Gloss values decrease as storage time is increased, and the type of test solution has a moderate effect. The initial surface quality and polishability are better with harder materials. Despite low hardness, the soft materials do not become rougher due to storage.
Clinical consequence
An influence of the splint materials on the investigated parameters could not be proven: printed and even softer splints can also show good color, gloss, and roughness resistance after 4 week application. Color, gloss, and roughness correlate in some, but not all respects.
References
Riley P, Glenny A-M, Worthington HV et al (2020) Oral splints for temporomandibular disorder or bruxism: a systematic review. Br Dent J 228:191–197. https://doi.org/10.1038/s41415-020-1250-2
Alajbeg IZ, Gikić M, Valentić-Peruzović M (2014) Changes in pain intensity and oral health-related quality of life in patients with temporomandibular disorders during stabilization splint therapy–a pilot study. Acta Clin Croat 53:7–16
Vrbanović E, Alajbeg IZ (2019) Long-term effectiveness of occlusal splint therapy compared to placebo in patients with chronic temporomandibular disorders. Acta Stomatol Croat 53:195–206. https://doi.org/10.15644/asc53/3/1
Zhang C, Wu J-Y, Deng D-L et al (2016) Efficacy of splint therapy for the management of temporomandibular disorders: a meta-analysis. Oncotarget 7:84043–84053. https://doi.org/10.18632/oncotarget.13059
Kass CA, Tregaskes JN (1978) Occlusal splint fabrication. J Prosthet Dent 40:461–463. https://doi.org/10.1016/0022-3913(78)90133-6
Becker CM, Kaiser DA, Lemm RB (1974) A simplified technique for fabrication fo night guards. J Prosthet Dent 32:582–589. https://doi.org/10.1016/0022-3913(74)90015-8
Bohnenkamp DM (1996) Dimensional stability of occlusal splints. J Prosthet Dent 75:262–268. https://doi.org/10.1016/s0022-3913(96)90482-5
Miyazaki T, Hotta Y, Kunii J et al (2009) A review of dental CAD/CAM: current status and future perspectives from 20 years of experience. Dent Mater J 28:44–56. https://doi.org/10.4012/dmj.28.44
Tomita Y, Uechi J, Konno M et al (2018) Accuracy of digital models generated by conventional impression/plaster-model methods and intraoral scanning. Dent Mater J 37:628–633. https://doi.org/10.4012/dmj.2017-208
Lauren M, McIntyre F (2008) A new computer-assisted method for design and fabrication of occlusal splints. Am J Orthod Dentofacial Orthop 133:S130–S135. https://doi.org/10.1016/j.ajodo.2007.11.018
Lutz A-M, Hampe R, Roos M et al (2019) Fracture resistance and 2-body wear of 3-dimensional-printed occlusal devices. J Prosthet Dent 121:166–172. https://doi.org/10.1016/j.prosdent.2018.04.007
Kessler A, Hickel R, Reymus M (2020) 3D printing in dentistry-state of the art. Oper Dent 45:30–40. https://doi.org/10.2341/18-229-L
Marcel R, Reinhard H, Andreas K (2020) Accuracy of CAD/CAM-fabricated bite splints: milling vs 3D printing. Clin Oral Investig 24:4607–4615. https://doi.org/10.1007/s00784-020-03329-x
Wulff J, Schmid A, Huber C et al (2021) Dynamic fatigue of 3D-printed splint materials. J Mech Behav Biomed Mater 124:104885. https://doi.org/10.1016/j.jmbbm.2021.104885
Kim D, Shim J-S, Lee D et al. (2020) Effects of post-curing time on the mechanical and color properties of three-dimensional printed crown and bridge materials. Polymers (Basel) 12. https://doi.org/10.3390/polym12112762
Berli C, Thieringer FM, Sharma N et al (2020) Comparing the mechanical properties of pressed, milled, and 3D-printed resins for occlusal devices. J Prosthet Dent 124:780–786. https://doi.org/10.1016/j.prosdent.2019.10.024
Grymak A, Aarts JM, Ma S et al (2021) Comparison of hardness and polishability of various occlusal splint materials. J Mech Behav Biomed Mater 115:104270. https://doi.org/10.1016/j.jmbbm.2020.104270
Edelhoff D, Schweiger J, Prandtner O et al (2017) CAD/CAM splints for the functional and esthetic evaluation of newly defined occlusal dimensions. Quintessence Int 48:181–191. https://doi.org/10.3290/j.qi.a37641
Rosentritt M, Huber C, Strasser T et al (2021) Investigating the mechanical and optical properties of novel Urethandimethacrylate (UDMA) and Urethanmethacrylate (UMA) based rapid prototyping materials. Dent Mater. https://doi.org/10.1016/j.dental.2021.08.009
Kang A, Son S-A, Hur B et al (2012) The color stability of silorane- and methacrylate-based resin composites. Dent Mater J 31:879–884. https://doi.org/10.4012/dmj.2012-082
Leite ML, de Alencar E, Silva S, Sales FD, da Cunha ME, Meireles SS et al (2014) The effect of drinks on color stability and surface roughness of nanocomposites. Eur J Dent 8:330–336. https://doi.org/10.4103/1305-7456.137640
Guler AU, Yilmaz F, Kulunk T et al (2005) Effects of different drinks on stainability of resin composite provisional restorative materials. J Prosthet Dent 94:118–124. https://doi.org/10.1016/j.prosdent.2005.05.004
Ardu S, Duc O, Di Bella E et al (2017) Color stability of recent composite resins. Odontology 105:29–35. https://doi.org/10.1007/s10266-016-0234-9
Ruyter IE, Nilner K, Moller B (1987) Color stability of dental composite resin materials for crown and bridge veneers. Dent Mater 3:246–251. https://doi.org/10.1016/S0109-5641(87)80081-7
American Dental Association Product Review (2010) Polishing systems. American dental Association, Chicago
Cook MP, Thomas K (1990) Evaluation of gloss meters for measurement of moulded plastics. Polym Testing 9:233–244. https://doi.org/10.1016/0142-9418(90)90010-B
Pala K, Tekçe N, Tuncer S et al (2016) Evaluation of the surface hardness, roughness, gloss and color of composites after different finishing/polishing treatments and thermocycling using a multitechnique approach. Dent Mater J 35:278–289. https://doi.org/10.4012/dmj.2015-260
Lai G, Zhao L, Wang J et al (2018) Surface properties and color stability of dental flowable composites influenced by simulated toothbrushing. Dent Mater J 37:717–724. https://doi.org/10.4012/dmj.2017-233
Aydın N, Topçu F-T, Karaoğlanoğlu S et al (2021) Effect of finishing and polishing systems on the surface roughness and color change of composite resins. J Clin Exp Dent 13:e446–e454. https://doi.org/10.4317/jced.58011
Jain V, Platt JA, Moore K et al (2013) Color stability, gloss, and surface roughness of indirect composite resins. J Oral Sci 55:9–15. https://doi.org/10.2334/josnusd.55.9
Kamonkhantikul K, Arksornnukit M, Takahashi H et al (2014) Polishing and toothbrushing alters the surface roughness and gloss of composite resins. Dent Mater J 33:599–606. https://doi.org/10.4012/dmj.2014-111
Heintze SD, Forjanic M, Rousson V (2006) Surface roughness and gloss of dental materials as a function of force and polishing time in vitro. Dent Mater 22:146–165. https://doi.org/10.1016/j.dental.2005.04.013
Takahashi R, Jin J, Nikaido T et al (2013) Surface characterization of current composites after toothbrush abrasion. Dent Mater J 32:75–82. https://doi.org/10.4012/dmj.2012-160
Tellefsen G, Liljeborg A, Johannsen A et al (2011) The role of the toothbrush in the abrasion process. Int J Dent Hyg 9:284–290. https://doi.org/10.1111/j.1601-5037.2011.00505.x
ISO ISO 11664–4. 2008: Colorimetry - Part 4: CIE 1976 L*a*b* Colour space2008.
ISO ISO 2813. 2014: Paints and varnishes – determination of gloss value at 20°, 60°, 85°2014
Reymus M, Lümkemann N, Stawarczyk B (2019) 3D-printed material for temporary restorations: impact of print layer thickness and post-curing method on degree of conversion. Int J Comput Dent 22:231–237
Reymus M, Stawarczyk B (2020) Influence of different postpolymerization strategies and artificial aging on hardness of 3D-printed resin materials: an in vitro study. Int J Prosthodont 33:634–640. https://doi.org/10.11607/ijp.6634
Reymus M, Stawarczyk B (2021) In vitro study on the influence of postpolymerization and aging on the Martens parameters of 3D-printed occlusal devices. J Prosthet Dent 125:817–823. https://doi.org/10.1016/j.prosdent.2019.12.026
Domingos PA, dos Santos G, Sasso PPN, Oliveira AL, Martins B, de, et al (2011) Composite resin color stability: influence of light sources and immersion media. J Appl Oral Sci 19:204–211. https://doi.org/10.1590/s1678-77572011000300005
Um CM, Ruyter IE (1991) Staining of resin-based veneering materials with coffee and tea. Quintessence Int 22:377–386
Satou N, Khan AM, Matsumae I et al (1989) In vitro color change of composite-based resins. Dent Mater 5:384–387. https://doi.org/10.1016/0109-5641(89)90105-x
Ferracane JL (1985) Correlation between hardness and degree of conversion during the setting reaction of unfilled dental restorative resins. Dent Mater 1:11–14. https://doi.org/10.1016/S0109-5641(85)80058-0
Ferracane JL, Moser JB, Greener EH (1985) Ultraviolet light-induced yellowing of dental restorative resins. J Prosthet Dent 54:483–487. https://doi.org/10.1016/0022-3913(85)90418-4
Waerhaug J (1980) Temporary restorations: advantages and disadvantages. Dent Clin North Am 24:305–316
Pipko DJ, el-Sadeek M, (1972) An in vitro investigation of abrasion and staining of dental resins. J Dent Res 51:689–705. https://doi.org/10.1177/00220345720510030201
Hachiya Y, Iwaku M, Hosoda H et al (1984) Relation of finish to discoloration of composite resins. J Prosthet Dent 52:811–814. https://doi.org/10.1016/s0022-3913(84)80010-4
Shintani H, Satou J, Satou N et al (1985) Effects of various finishing methods on staining and accumulation of Streptococcus mutans HS-6 on composite resins. Dent Mater 1:225–227. https://doi.org/10.1016/S0109-5641(85)80046-4
van Groeningen G, Jongebloed W, Arends J (1986) Composite degradation in vivo. Dent Mater 2:225–227. https://doi.org/10.1016/S0109-5641(86)80018-5
Dietschi D, Campanile G, Holz J et al (1994) Comparison of the color stability of ten new-generation composites: an in vitro study. Dent Mater 10:353–362. https://doi.org/10.1016/0109-5641(94)90059-0
Sarac D, Sarac YS, Kulunk S et al (2006) The effect of polishing techniques on the surface roughness and color change of composite resins. J Prosthet Dent 96:33–40. https://doi.org/10.1016/j.prosdent.2006.04.012
Kamonkhantikul K, Arksornnukit M, Lauvahutanon S et al (2016) Toothbrushing alters the surface roughness and gloss of composite resin CAD/CAM blocks. Dent Mater J 35:225–232. https://doi.org/10.4012/dmj.2015-228
Chang Y-H, Lee C-Y, Hsu M-S et al (2021) Effect of toothbrush/dentifrice abrasion on weight variation, surface roughness, surface morphology and hardness of conventional and CAD/CAM denture base materials. Dent Mater J 40:220–227. https://doi.org/10.4012/dmj.2019-226
Valente LL, Peralta SL, Ogliari FA et al (2013) Comparative evaluation of dental resin composites based on micron- and submicron-sized monomodal glass filler particles. Dent Mater 29:1182–1187. https://doi.org/10.1016/j.dental.2013.09.006
Murakami M (2009) Surface properties of an indirect composite polymerized with five laboratory light polymerization systems. J Oral Sci 51:215–221. https://doi.org/10.2334/josnusd.51.215
Heintze SD, Forjanic M, Ohmiti K et al (2010) Surface deterioration of dental materials after simulated toothbrushing in relation to brushing time and load. Dent Mater 26:306–319. https://doi.org/10.1016/j.dental.2009.11.152
Barbieri GM, Mota EG, Rodrigues-Junior SA et al (2011) Effect of whitening dentifrices on the surface roughness of commercial composites. J Esthet Restor Dent 23:338–345. https://doi.org/10.1111/j.1708-8240.2011.00426.x
Tuncer S, Demirci M, Tiryaki M et al (2013) The effect of a modeling resin and thermocycling on the surface hardness, roughness, and color of different resin composites. J Esthet Restor Dent 25:404–419. https://doi.org/10.1111/jerd.12063
Söderholm KJ, Zigan M, Ragan M et al (1984) Hydrolytic degradation of dental composites. J Dent Res 63:1248–1254. https://doi.org/10.1177/00220345840630101701
Quirynen M, van der Mei HC, Bollen CM et al (1993) An in vivo study of the influence of the surface roughness of implants on the microbiology of supra- and subgingival plaque. J Dent Res 72:1304–1309. https://doi.org/10.1177/00220345930720090801
Bollen CM, Papaioanno W, van Eldere J et al (1996) The influence of abutment surface roughness on plaque accumulation and peri-implant mucositis. Clin Oral Implants Res 7:201–211. https://doi.org/10.1034/j.1600-0501.1996.070302.x
Strasser T, Preis V, Behr M et al (2018) Roughness, surface energy, and superficial damages of CAD/CAM materials after surface treatment. Clin Oral Investig 22:2787–2797. https://doi.org/10.1007/s00784-018-2365-6
Więckiewicz M, Boening KW, Richter G et al (2014) The use of light-cured resin as an alternative method of occlusal splints manufacturing--in vitro study. Adv Clin Exp Med 23:977–985. https://doi.org/10.17219/acem/37354
Danesh G, Lippold C, Ziebura T et al (2006) In-vitro investigation on suitability of light-cured resins for interocclusal splints : part II: surface hardness. J Orofac Orthop 67:138–147. https://doi.org/10.1007/s00056-006-5032-5
Zhang Z-C, Li P-L, Chu F-T et al (2019) Influence of the three-dimensional printing technique and printing layer thickness on model accuracy (Einfluss der dreidimensionalen Drucktechnik und Schichtstärke auf die Modellgenauigkeit). J Orofac Orthop 80:194–204. https://doi.org/10.1007/s00056-019-00180-y
Kraemer Fernandez P, Unkovskiy A, Benkendorff V et al. (2020) Surface characteristics of milled and 3D printed denture base materials following polishing and coating: an in-vitro study. Materials (Basel) 13. https://doi.org/10.3390/ma13153305
Örtengren U, Andersson F, Elgh U et al (2001) Influence of pH and storage time on the sorption and solubility behaviour of three composite resin materials. J Dent 29:35–41. https://doi.org/10.1016/S0300-5712(00)00055-5
Augusto MG, de Andrade GS, Caneppele TMF et al (2020) Nanofilled bis-acryl composite resin materials: is it necessary to polish? J Prosthet Dent 124:494.e1-494.e5. https://doi.org/10.1016/j.prosdent.2020.03.015
Watanabe T, Miyazaki M, Takamizawa T et al (2005) Influence of polishing duration on surface roughness of resin composites. J Oral Sci 47:21–25. https://doi.org/10.2334/josnusd.47.21
Stawarczyk B, Sener B, Trottmann A et al (2012) Discoloration of manually fabricated resins and industrially fabricated CAD/CAM blocks versus glass-ceramic: effect of storage media, duration, and subsequent polishing. Dent Mater J 31:377–383. https://doi.org/10.4012/dmj.2011-238
Da Costa JB, Ferracane JL, Amaya-Pajares S et al (2021) Visually acceptable gloss threshold for resin composite and polishing systems. J Am Dent Assoc 152:385–392. https://doi.org/10.1016/j.adaj.2020.09.027
Dayan C, Guven MC, Gencel B et al (2019) A comparison of the color stability of conventional and CAD/CAM polymethyl methacrylate denture base materials. Acta Stomatol Croat 53:158–167. https://doi.org/10.15644/asc53/2/8
Hollis S, Eisenbeisz E, Versluis A (2015) Color stability of denture resins after staining and exposure to cleansing agents. J Prosthet Dent 114:709–714. https://doi.org/10.1016/j.prosdent.2015.06.001
Sirovica S, Guo Y, Guan R et al (2020) Photo-polymerisation variables influence the structure and subsequent thermal response of dental resin matrices. Dent Mater 36:343–352. https://doi.org/10.1016/j.dental.2019.12.004
Perea-Lowery L, Gibreel M, Vallittu PK et al (2021) Evaluation of the mechanical properties and degree of conversion of 3D printed splint material. J Mech Behav Biomed Mater 115:104254. https://doi.org/10.1016/j.jmbbm.2020.104254
Acknowledgements
We would like to thank individual manufacturers for providing materials.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
For this type of study, formal consent is not required.
Conflict of interest
Verena Hickl declares that she has no conflict of interest.
Dr. Alois Schmid declares that he has no conflict of interest.
Dr. Thomas Strasser declares that she has no conflict of interest.
Prof. Dr. Martin Rosentritt declares that he has third-party research projects with different dental companies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hickl, V., Strasser, T., Schmid, A. et al. Effects of storage and toothbrush simulation on color, gloss, and roughness of CAD/CAM, hand-cast, thermoforming, and 3D-printed splint materials. Clin Oral Invest 26, 4183–4194 (2022). https://doi.org/10.1007/s00784-022-04391-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04391-3