Abstract
Objective
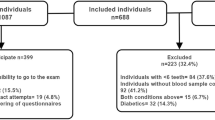
The aim of the present study was to assess the extent and severity of periodontal disease among type 1 diabetic patients (T1DM) and to investigate the possible association with systemic markers of glucose control and variability.
Material and methods
Patients were consecutively enrolled in a Diabetic Unit. A full-mouth periodontal evaluation was performed, and data on systemic markers of diabetes were collected. Descriptive statistics and logistic and linear models were performed.
Results
A total of 136 T1DM patients (mean age: 45.5 ± 14.6 years) were examined. Periodontitis was detected in 62% of cases (mean CAL: 3.0 ± 0.9 mm): stage III periodontitis was diagnosed in 32% of patients while stage IV in 8%. Mean level of glycated hemoglobin (HbA1c) was 7.5% ± 1.4. Among the investigated factors, mean CAL (p=0.040) was associated with HbA1c ≥ 7%; 93% of patients with mean CAL > 6 mm showed HbA1c ≥ 7%. Mean CAL (p=0.004), mean PPD (p=0.005), mean FMPS (p=0.030), and stage III/IV periodontitis (p=0.018) predict glucose coefficient of variation (CV).
Conclusions
Periodontitis showed a relevant prevalence in the present, well-controlled T1DM population and predicts poor glycemic control (HbA1c ≥7%) and higher glucose variability. The present findings suggest that periodontal infection may have systemic effects also in T1DM patients.
Clinical relevance
The extent and severity of periodontitis and its possible systemic effects in T1DM patients could be underestimated.
Similar content being viewed by others
References
Sanz M, Quirynen M, European Workshop in Periodontology Group A (2005) Advances in the aetiology of periodontitis. Group A: consensus report of the 5th European Workshop in Periodontology. J Clin Periodontol 32(Suppl. 6):54–56. https://doi.org/10.1111/j.1600-051X.2005.00827.x
Kassebaum NJ, Bernab E, Dahiya M, Bhandari B, Murray CJ, Marcenes W (2014) Global burden of severe periodontitis in 1990-2010: a systematic review and meta-regression. J Dental Re 93(11):1045–1053. https://doi.org/10.1177/0022034514552491
Chapple IL, Genco R, Working group 2 of the joint EFP/AAP workshop (2013) Diabetes and periodontal diseases: consensus report of the Joint EFP/AAP Workshop on Periodontitis and Systemic Diseases. J Periodontol 84(4 Suppl):S106–S112. https://doi.org/10.1902/jop.2013.1340011
Kapila YL (2021) Oral health’s inextricable connection to systemic health: special populations bring to bear multimodal relationships and factors connecting periodontal disease to systemic diseases and conditions. Periodontol 87(1):11–16. https://doi.org/10.1111/prd.12398
Cairo F, Castellani S, Gori AM, Nieri M, Baldelli G, Abbate R, Pini-Prato GP (2008) Severe periodontitis in young adults is associated with sub-clinical atherosclerosis. J Clin Periodontol 35(6):465–472. https://doi.org/10.1111/j.1600-051X.2008.01228.x
Cairo F, Castellani S, Gori AM, Nieri M, Abbate R, Pini-Prato GP (2009) Periodontol variables predict sub-clinical atherosclerosis and systemic inflammation in young adults. Eur J Oral Implant 2:125–133
D’Aiuto F, Gkranias N, Bhowruth D, Khan T, Orlandi M, Suvan J, Masi S, Tsakos G, Hurel S, Hingorani AD, Donos N, Deanfield JE, TASTE Group (2018) Systemic effects of periodontitis treatment in patients with type 2 diabetes: a 12 month, single-centre, investigator-masked, randomised trial. Lancet Diabetes Endocrinol 6(12):954–965. https://doi.org/10.1016/S2213-8587(18)30038-X
Cairo F, Gaeta C, Dorigo W, Oggioni MR, Pratesi C, Pini Prato GP, Pozzi G (2004) Periodontal pathogens in atheromatous plaques. A controlled clinical and laboratory plaques. J Periodontal Res 39:442–446. https://doi.org/10.1111/j.1600-0765.2004.00761.x
Genco RJ, Borgnakke WS (2013) Diabetes as a potential risk for periodontitis: association studies. Periodontol 83(1):40–45. https://doi.org/10.1111/prd.12270
Genco RJ, Borgnakke WS (2020) Diabetes as a potential risk for periodontitis: association studies. Periodontol 83(1):40–45. https://doi.org/10.1111/prd.12270
Polak D, Sanui T, Nishimura F, Shapira L (2020) Diabetes as a risk factor for periodontal disease-plausible mechanisms. Periodontol 83(1):46–58. https://doi.org/10.1111/prd.12298
Golub LM, Schneir M, Ramamurthy NS (1978) Enhanced collagenase activity in diabetic rat gingiva: in vitro and in vivo evidence. J Dent Res 57(3):520–525. https://doi.org/10.1177/00220345780570032101
Federoff HJ, Lawrence D, Brownlee M (1993) Nonenzymatic glycosylation of laminin and the laminin peptide CIKVAVS inhibits neurite outgrowth. Diabetes 42(4):509–513. https://doi.org/10.2337/diab.42.4.509
Zambon JJ, Reynolds H, Fisher JG, Shlossman M, Dunford R, Genco RJ (1988) Microbiological and immunological studies of adult periodontitis in patients with noninsulin-dependent diabetes mellitus. J Periodontol 59(1):23–31. https://doi.org/10.1902/jop.1988.59.1.23
Salvi GE, Lawrence HP, Offenbacher S, Beck JD (1997) Influence of risk factors on the pathogenesis of periodontitis. Periodontol 14:173–201. https://doi.org/10.1111/j.1600-0757.1997.tb00197.x
Ko KI, Sculean A, Graves DT (2021) Diabetic wound healing in soft and hard oral tissues. Transl Res 236:72–86. https://doi.org/10.1016/j.trsl.2021.05.001
Sanz M, Ceriello A, Buysschaert M, Chapple I, Demmer RT, Graziani F, Herrera D, Jepsen S, Lione L, Madianos P, Mathur M, Montanya E, Shapira L, Tonetti M, Vegh D (2018) Scientific evidence on the links between periodontal diseases and diabetes: consensus report and guidelines of the joint workshop on periodontal diseases and diabetes by the International Diabetes Federation and the European Federation of Periodontology. J Clin Periodontol 45(2):138–149. https://doi.org/10.1111/jcpe.12808
Genco RJ, Graziani F, Hasturk H (2020) Effects of periodontal disease on glycemic control, complications, and incidence of diabetes mellitus. Periodontol 83(1):59–65. https://doi.org/10.1111/prd.12271
Graziani F, Gennai S, Solini A, Petrini M (2018) A systematic review and meta-analysis of epidemiologic observational evidence on the effect of periodontitis on diabetes An update of the EFP-AAP review. J Clin Periodontol 45(2):167–187. https://doi.org/10.1111/jcpe.12837
Engebretson S, Kocher T (2013) Evidence that periodontal treatment improves diabetes outcomes: a systematic review and meta-analysis. J Periodontol 84(Suppl 4):S153–S169. https://doi.org/10.1902/jop.2013.1340017
Dicembrini I, Serni L, Monami M, Caliri M, Barbato L, Cairo F, Mannucci E (2020) Type 1 diabetes and periodontitis: prevalence and periodontal destruction-a systematic review. Acta Diabetol 57(12):1405–1412. https://doi.org/10.1007/s00592-020-01531-7
Dicembrini I, Barbato L, Serni L, Caliri M, Pala L, Cairo F, Mannucci E (2021) Glucose variability and periodontal disease in type 1 diabetes: a cross-sectional study-the “PAROdontopatia e DIAbete” (PARODIA) project. Acta Diabetol 58(10):1367–1371. https://doi.org/10.1007/s00592-021-01720-y
Tonetti MS, Greenwell H, Kornman KS (2018) Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J Clin Periodontol 45(Suppl 20):S149–S161. https://doi.org/10.1111/jcpe.12945
Hsieh FY (1989) Sample size tables for logistic regression. Stat Med 8(7):795–802. https://doi.org/10.1002/sim.4780080704
Hugoson A, Norderyd O, Slotte C, Thorstensson H (1998) Distribution of periodontal disease in a Swedish adult population 1973, 1983 and 1993. J Clin Periodontol 25:542–548. https://doi.org/10.1111/j.1600-051x.1998.tb02485.x
Tonetti MS, Jepsen S, Jin L, Otomo-Corgel J (2017) Impact of the global burden of periodontal diseases on health, nutrition and wellbeing of mankind: a call for global action. J Clin Periodontol 44:456–462. https://doi.org/10.1111/jcpe.12732
Preshaw PM, Alba AL, Herrera D, Jepsen S, Kostantinidis A, Makrilakis K, Taylor R (2012) Periodontitis and diabetes: a two-way relationship. Diabetologia 55(1):21–31. https://doi.org/10.1007/s00125-011-2342-y
Taylor GW (2001) Bidirectional interrelationships between diabetes and periodontal diseases: an epidemiologic perspective. Ann Periodontol 6:99–112. https://doi.org/10.1902/annals.2001.6.1.99
Lalla E, Cheng B, Lal S, Kaplan S, Softness B, Greenberg E, Goland RS, Lamster I (2007) Diabetes mellitus promotes periodontal destruction in children. J Clin Periodontol 34:294–298. https://doi.org/10.1111/j.1600-051X.2007.01054.x
Funding
This study was self-funded.
Author information
Authors and Affiliations
Ethics declarations
Ethical approval
The study protocol was approved by the Ethical Board (PARODIA Project, approval number: CEAVC 30952/2019).
Conflict of interest
Francesco Cairo has received consultancy fees and grants from Straumann and Geistlich Biomaterials. Ilaria Dicembrini has received speaking fees from Merck, Novartis, AstraZeneca, Bristol Myers Squibb, Boehringer-Ingelheim, Eli-Lilly, Novo Nordisk, Sanofi, and Novartis. Edoardo Mannucci has received consultancy fees from Merck and Novartis; speaking fees from AstraZeneca, Bristol Myers Squibb, Boehringer-Ingelheim, Eli-Lilly, Merck, Novo Nordisk, Sanofi; and Novartis, and research grants from Merck, Novartis, and Takeda. Other authors have nothing to disclose.
Informed consent
Informed consent was obtained from all subjects included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cairo, F., Dicembrini, I., Serni, L. et al. Periodontitis predicts HbA1c levels and glucose variability in type 1 diabetic patients: the PARODIA Florence Project study. Clin Oral Invest 26, 3585–3591 (2022). https://doi.org/10.1007/s00784-021-04326-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-021-04326-4