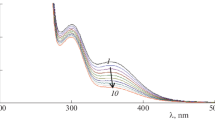
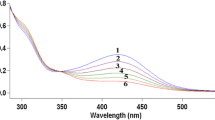
Abstract
The kinetics of the oxidation of ruthenium(III)-catalyzed oxidation of pentoxifylline (PTX) by diperiodatocuprate(III) (DPC) in aqueous alkaline medium at a constant ionic strength of 0.30 mol dm−3 was studied spectrophotometrically. The reaction between PTX and DPC in alkaline medium in the presence of Ru(III) exhibits 1:2 stoichiometry (PTX:DPC). The reaction was of first order in DPC, less than the unit order in [PTX] and [OH−] and negative fractional order in [IO4 −]. The order in [Ru(III)] was unity. Intervention of free radicals was observed in the reaction. The main products were identified by TLC and spectral studies including LC-MS. The oxidation reaction in alkaline medium has been shown to proceed via a Ru(III)-PTX complex, which reacts with monoperiodatocuprate(III) to decompose in a rate determining step followed by a fast step to give the products. The reaction constants involved in different steps of the mechanism were calculated. The activation parameters with respect to the slow step of the mechanism were computed and discussed, and thermodynamic quantities were also determined. The active species of catalyst and oxidant have been identified.
Graphical abstract










Similar content being viewed by others
References
Reddy KB (1990) Transition Met Chem 15:9
Sethuram B (2003) Some aspects of electron transfer reactions involving organic molecules. Allied Publishers (P) Ltd, New Delhi
Delanian S, Porcher R, Rudant J, Lefaix J-L (2005) J Clin Oncol 23:8570
Rott O, Cash E, Fleischer B (2005) Eur J Immun 23:1745
Berman B, Duncan MR (1989) J Invest Derm 92:605
Das AK (2001) Coord Chem Revs 213:307
Shivananda KN, Lakshmi B, Jagdeesh RV, Puttaswamy, Mahendra KN (2007) Appl Cat A Gen 326:202
Tandon PK, Mehrotra A, Shrivastava M, Dhusia M, Singh SB (2007) Transition Met Chem 32:991
Singh AK, Srivastava S, Srivastava J, Srivastava R, Singh P (2007) J Mol Cat A Chem 278:72
Shetti NP, Hosamani RR, Nandibewoor ST (2009) Open Cat J 2:112
Jagdeesh RV, Puttaswamy (2008) J Phys Org Chem 21:844
Gavars R, Baumane L, Stradyn Y, Chekavichus B, Duburs G (1998) Chem Heterocycl Comp 34:950
Moelwyn-Hughes EA (1947) Kinetics of reactions in solutions. Oxford University Press, London, p 297
Reddy KB, Sethuram B, Navneeth Rao T (1987) Z Phys Chem 268:706
Bailar JC Jr, Emelens HJ, Nyholm SR, Trotman-Dikenson AF (1975) Comprehensive inorganic chemistry, vol 2. Pergamon Press, Oxford, p 1456
Niu W, Zhu Y, Hu K, Tong C, Yang H (1996) Int J Chem Kinet 28:899
Jose T, Tuwar SM (2007) J Mol Struct 827:137
Connick RE, Fine DA (1960) J Am Chem Soc 82:4187
Cotton FA, Wilkinson G, Murillo CA, Bochmann M (1999) Advanced inorganic chemistry, 6th edn. Wiley, New York
Hiremath DC, Kiran TS, Nandibewoor ST (2007) Int J Chem Kinet 39:236
Vinod KN, Puttaswamy, Ninge Gowda KN (2009) J Mol Cat A General 298:60
Rangappa KS, Ragavendra MP, Mahadevappa DS, Channegouda D (1998) J Org Chem 63:531
Weissberger A, Lewis ES (eds) (1974) Investigation of rates and mechanism of reactions in techniques of chemistry, vol 4. Wiley, New York, p 421
Farokhi SA, Nandibewoor ST (2003) Tetrahedron 59:7595
Kiran TS, Hiremath CV, Nandibewoor ST (2006) Appl Cat A Gen 305:79
Reddy CS, Vijay Kumar T (1995) Indian J Chem 34:615
Murthy CP, Sethuram B, Navaneeth Rao T (1981) Z Phys Chem 262:252
Jeffery GH, Bassett J, Mendham J, Denny RC (1996) Vogel’s text book of quantitative chemical analysis, ELBS, 5th edn. Longman, Essex, p 455
Panigrahi GP, Misro PK (1978) Indian J Chem 16:762
Feigl F (1975) Spot tests in organic analysis. Elsevier, New York, p 333
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
According to Scheme 1,
The total concentration of DPC, [DPC]T, is given by (subscripts T and f stand for total and free, respectively)
Similarly,
In view of the low concentrations of Ru(III) used, we have
Similarly,
Substituting Eqs. (12), (13), (14), and (15) in Eq. (11) and omitting the T and f subscripts, we get
The terms (K 3[H3IO6 2−][PTX]) and (K 1 K 3[PTX][OH−][H3IO6 2−]) of the denominator are neglected in view of their lower values as compared to the periodate used in the study. Therefore,
Rights and permissions
About this article
Cite this article
Malode, S.J., Abbar, J.C. & Nandibewoor, S.T. Mechanistic investigations of ruthenium(III) catalyzed oxidation of pentoxifylline by copper(III) periodate complex in aqueous alkaline medium. Monatsh Chem 142, 469–479 (2011). https://doi.org/10.1007/s00706-011-0458-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-011-0458-x