Abstract
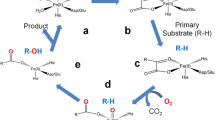
Human deoxyhypusine hydroxylase (hDOHH) is an enzyme that is involved in the critical post-translational modification of the eukaryotic translation initiation factor 5A (eIF5A). Following the conversion of a lysine residue on eIF5A to deoxyhypusine (Dhp) by deoxyhypusine synthase, hDOHH hydroxylates Dhp to yield the unusual amino acid residue hypusine (Hpu), a modification that is essential for eIF5A to promote peptide synthesis at the ribosome, among other functions. Purification of hDOHH overexpressed in E. coli affords enzyme that is blue in color, a feature that has been associated with the presence of a peroxo-bridged diiron(III) active site. To gain further insight into the nature of the diiron site and how it may change as hDOHH goes through the catalytic cycle, we have conducted X-ray absorption spectroscopic studies of hDOHH on five samples that represent different species along its reaction pathway. Structural analysis of each species has been carried out, starting with the reduced diferrous state, proceeding through its O2 adduct, and ending with a diferric decay product. Our results show that the Fe⋯Fe distances found for the five samples fall within a narrow range of 3.4–3.5 Å, suggesting that hDOHH has a fairly constrained active site. This pattern differs significantly from what has been associated with canonical dioxygen activating nonheme diiron enzymes, such as soluble methane monooxygenase and Class 1A ribonucleotide reductases, for which the Fe⋯Fe distance can change by as much as 1 Å during the redox cycle. These results suggest that the O2 activation mechanism for hDOHH deviates somewhat from that associated with the canonical nonheme diiron enzymes, opening the door to new mechanistic possibilities for this intriguing family of enzymes.
Similar content being viewed by others
References
Lundin D, Poole AM, Sjöberg B-M, Högbom M (2012) J Biol Chem 287:20565–20575
Nordlund P, Eklund H (1995) Curr Opin Struct Biol 5:758–766
Liu X, Theil EC (2005) Acc Chem Res 38:167–175
Nordlund P, Reichard P (2006) Annu Rev Biochem 75:681–706
Wallar BJ, Lipscomb JD (1996) Chem Rev 96:2625–2658
Tinberg CE, Lippard SJ (2011) Acc Chem Res 44:280–288
Bochevarov AD, Li J, Song WJ, Friesner RA, Lippard SJ (2011) J Am Chem Soc 133:7384–7397
Fox BG, Lyle KS, Rogge CE (2004) Acc Chem Res 37:421–429
Choi YS, Zhang H, Brunzelle JS, Nair SK, Zhao H (2008) Proc Natl Acad Sci USA 105:6858–6863
Makris TM, Vu VV, Meier KK, Komor AJ, Rivard BS, Münck E, Que L, Lipscomb JD (2015) J Am Chem Soc 137:1608–1617
Krebs C, Bollinger JM, Booker SJ (2011) Curr Opin Chem Biol 15:291–303
Das D, Eser BE, Han J, Sciore A, Marsh ENG (2011) Angew Chem Int Ed 50:7148–7152
Kim YS, Kang KR, Wolff EC, Bell JK, McPhie P, Park MH (2006) J Biol Chem 281:13217–13225
Park MH (2006) J Biochem 139:161–169
Saini P, Eyler DE, Green R, Dever TE (2009) Nature 459:118–121
Gutierrez E, Shin B-S, Woolstenhulme CJ, Kim J-R, Saini P, Buskirk AR, Dever TE (2013) Mol Cell 51:35–45
Park MH, Nishimura K, Zanelli CF, Valentini SR (2010) Amino Acids 38:491–500
Kaiser A (2012) Amino Acids 42:679–684
Greganova E, Altmann M, Bütikofer P (2011) FEBS J 278:2613–2624
Vu VV, Emerson JP, Martinho M, Kim YS, Münck E, Park MH, Que L (2009) Proc Natl Acad Sci USA 106:14814–14819
Liu KE, Valentine AM, Wang D, Huynh BH, Edmondson DE, Salifoglou A, Lippard SJ (1995) J Am Chem Soc 117:10174–10185
Bollinger JM, Krebs C, Vicol A, Chen S, Ley BA, Edmondson DE, Huynh BH (1998) J Am Chem Soc 120:1094–1095
Baldwin J, Krebs C, Saleh L, Stelling M, Huynh BH, Bollinger JM, Riggs-Gelasco P (2003) Biochemistry 42:13269–13279
Moënne-Loccoz P, Baldwin J, Ley BA, Loehr TM, Bollinger JM (1998) Biochemistry 37:14659–14663
Skulan AJ, Brunold TC, Baldwin J, Saleh L, Bollinger JM, Solomon EI (2004) J Am Chem Soc 126:8842–8855
Krebs C, Bollinger JM, Theil EC, Huynh BH (2002) J Biol Inorg Chem 7:863–869
Broadwater JA, Ai J, Loehr TM, Sanders-Loehr J, Fox BG (1998) Biochemistry 37:14664–14671
Broadwater JA, Achim C, Münck E, Fox BG (1999) Biochemistry 38:12197–12204
Hwang J, Krebs C, Huynh BH, Edmondson DE, Theil EC, Penner-Hahn JE (2000) Science 287:122–125
Pereira AS, Small W, Krebs C, Tavares P, Edmondson DE, Theil EC, Huynh BH (1998) Biochemistry 37:9871–9876
Solomon EI, Park, K (2016) J Biol Inorg Chem. doi:10.1007/s00775-016-1372-9
Solomon EI, Brunold TC, Davis MI, Kemsley JN, Lee S-K, Lehnert N, Neese F, Skulan AJ, Yang Y-S, Zhou J (2000) Chem Rev 100:235–350
Liu KE, Wang D, Huynh BH, Edmondson DE, Salifoglou A, Lippard SJ (1994) J Am Chem Soc 116:7465–7466
Korboukh VK, Li N, Barr EW, Bollinger JM, Krebs C (2009) J Am Chem Soc 131:13608–13609
Murray LJ, Naik SG, Ortillo DO, García-Serres R, Lee JK, Huynh BH, Lippard SJ (2007) J Am Chem Soc 129:14500–14510
Han Z, Sakai N, Böttger LH, Klinke S, Hauber J, Trautwein AX, Hilgenfeld R (2015) Structure 23:882–892
Rudd DJ, Sazinsky MH, Lippard SJ, Hedman B, Hodgson KO (2005) Inorg Chem 44:4546–4554
Shu L, Nesheim JC, Kauffmann K, Münck E, Lipscomb JD, Que L (1997) Science 275:515–518
Rudd DJ, Sazinsky MH, Merkx M, Lippard SJ, Hedman B, Hodgson KO (2004) Inorg Chem 43:4579–4589
Shu L, Liu Y, Lipscomb JD, Que L Jr (1996) J Biol Inorg Chem 1:297–304
Dassama LMK, Silakov A, Krest CM, Calixto JC, Krebs C, Bollinger JM, Green MT (2013) J Am Chem Soc 135:16758–16761
Griese JJ, Kositzki R, Schrapers P, Branca RMM, Nordström A, Lehtiö J, Haumann M, Högbom M (2015) J Biol Chem 290:25254–25272
Colpas GJ, Maroney MJ, Bagyinka C, Kumar M, Willis WS, Suib SL, Mascharak PK, Baidya N (1991) Inorg Chem 30:920–928
Wirt MD, Sagi I, Chen E, Frisbie SM, Lee R, Chance MR (1991) J Am Chem Soc 113:5299–5304
Penner-Hahn JE, Fronko RM, Pecoraro VL, Yocum CF, Betts SD, Bowlby NR (1990) J Am Chem Soc 112:2549–2557
Rudd DJ, Goldsmith CR, Cole AP, Stack TDP, Hodgson KO, Hedman B (2005) Inorg Chem 44:1221–1229
de Groot F (2001) Chem Rev 101:1779–1808
Sarangi R (2013) Coord Chem Rev 257:459–472
Randall CR, Shu L, Chiou Y-M, Hagen KS, Ito M, Kitajima N, Lachicotte RJ, Zang Y, Que L (1995) Inorg Chem 34:1036–1039
Cranswick MA, Meier KK, Shan X, Stubna A, Kaizer J, Mehn MP, Münck E, Que L (2012) Inorg Chem 51:10417–10426
Pap JS, Cranswick MA, Balogh-Hergovich E, Baráth G, Giorgi M, Rohde GT, Kaizer J, Speier G, Que L (2013) Eur J Inorg Chem 2013:3858–3866
Frisch JR, Vu VV, Martinho M, Münck E, Que L (2009) Inorg Chem 48:8325–8336
Westre TE, Kennepohl P, DeWitt JG, Hedman B, Hodgson KO, Solomon EI (1997) J Am Chem Soc 119:6297–6314
Carson EC, Lippard SJ (2006) Inorg Chem 45:837–848
Ou C-C, Lalancette RA, Potenza JA, Schugar HJ (1978) J Am Chem Soc 100:2053–2057
Schmitt W, Murugesu M, Goodwin JC, Hill JP, Mandel A, Bhalla R, Anson CE, Heath SL, Powell AK (2001) Polyhedron 20:1687–1697
Thich JA, Ou CC, Powers D, Vasiliou B, Mastropaolo D, Potenza JA, Schugar HJ (1976) J Am Chem Soc 98:1425–1433
Yoon S, Lippard SJ (2004) J Am Chem Soc 126:2666–2667
Makhlynets OV, Oloo WN, Moroz YS, Belaya IG, Palluccio TD, Filatov AS, Müller P, Cranswick MA, Que L, Rybak-Akimova EV (2014) Chem Commun 50:645–648
Yoon S, Lippard SJ (2005) J Am Chem Soc 127:8386–8397
Makris TM, Knoot CJ, Wilmot CM, Lipscomb JD (2013) Biochemistry 52:6662–6671
Whittington DA, Lippard SJ (2001) J Am Chem Soc 123:827–838
Elango N, Radhakrishnan R, Froland WA, Wallar BJ, Earhart CA, Lipscomb JD, Ohlendorf DH (1997) Protein Sci 6:556–568
Logan DT, Su XD, Aberg A, Regnstrom K, Hajdu J, Eklund H, Nordlund P (1996) Structure 4:1053–1064
Eriksson M, Jordan A, Eklund H (1998) Biochemistry 37:13359–13369
Lindqvist Y, Huang W, Schneider G, Shanklin J (1996) EMBO J 15:4081–4092
Holmes MA, Le Trong I, Turley S, Sieker LC, Stenkamp RE (1991) J Mol Biol 218:583–593
Holmes MA, Stenkamp RE (1991) J Mol Biol 220:723–737
Dong Y, Yan S, Young VG, Que L (1996) Angew Chem Int Ed 35:618–620
Ookubo T, Sugimoto H, Nagayama T, Masuda H, Sato T, Tanaka K, Maeda Y, Ōkawa H, Hayashi Y, Uehara A, Suzuki M (1996) J Am Chem Soc 118:701–702
Kim K, Lippard SJ (1996) J Am Chem Soc 118:4914–4915
Riggs-Gelasco PJ, Shu L, Chen S, Burdi D, Huynh BH, Que L, Stubbe J (1998) J Am Chem Soc 120:849–860
Korendovych IV, Kryatov SV, Reiff WM, Rybak-Akimova EV (2005) Inorg Chem 44:8656–8658
Kryatov SV, Taktak S, Korendovych IV, Rybak-Akimova EV, Kaizer J, Torelli S, Shan X, Mandal S, MacMurdo VL (2005) Mairata i Payeras A, Que L. Inorg Chem 44:85–99
Chaudhury P, Wieghardt K, Nuber B, Weiss J (1985) Angew Chem Int Ed 24:778–779
Yoon S, Lippard SJ (2004) J Am Chem Soc 126:16692–16693
Yoon S, Kelly AE, Lippard SJ (2004) Polyhedron 23:2805–2812
Mitra M, Lloret-Fillol J, Haukka M, Costas M, Nordlander E (2014) Chem Commun 50:1408–1410
Widger LR, Siegler MA, Goldberg DP (2013) Polyhedron 58:179–189
Zhang Z-T, Cheng X-L (2005) Acta Crystallogr C 61:m529–m531
Liang Y, Li W, Guo B-J (2005) Acta Crystallogr Sect E Struct Reports Online 61:m1782–m1784
Galet A, Muñoz MC, Agustí G, Martínez V, Gaspar AB, Real JA (2005) Z Anorg Allg Chem 631:2092–2095
Turowski PN, Armstrong WH, Liu S, Brown SN, Lippard SJ (1994) Inorg Chem 33:636–645
Zhang X, Furutachi H, Fujinami S, Nagatomo S, Maeda Y, Watanabe Y, Kitagawa T, Suzuki M (2005) J Am Chem Soc 127:826–827
Shakya R, Powell DR, Houser RP (2009) Eur J Inorg Chem 2009:5319–5327
Jozwiuk A, Ingram AL, Powell DR, Moubaraki B, Chilton NF, Murray KS, Houser RP (2014) Dalton Trans 43:9740–9753
Hazell A, Jensen KB, McKenzie CJ, Toftlund H (1994) Inorg Chem 33:3127–3134
Dong Y, Fujii H, Hendrich MP, Leising RA, Pan G, Randall CR, Wilkinson EC, Zang Y, Que L (1995) J Am Chem Soc 117:2778–2792
Ogo S, Wada S, Watanabe Y, Iwase M, Wada A, Harata M, Jitsukawa K, Masuda H, Einaga H (1998) Angew Chem Int Ed 37:2102–2104
MacBeth CE, Gupta R, Mitchell-Koch KR, Young VG, Lushington GH, Thompson WH, Hendrich MP, Borovik AS (2004) J Am Chem Soc 126:2556–2567
Yamashita M, Furutachi H, Tosha T, Fujinami S, Saito W, Maeda Y, Takahashi K, Tanaka K, Kitagawa T, Suzuki M (2007) J Am Chem Soc 129:2–3
Celenligil-Cetin R, Paraskevopoulou P, Dinda R, Staples RJ, Sinn E, Rath NP, Stavropoulos P (2008) Inorg Chem 47:1165–1172
Mukherjee J, Lucas RL, Zart MK, Powell DR, Day VW, Borovik AS (2008) Inorg Chem 47:5780–5786
Majumdar A, Lippard SJ (2013) Inorg Chem 52:13292–13294
Majumdar A, Apfel U-P, Jiang Y, Moënne-Loccoz P, Lippard SJ (2014) Inorg Chem 53:167–181
Armstrong WH, Lippard SJ (1984) J Am Chem Soc 106:4632–4633
DeWitt JG, Bentsen JG, Rosenzweig AC, Hedman B, Green J, Pilkington S, Papaefthymiou GC, Dalton H, Hodgson KO, Lippard SJ (1991) J Am Chem Soc 113:9219–9235
Scarrow RC, Maroney MJ, Palmer SM, Que L, Roe AL, Salowe SP, Stubbe J (1987) J Am Chem Soc 109:7857–7864
Vu VV, Makris TM, Lipscomb JD, Que L (2011) J Am Chem Soc 133:6938–6941
Cramer SP, Hodgson KO, Stiefel EI, Newton WE (1978) J Am Chem Soc 100:2748–2761
Sousa CM, Carpentier P, Matias PM, Testa F, Pinho F, Sarti P, Giuffrè A, Bandeiras TM, Romão CV (2015) Acta Crystallogr D Biol Crystallogr 71:2236–2247
Gudmundsson M, Kim S, Wu M, Ishida T, Momeni MH, Vaaje-Kolstad G, Lundberg D, Royant A, Ståhlberg J, Eijsink VGH, Beckham GT, Sandgren M (2014) J Biol Chem 289:18782–18792
Sigfridsson KGV, Chernev P, Leidel N, Popovic-Bijelic A, Gräslund A, Haumann M (2013) J Biol Chem 288:9648–9661
Bai Y, McCoy JG, Levin EJ, Sobrado P, Rajashankar KR, Fox BG, Zhou M (2015) Nature 524:252–256
Rosenzweig AC, Nordlund P, Takahara PM, Frederick CA, Lippard SJ (1995) Chem Biol 2:409–418
Nordlund P, Eklund H (1993) J Mol Biol 232:123–164
Guy JE, Whittle E, Kumaran D, Lindqvist Y, Shanklin J (2007) J Biol Chem 282:19863–19871
Shu L, Broadwater JA, Achim C, Fox BG, Münck E, Que L Jr (1998) J Biol Inorg Chem 3:392–400
Rardin RL, Tolman WB, Lippard SJ (1991) New J Chem 15:417–430
Banerjee R, Proshlyakov Y, Lipscomb JD, Proshlyakov DA (2015) Nature 518:431–434
Magnus KA, Hazes B, Ton-That H, Bonaventura C, Bonaventura J, Hol WG (1994) Proteins 19:302–309
Kitajima N, Fujisawa K, Fujimoto C, Morooka Y, Hashimoto S, Kitagawa T, Toriumi K, Tatsumi K, Nakamura A (1992) J Am Chem Soc 114:1277–1291
Lewis EA, Tolman WB (2004) Chem Rev 104:1047–1076
Funahashi Y, Nishikawa T, Wasada-Tsutsui Y, Kajita Y, Yamaguchi S, Arii H, Ozawa T, Jitsukawa K, Tosha T, Hirota S, Kitagawa T, Masuda H (2008) J Am Chem Soc 130:16444–16445
Xue G, Fiedler AT, Martinho M, Munck E, Que L (2008) Proc Natl Acad Sci 105:20615–20620
Xue G, Geng C, Ye S, Fiedler AT, Neese F, Que L (2013) Inorg Chem 52:3976–3984
Xue G, De Hont R, Münck E, Que L (2010) Nat Chem 2:400–405
Siegbahn PEM, Crabtree RH (1997) J Am Chem Soc 119:3103–3113
Rinaldo D, Philipp DM, Lippard SJ, Friesner RA (2007) J Am Chem Soc 129:3135–3147
Moënne-Loccoz P, Krebs C, Herlihy K, Edmondson DE, Theil EC, Huynh BH, Loehr TM (1999) Biochemistry 38:5290–5295
Murray LJ, García-Serres R, Naik S, Huynh BH, Lippard SJ (2006) J Am Chem Soc 128:7458–7459
Park JH, Dias CAO, Lee SB, Valentini SR, Sokabe M, Fraser CS, Park MH (2011) Protein Eng Des Sel 24:301–309
George GN (1990) EXAFSPAK: A suite of computer programs for analysis of X-ray absorption spectra. http://ssrl.slac.stanford.edu/exafspak.html. Accessed 28 June 2016
Ankudinov AL, Ravel B, Rehr JJ, Conradson SD (1998) Phys Rev B 58:7565–7576
Wojdyr M (2010) J Appl Crystallogr 43:1126–1128
Knoot CJ, Kovaleva EG, Lipscomb JD (2016) J Biol Inorg Chem. doi:10.1007/s00775-016-1363-x
Komor AJ, Rivard BS, Fan R, Guo Y, Que L Jr, Lipscomb JD (2016) J Am Chem Soc 138:7411–7421
Acknowledgments
This work was supported by the National Institutes of Health grant GM-38767 to L. Q. and postdoctoral fellowship 5F32GM106612-02 to L. M. E. and by the Intramural Research Program of the National Institute of Dental and Craniofacial Research. V. V. V. is grateful to the Vietnam Education Foundation for providing him with a predoctoral fellowship. XAS data were collected on Beamline 7–3 at the Stanford Synchrotron Radiation Lightsource, SLAC National Accelerator Laboratory and at Beamline X3B of the National Synchrotron Light Source (NSLS). SLAC is supported by the US Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences under Contract No. DE-AC02-76SF00515. Use of Beamline 7–3 is supported by the DOE Office of Biological and Environmental Research, and by the National Institutes of Health, National Institute of General Medical Sciences (including P41GM103393). NSLS is a DOE Office of Science User Facility operated for the DOE Office of Science by Brookhaven National Laboratory under Contract No. DE-AC02-98CH10886. We thank Dr. Caleb Allpress for helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jasniewski, A.J., Engstrom, L.M., Vu, V.V. et al. X-ray absorption spectroscopic characterization of the diferric-peroxo intermediate of human deoxyhypusine hydroxylase in the presence of its substrate eIF5a. J Biol Inorg Chem 21, 605–618 (2016). https://doi.org/10.1007/s00775-016-1373-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-016-1373-8