Abstract
Background
To explore the association between obesity and tooth wear among American adults and the role of sugar-sweetened acidic drinks consumption in explaining that association.
Methods
We analyzed data from 3541 adult participants in the National Health and Nutrition Examination Survey. Obesity was determined using the body mass index and tooth wear was assessed using the modified tooth wear index. Daily intake of four categories of drinks was estimated as the average (drinks/day) of two consecutive 24-h dietary recalls. The association between obesity and number of surfaces with moderate-to-severe tooth wear was assessed in hurdle models adjusting for sociodemographic factors, acid reflux medication, and dental insurance.
Results
Overweight and obese adults had more surfaces with moderate-to-severe tooth wear than those with normal body size, after adjusting for confounders. The consumption of sugar-sweetened acidic drinks explained part, but not all the above association. More specifically, the estimate for obesity was fully attenuated, whereas the estimate for overweight was slightly attenuated but remained significant.
Conclusion
Obesity was positively associated with tooth wear in American adults. This association was only partially accounted for by the consumption of sugar-sweetened acidic drinks, a common risk factor for both conditions.
Practical implications
Dentists must be aware of the health consequences of sugar-sweetened acidic drinks and advocate for reduction in consumption and/or substitution with healthier alternatives.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Tooth wear is the loss of dental hard tissues resulting of the interaction between teeth and other materials (abrasion), tooth-to-tooth contact (attrition), or dissolution of hard tissues by acidic substances not caused by bacteria (erosion) [1, 2]. As the prevalence of tooth wear is raising [3, 4], a focus on prevention may prolong the health of the dentition. At the same time, obesity is increasing at an alarming rate globally to the point it is now considered a global pandemic [5, 6].
A few studies have investigated the association between obesity and tooth wear [7,8,9,10], with most evidence coming from studies in children [7,8,9]. However, results are still contradicting as one study in England showed that obese children were more likely to have erosive tooth wear than normal weight children [8], whereas studies in the USA [7] and Brazil [9] reported no association between the two conditions. A study among Swedish young adults found that overweight and obesity were more common among participants with dental erosion than among those with no erosion. However, results were merely descriptive since estimates were not adjusted for potential confounders [10].
There are several potential reasons why obese individuals may be more likely to have tooth wear. First and foremost is the confounding role of factors associated with both obesity and tooth wear. One of those factors is the increased consumption of soft drinks, which is associated with both obesity [11, 12] and tooth wear [13, 14]. Globally, a rise in soft drinks consumption [15, 16] parallels those in obesity [5, 6] and tooth wear [3, 4]. However, while it is the sugar content of soft drinks (often referred to as sugar-sweetened beverages) what leads to obesity [15], it is the acidic nature of some drinks (such as carbonated drinks and acidic fruit juices) that leads to tooth wear [14]. As such, the key confounder here is the consumption of sugar-sweetened acidic drinks. Beyond the confounding role of sugar-sweetened acidic drinks, it is possible that obesity has an independent effect on tooth wear. Abdominal obesity could be associated with esophageal dysfunction, such as increased acid exposure and reflux symptoms, which in turn may lead to a higher risk of intrinsic erosive tooth wear [17, 18]. More pathological tooth wear and more vomiting and gastroesophageal reflux has also been observed among morbidly obese adults (waiting for bariatric surgery) than healthy controls [19].
The above factors may be interrelated but have yet to be fully investigated. Therefore, the aim of this study was to explore the association between obesity and tooth wear among American adults and the role of the consumption of sugar-sweetened acidic drinks in explaining that association.
Methods
Data source
We used data from the National Health and Nutrition Examination Survey (NHANES) 2003–04, which is part of a series of surveys conducted by the Center for Disease Control and Prevention (CDC) to assess the health and nutritional status of the non-institutionalized civilian population aged 2 years and older residing in the USA [20]. The 2003–04 NHANES is the only survey that included an expanded oral health section providing information on oral health conditions that were never assessed previously and have not been assessed again since then.
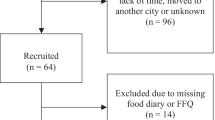
The selection of survey participants was conducted by stratified, multistage probability sampling to recruit a representative sample. Oversampling was conducted on certain subgroups which included non-Hispanic blacks, Mexican Americans, and participants aged 12 to 19 years and 60 years or more. Data were collected through phone interviews, face-to-face interviews, and clinical examinations. A total of 10,122 individuals of the eligible population agreed to take part in the survey (79% response rate). The expanded oral health examination was conducted on participants aged 13 years and older and included 8272 participants (76%) [20]. The NHANES 2003–04 survey included 4308 participants aged 18 years and over with complete tooth wear. We excluded 767 participants because of missing data on obesity, dietary data, and confounders. Therefore, the study sample included 3541 participants.
Variables selection
The outcome variable for this study was tooth wear, which was assessed using the modified tooth wear index (TWI), has been used in the UK Adult Dental Health Survey [21]. The clinical assessment included visual examination of the facial, lingual, and incisal surfaces of the 12 anterior teeth as well as occlusal surfaces of the first molars (40 tooth surfaces overall). Tooth surfaces were scored as sound (any wear restricted to the enamel); mild tooth wear (loss of enamel just exposing dentine); moderate tooth wear (dentine exposure greater than one-third of the surface); or severe tooth wear (complete loss of enamel, pulp exposure, or exposure of secondary dentine). Oral clinical examinations were conducted by two trained and calibrated examiners. Inter-examiner reliability for the tooth wear examination gave a percent agreement of 87.7% and a kappa of 0.80 [20]. The number of surfaces with moderate-to-severe tooth wear was the outcome for this study.
The exposure was obesity measured using the body mass index (BMI). Trained health technicians measured participants’ weight and height using standard protocols and equipment [22]. BMI was calculated as body weight (in kilograms) divided by the square of height (in meters) and used to classify participants as normal (BMI < 25), overweight (25 < BMI < 29.9), or obese (BMI > 30).
Several confounders were also selected for analysis based on previous studies [23, 24]. A basic set of confounders included demographic characteristics (sex, age, race/ethnicity), socioeconomic position (education and poverty income ratio), acid reflux medication (antacids and anti-reflux medication, as a proxy for gastroesophageal reflux symptoms), and dental insurance coverage. The key confounder in our analysis was the intake of sugar-sweetened acidic drinks, which was estimated from two non-consecutive 24-h recalls [25]. The first 24-h dietary recall was performed in person by trained interviewers in the mobile examination center, while the second 24-h dietary recall was conducted by telephone 3 to 10 days after the examination. Dietary recall data were used to measure an average intake (drinks/day) across the 2 days of four different categories of drinks: sugar-sweetened acidic drinks (SSA: sweetened soda, non-100% fruit juice, sweetened sports drinks), sugar-sweetened non-acidic drinks (SSNA: sugar-sweetened coffee, tea- and milk-based products with sugar), non-sugar-sweetened acidic drinks (NSSA: diet soft drinks, 100% fruit juice or flavored sparkling water), non-sugar-sweetened non-acidic drinks (NSSNA: non-sugar-sweetened coffee, tea- and milk-based products). The specific USDA food codes included in each of the four categories of drinks are listed in the Appendix.
Data analysis
Data were analyzed using Stata/SE version 14.1 (Stata Corp., College Station TX, USA). All analyses were weighted to produce a representative sample and to take account of the variation in cluster size and different sampling rates for clusters within strata.
The distribution of the number of surfaces with moderate-to-severe tooth wear was positively skewed, with many participants having zero counts. Therefore, a hurdle model was preferred to model associations with the number of surfaces with moderate-to-severe tooth wear. Hurdle models account for the excessive proportion of zero values and over-dispersion [26, 27]. They include two components. The first component estimates the probability of being a non-zero (positive) count from a binary logit model. Odds ratios (OR) were reported for this component of the model, which represent the probability of having moderate-to-severe tooth wear. The second component estimates the mean number of surfaces with moderate-to-severe tooth wear among those with positive counts only (those with tooth wear) using a truncated negative binomial model. Rate ratios (RRs) were reported for this component, which represent how higher/lower the mean number of surfaces with moderate-to-severe tooth wear was in one group compared to the reference group, among those with the condition [26, 27].
We first examined the association between each type of drink (SSA, SSNA, NSSA, NSSNA) and the number of surfaces with moderate-to-severe tooth wear through a series of regression models: Model 1A was unadjusted; model 1B was adjusted for confounders (sex, age groups, race/ethnicity, education, poverty income ratio, dental insurance, and acid reflux medication); and model 2C was additionally adjusted for the remaining types of drinks. We then examined the association between BMI and the number of surfaces with moderate-to-severe tooth wear through a series of regression models: model 2A was unadjusted; model 2B was adjusted for confounders (sex, age groups, race/ethnicity, education, poverty income ratio, dental insurance, and acid reflux medication); models 2C to 2F were additionally adjusted for one type of drink at a time (model 2C: SSA, model 2D: SSNA, model 2E: NSSA, model 2F: NSSNA); and model 3F was adjusted for confounders and all four types of drinks simultaneously.
Results
There were some differences between the study sample and those not included due to missing values on covariates (Table 1). On average, participants in the study sample were older (43.9 versus 39.9 years, p = 0.001) and reported greater consumption of NSSNA drinks (1.51 versus 1.24 drinks/day, p = 0.038) than those excluded. In the study sample, the prevalence of moderate-to-severe tooth wear was 12.1%, with an average of 3.4 (95% confidence interval 2.9–3.9) surfaces affected among those with the condition. Also, 33.7% and 32% of adults were overweight and obese, respectively.
The association of the daily intake of different types of drinks with moderate-to-severe tooth wear is presented in Table 2. The intakes of SSA, SSNA, NSSA, and NSSNA drinks were not associated with the probability of having moderate-to-severe tooth wear. However, the intakes of SSA and NSSNA drinks were significantly associated with the number of surfaces with moderate-to-severe tooth wear among those with the condition (model 1B). In those with tooth wear, the number of surfaces affected was 1.40 (95% CI 1.14–1.73) times higher per additional SSA drink consumed per day. On the other hand, the number of surfaces with moderate-to-severe tooth wear was 17% (RR 0.83, 95% CI 0.75–0.93) lower per additional NSSNA drinks consumed per day. These two associations were slightly attenuated but remained significant after further adjustment for other types of drinks (RR of 1.34 and 0.86 for SSA and NSSNA drinks from model 1C, respectively).
BMI was not associated with tooth wear in the unadjusted model (model 2A in Table 3). After adjustment for confounders (model 2B), BMI was not significantly associated with the probability of having moderate-to-severe tooth wear, but it was associated with the number of surfaces with moderate-to-severe tooth wear among those with the condition. In those with tooth wear, the number of surfaces affected was, respectively, 1.72 (95% CI 1.09–2.71) and 1.61 (95% CI 1.00–2.58) times higher in overweight and obese adults than in those with normal body size. However, the estimate for obese adults was fully attenuated after further adjustment for either SSA, NSSA, or NSSNA drinks (models 2C, 2E, and 2F, respectively). The estimate for obese adults remained significant after adjustment for SSNA drinks (RR 1.62, 95% CI 1.01–2.58 from model 2D). When adjusted for confounders and all four types of drinks (model 2G), the estimate for overweight remained significant (RR 1.56, 95% CI 1.04–2.34), while the estimate for obese adults was fully attenuated (RR 1.41, 95% CI 0.90–2.21).
Discussion
This study first found that obesity was positively associated with tooth wear, independently of known risk factors for tooth wear. Interestingly, this association was found with the number of surfaces with moderate-to-severe tooth wear among those with the condition (i.e., an indication of its severity), but not with the prevalence of tooth wear. Our finding reinforces the opinion that a binary classification of individuals as simply having or not having tooth wear (prevalence) hides fine-grained information on the extent of the condition, and it may potentially mask associations [24].
Beyond this initial finding, we also set up to explore the role of the consumption of SSA drinks in explaining the above association because this category of beverages is associated with both obesity and tooth wear. Therefore, the consumption of SSA drinks may be a common risk factor for both conditions. As expected, SSA drinks consumption was positively associated with tooth wear and explained part of the association between obesity and tooth wear. Indeed, the estimate for obesity, but not that for overweight, becomes fully attenuated when SSA drinks consumption was included in the regression model. Accounting for other categories of drinks did not alter that finding. It is possible that obese adults have already been diagnosed with the condition and cut down on their usual intake of sugar-sweetened beverages (some of which are acidic). Contrarily, overweight adults might still be indulging in their (high) consumption of SSA drinks. The fact that the association between overweight and tooth wear was not explained away by the intake of SSA drinks implies that other factors may be at play. There is considerable evidence linking obesity with gastroesophageal reflux disease (GERD) symptoms [17, 18], a cause of erosive tooth wear [28, 29]. Increased abdominal pressure caused by central adiposity relaxes the lower esophageal sphincter, thus exposing the esophageal and oral mucosa to gastric content [17]. On the other hand, adipose tissue is metabolically active and induces the production of inflammatory cytokines which may play a role in GERD [18].
An additional finding of this study was that the consumption of NSSNA drinks was inversely associated with tooth wear (among those with the condition only), suggesting a potential protective effect. In this study, NSSNA drinks included items like unsweetened tea, coffee, milk, and non-sugar-sweetened milk-based products. Previously, it has been reported that milk-based products were associated with reduced tooth wear [14], although another review reported no association [13]. This protective effect could be due to substitution of the acidic beverage with a less acidic counterpart [14] or, less likely, due to the calcium and phosphate levels in the drinks encouraging remineralization or providing sacrificial minerals in the event of an acid challenge [30,31,32].
The present findings have implications on practice and future research. These results suggest that SSA drinks consumption is a common root for obesity and tooth wear. Addressing their consumption provides an important target area for healthcare practitioners to reduce the health impact of both conditions. It is probably easier to advocate their substitution with NSSNA drinks rather than reducing their consumption all together and recent behavioral research has shown relative success with this method [33]. Further research in this area is needed, and it would be useful to include tooth wear assessment in national health surveys to continue this work. There is also a need for longitudinal studies using multiple dietary assessments, repeated body measurements, and more accurate quantification of tooth wear progression to confirm this relationship.
Some limitations of this study need to be addressed. First, this study was based on cross-sectional data and as such unable to test for causal associations. Second, our study sample represented 82% of eligible adults, which may raise concerns about representativeness. Since older participants and high-NSSNA drinkers were more likely to be included in the study sample, the present findings should be generalized with caution. Third, tooth wear assessment was based on partial-mouth examination which may not be as valid as full-mouth examination [34]. However, past studies have indicated that anterior teeth and the occlusal surface of first molars are the most commonly affected by tooth wear [35, 36]. Fourth, we assessed participants’ dietary intake through 24-h recalls, which do not measure long-term dietary habits of individuals and are prone to recall bias and under/over-reporting. Compared with food frequency questionnaire data also available in NHANES, the use of multiple 24-h recalls provided information regarding a significantly larger number of drinks that were combined into four categories. Finally, we were unable to control for GERD (although we used a proxy based on acid reflux medication) or vomiting eating disorders which are known risk factors for tooth wear.
In conclusion, obesity was positively associated with tooth wear in this national sample of American adults. This association was partially accounted for by the consumption of sugar-sweetened acidic drinks, a common risk factor for both conditions. All the findings were observed with the extent (among those with the condition) but not the prevalence of moderate-to-severe tooth wear.
References
Shellis RP, Addy M (2014) The interactions between attrition, abrasion and erosion in tooth wear. Monogr Oral Sci 25:32–45. https://doi.org/10.1159/000359936
Carvalho TS, Colon P, Ganss C, Huysmans MC, Lussi A, Schlueter N, Schmalz G et al (2015) Consensus report of the European Federation of Conservative Dentistry: erosive tooth wear--diagnosis and management. Clin Oral Investig 19:1557–1561. https://doi.org/10.1007/s00784-015-1511-7
Salas MM, Nascimento GG, Huysmans MC, Demarco FF (2015) Estimated prevalence of erosive tooth wear in permanent teeth of children and adolescents: an epidemiological systematic review and meta-regression analysis. J Dent 43:42–50. https://doi.org/10.1016/j.jdent.2014.10.012
Schlueter N, Luka B (2018) Erosive tooth wear - a review on global prevalence and on its prevalence in risk groups. Br Dent J 224:364–370. https://doi.org/10.1038/sj.bdj.2018.167
Popkin BM, Adair LS, Ng SW (2012) Global nutrition transition and the pandemic of obesity in developing countries. Nutr Rev 70:3–21. https://doi.org/10.1111/j.1753-4887.2011.00456.x
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, Mullany EC et al (2014) Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384:766–781. https://doi.org/10.1016/s0140-6736(14)60460-8
McGuire J, Szabo A, Jackson S, Bradley TG, Okunseri C (2009) Erosive tooth wear among children in the United States: relationship to race/ethnicity and obesity. Int J Paediatr Dent 19:91–98. https://doi.org/10.1111/j.1365-263X.2008.00952.x
Tong HJ, Rudolf MC, Muyombwe T, Duggal MS, Balmer R (2014) An investigation into the dental health of children with obesity: an analysis of dental erosion and caries status. Eur Arch Paediatr Dent 15:203–210. https://doi.org/10.1007/s40368-013-0100-1
Salas MM, Vargas-Ferreira F, Nascimento GG, Huysmanns MC, Demarco FF (2018) Tooth erosion association with obesity: findings from a Brazilian survey in schoolchildren. Pesquisa Brasileira em Odontopediatria e Clinica Integrada 18:e3764
Isaksson H, Birkhed D, Wendt LK, Alm A, Nilsson M, Koch G (2014) Prevalence of dental erosion and association with lifestyle factors in Swedish 20-year olds. Acta Odontol Scand 72:448–457. https://doi.org/10.3109/00016357.2013.859727
Trumbo PR, Rivers CR (2014) Systematic review of the evidence for an association between sugar-sweetened beverage consumption and risk of obesity. Nutr Rev 72:566–574. https://doi.org/10.1111/nure.12128
Keller A, Bucher Della Torre S (2015) Sugar-sweetened beverages and obesity among children and adolescents: a review of systematic literature reviews. Childhood Obes (Print) 11:338–346. https://doi.org/10.1089/chi.2014.0117
Li H, Zou Y, Ding G (2012) Dietary factors associated with dental erosion: a meta-analysis. PLoS One 7:e42626. https://doi.org/10.1371/journal.pone.0042626
Salas MM, Nascimento GG, Vargas-Ferreira F, Tarquinio SB, Huysmans MC, Demarco FF (2015) Diet influenced tooth erosion prevalence in children and adolescents: results of a meta-analysis and meta-regression. J Dent 43:865–875. https://doi.org/10.1016/j.jdent.2015.05.012
Popkin BM, Hawkes C (2016) Sweetening of the global diet, particularly beverages: patterns, trends, and policy responses. Lancet Diabetes Endocrinol 4:174–186. https://doi.org/10.1016/s2213-8587(15)00419-2
Dunford EK, Popkin BM (2017) Disparities in snacking trends in US adults over a 35 year period from 1977 to 2012. Nutrients 9:809. https://doi.org/10.3390/nu9080809
Friedenberg FK, Xanthopoulos M, Foster GD, Richter JE (2008) The association between gastroesophageal reflux disease and obesity. Am J Gastroenterol 103:2111–2122. https://doi.org/10.1111/j.1572-0241.2008.01946.x
Emerenziani S, Rescio MP, Guarino MP, Cicala M (2013) Gastro-esophageal reflux disease and obesity, where is the link? World J Gastroenterol 19:6536–6539. https://doi.org/10.3748/wjg.v19.i39.6536
Alves MS, da Silva FA, Araujo SG, de Carvalho AC, Santos AM, de Carvalho AL (2012) Tooth wear in patients submitted to bariatric surgery. Braz Dent J 23:160–166. https://doi.org/10.1590/S0103-64402012000200012
Dye BA, Nowjack-Raymer R, Barker LK, Nunn JH, Steele JG, Tan S, Lewis BG et al (2008) Overview and quality assurance for the oral health component of the National Health and Nutrition Examination Survey (NHANES), 2003-04. J Public Health Dent 68:218–226. https://doi.org/10.1111/j.1752-7325.2007.00076.x
Li MH, Bernabe E (2016) Tooth wear and quality of life among adults in the United Kingdom. J Dent 55:48–53. https://doi.org/10.1016/j.jdent.2016.09.013
Centers for Disease Control and Prevention (2000) National Health and Nutrition Examination Survey - Anthropometry Procedures Manual. National Center for Health Statistics
O'Toole S, Bernabe E, Moazzez R, Bartlett D (2017) Timing of dietary acid intake and erosive tooth wear: a case-control study. J Dent 56:99–104. https://doi.org/10.1016/j.jdent.2016.11.005
Al-Zwaylif LH, O'Toole S, Bernabe E (2018) Type and timing of dietary acid intake and tooth wear among American adults. J Public Health Dent. https://doi.org/10.1111/jphd.12264
Raper N, Perloff B, Ingwersen L, Steinfeldt L, Anand J (2004) An overview of USDA’s dietary intake data system. J Food Compos Anal 17:545–555. https://doi.org/10.1016/j.jfca.2004.02.013
Cameron AC, Trivedi PK (2013) Regression analysis of count data. Cambridge University Press, Cambridge
Hilbe JM (2014) Modeling count data. Cambridge University Press, Cambridge
Moazzez R, Bartlett D (2014) Intrinsic causes of erosion. Monogr Oral Sci 25:180–196. https://doi.org/10.1159/000360369
Wilder-Smith CH, Materna A, Martig L, Lussi A (2017) Longitudinal study of gastroesophageal reflux and erosive tooth wear. BMC Gastroenterol 17:113–116. https://doi.org/10.1186/s12876-017-0670-1
Aimutis WR (2004) Bioactive properties of milk proteins with particular focus on anticariogenesis. J Nutr 134:989s–995s. https://doi.org/10.1093/jn/134.4.989S
Barbour ME, Shellis RP, Parker DM, Allen GC, Addy M (2008) Inhibition of hydroxyapatite dissolution by whole casein: the effects of pH, protein concentration, calcium, and ionic strength. Eur J Oral Sci 116:473–478. https://doi.org/10.1111/j.1600-0722.2008.00565.x
Al-Khateeb SN, Tarazi SJ, Al Maaitah EF, Al-Batayneh OB, Abu Alhaija ES (2014) Does acid etching enhance remineralisation of arrested white spot lesions? Eur Arch Paediatr Dentist 15:413–419. https://doi.org/10.1007/s40368-014-0131-2
O'Toole S, Newton T, Moazzez R, Hasan A, Bartlett D (2018) Randomised controlled clinical trial investigating the impact of implementation planning on behaviour related to the diet. Sci Rep 8:8024–8026. https://doi.org/10.1038/s41598-018-26418-0
Ganss C, Young A, Lussi A (2011) Tooth wear and erosion: methodological issues in epidemiological and public health research and the future research agenda. Community Dent Health 28:191–195. https://doi.org/10.1922/CDH_2796Ganss05
Steele JG, Walls AW (2000) Using partial recording to assess tooth wear in older adults. Community Dent Oral Epidemiol 28:18–25. https://doi.org/10.1034/j.1600-0528.2000.280103.x
Johansson AK, Omar R, Carlsson GE, Johansson A (2012) Dental erosion and its growing importance in clinical practice: from past to present. Int J Dentist 2012:632907. https://doi.org/10.1155/2012/632907
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Written informed consent was obtained from every participant in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 40 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Kamal, Y., O’Toole, S. & Bernabé, E. Obesity and tooth wear among American adults: the role of sugar-sweetened acidic drinks. Clin Oral Invest 24, 1379–1385 (2020). https://doi.org/10.1007/s00784-019-03079-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-019-03079-5