Abstract
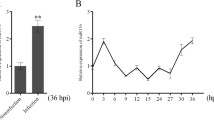
Influenza A virus (IAV) infections result in a large number of deaths and substantial economic losses each year. MicroRNAs repress gene expression and are involved in virus-host interactions. miR-29a is known to have anti-tumor and anti-fibrotic effects. However, the role of miR-29a in IAV infection is unclear. In the present study, we investigated the effect of miR-29a on IAV infection and the mechanisms by which it functions. IAV infection was found to cause decreased miR-29a expression in lung epithelial A549 cells and mouse lungs. Overexpression of miR-29a reduced IAV mRNA and protein levels and progeny virus production in HEK293 and A549 cells. Inhibition of IAV infection by miR-29a was observed with different strains of IAV, including A/PR/8/34, A/WSN/1933, and clinical isolates A/OK/3052/09 and A/OK/309/06 H3N2. Knockout of miR-29a using CRISPR/Cas9 resulted in an increase in viral mRNA and protein levels, confirming that miR-29a suppresses IAV infection. A 3’ untranslated region (3’-UTR) reporter assay showed that miR-29a had binding sites in the 3’-UTR of the Wnt-Ca2+ signaling receptor frizzled 5 gene, and overexpression of miR-29a reduced the level of the endogenous frizzled 5 protein. Wnt5a treatment of HEK293 and A549 cells enhanced IAV infection. Our results suggest that miR-29a inhibits IAV infection, probably via the frizzled 5 receptor.







Similar content being viewed by others
References
Shrestha S et al (2015) The role of influenza in the epidemiology of pneumonia. Sci Rep 5:15314
Taubenberger JK, Morens DM (2008) The pathology of influenza virus infections. Annu Rev Pathol 3:499–522
Wang Y et al (2007) MicroRNA: past and present. Front Biosci 12:2316–2329
Peng S et al (2018) Endogenous cellular MicroRNAs mediate antiviral defense against influenza A virus. Mol Ther Nucleic Acids 10:361–375
Song L et al (2010) Cellular microRNAs inhibit replication of the H1N1 influenza A virus in infected cells. J Virol 84(17):8849–8860
Ma YJ et al (2012) Cellular microRNA let-7c inhibits M1 protein expression of the H1N1 influenza A virus in infected human lung epithelial cells. J Cell Mol Med 16(10):2539–2546
Gui S et al (2015) Mir-302c mediates influenza A virus-induced IFNbeta expression by targeting NF-kappaB inducing kinase. FEBS Lett 589(24 Pt B):4112–4118
Ingle H et al (2015) The microRNA miR-485 targets host and influenza virus transcripts to regulate antiviral immunity and restrict viral replication. Sci Signal 8(406):ra126
Maemura T et al (2018) Lung-derived exosomal miR-483-3p regulates the innate immune response to influenza virus infection. J Infect Dis 217(9):1372–1382
Guan Z et al (2012) Induction of the cellular microRNA-29c by influenza virus contributes to virus-mediated apoptosis through repression of antiapoptotic factors BCL2L2. Biochem Biophys Res Commun 425(3):662–667
Pociask DA et al (2017) Epigenetic and transcriptomic regulation of lung repair during recovery from influenza infection. Am J Pathol 187(4):851–863
Preusse M, Schughart K, Pessler F (2017) Host genetic background strongly affects pulmonary microRNA expression before and during influenza A virus infection. Front Immunol 8:246
Wang Y et al (2009) Identification of differentially expressed miRNAs in chicken lung and trachea with avian influenza virus infection by a deep sequencing approach. BMC Genomics 10:512
Wang Y et al (2012a) Integrated analysis of microRNA expression and mRNA transcriptome in lungs of avian influenza virus infected broilers. BMC Genomics 13:278
Li Y et al (2010) MicroRNA expression and virulence in pandemic influenza virus-infected mice. J Virol 84(6):3023–3032
Rogers JV et al (2012) Preliminary microRNA analysis in lung tissue to identify potential therapeutic targets against H5N1 infection. Viral Immunol 25(1):3–11
Wu Z et al (2013) The inhibitory role of Mir-29 in growth of breast cancer cells. J Exp Clin Cancer Res 32:98
Zhang M et al (2016) Negative regulation of CDC42 expression and cell cycle progression by miR-29a in breast cancer. Open Med (Wars) 11(1):78–82
Liu X et al (2015) MicroRNA-29a inhibits cell migration and invasion via targeting Roundabout homolog 1 in gastric cancer cells. Mol Med Rep 12(3):3944–3950
Chen L et al (2014) miR-29a suppresses growth and invasion of gastric cancer cells in vitro by targeting VEGF-A. BMB Rep 47(1):39–44
Shi C et al (2017) miR-29a/b/c function as invasion suppressors for gliomas by targeting CDC42 and predict the prognosis of patients. Br J Cancer 117(7):1036–1047
Wang XS et al (2012b) MicroRNA-29a and microRNA-142-3p are regulators of myeloid differentiation and acute myeloid leukemia. Blood 119(21):4992–5004
Lu L et al (2014) MicroRNA-29a upregulates MMP2 in oral squamous cell carcinoma to promote cancer invasion and anti-apoptosis. Biomed Pharmacother 68(1):13–19
Pei YF, Lei Y, Liu XQ (2016) MiR-29a promotes cell proliferation and EMT in breast cancer by targeting ten eleven translocation 1. Biochim Biophys Acta 1862(11):2177–2185
Matsumoto Y et al (2016) MiR-29a assists in preventing the activation of human stellate cells and promotes recovery from liver fibrosis in mice. Mol Ther 24(10):1848–1859
Tan J et al (2014) MicroRNA-29 mediates TGFbeta1-induced extracellular matrix synthesis by targeting wnt/beta-catenin pathway in human orbital fibroblasts. Int J Clin Exp Pathol 7(11):7571–7577
Yang T et al (2013) miR-29 mediates TGFbeta1-induced extracellular matrix synthesis through activation of PI3K-AKT pathway in human lung fibroblasts. J Cell Biochem 114(6):1336–1342
Li SC et al (2016) Microarray study of pathway analysis expression profile associated with microRNA-29a with regard to murine cholestatic liver injuries. Int J Mol Sci 17(3):324
Luo Y et al (2015) miR-29a-3p attenuates hypoxic pulmonary hypertension by inhibiting pulmonary adventitial fibroblast activation. Hypertension 65(2):414–420
Tang B et al (2017) MicroRNA-29a regulates lipopolysaccharide (LPS)-induced inflammatory responses in murine macrophages through the Akt1/ NF-kappaB pathway. Exp Cell Res 360(2):74–80
Kong G et al (2011) Upregulated microRNA-29a by hepatitis B virus X protein enhances hepatoma cell migration by targeting PTEN in cell culture model. PLoS ONE 6(5):e19518
Brogaard L et al (2016) Late regulation of immune genes and microRNAs in circulating leukocytes in a pig model of influenza A (H1N2) infection. Sci Rep 6:21812
Song H et al (2013) Microarray analysis of microRNA expression in peripheral blood mononuclear cells of critically ill patients with influenza A (H1N1). BMC Infect Dis 13:257
Zhu Z et al (2014) Comprehensive characterization of serum microRNA profile in response to the emerging avian influenza A (H7N9) virus infection in humans. Viruses 6(4):1525–1539
Tambyah PA et al (2013) microRNAs in circulation are altered in response to influenza A virus infection in humans. PLoS ONE 8(10):e76811
Yang X et al (2019) miR-193b represses influenza A virus infection by inhibiting Wnt/beta-catenin signalling. Cell Microbiol 21(5):e13001
Guo Y et al (2014) Wnt3a mitigates acute lung injury by reducing P2X7 receptor-mediated alveolar epithelial type I cell death. Cell Death Dis 5:e1286
Matsuoka Y et al (2013) A comprehensive map of the influenza A virus replication cycle. BMC Syst Biol 7:97
Yan D et al (2013) Dual myxovirus screen identifies a small-molecule agonist of the host antiviral response. J Virol 87(20):11076–11087
Bottini A et al (2012) Identification of small molecules that interfere with H1N1 influenza A viral replication. ChemMedChem 7(12):2227–2235
Lutz A et al (2005) Virus-inducible reporter genes as a tool for detecting and quantifying influenza A virus replication. J Virol Methods 126(1–2):13–20
Chang H et al (2016) CRISPR/cas9, a novel genomic tool to knock down microRNA in vitro and in vivo. Sci Rep 6:22312
Baarsma HA, Konigshoff M (2017) “WNT-er is coming”: WNT signalling in chronic lung diseases. Thorax 72(8):746–759
Blumenthal A et al (2006) The Wingless homolog WNT5A and its receptor Frizzled-5 regulate inflammatory responses of human mononuclear cells induced by microbial stimulation. Blood 108(3):965–973
Kuhl M et al (2000) Ca(2+)/calmodulin-dependent protein kinase II is stimulated by Wnt and Frizzled homologs and promotes ventral cell fates in Xenopus. J Biol Chem 275(17):12701–12711
Liu C et al (2016) A secreted WNT-ligand-binding domain of FZD5 generated by a frameshift mutation causes autosomal dominant coloboma. Hum Mol Genet 25(7):1382–1391
Fang J et al (2012) Epigenetic changes mediated by microRNA miR29 activate cyclooxygenase 2 and lambda-1 interferon production during viral infection. J Virol 86(2):1010–1020
Zhang X et al (2014) Induction of the cellular miR-29c by influenza virus inhibits the innate immune response through protection of A20 mRNA. Biochem Biophys Res Commun 450(1):755–761
Cavodeassi F et al (2005) Early stages of zebrafish eye formation require the coordinated activity of Wnt11, Fz5, and the Wnt/beta-catenin pathway. Neuron 47(1):43–56
Liu C, Nathans J (2008) An essential role for frizzled 5 in mammalian ocular development. Development 135(21):3567–3576
Pandit KV, Milosevic J, Kaminski N (2011) MicroRNAs in idiopathic pulmonary fibrosis. Transl Res 157(4):191–199
Bibaki E et al (2018) miR-185 and miR-29a are similarly expressed in the bronchoalveolar lavage cells in IPF and lung cancer but common targets DNMT1 and COL1A1 show disease specific patterns. Mol Med Rep 17(5):7105–7112
Kawashita Y et al (2011) Circulating miR-29a levels in patients with scleroderma spectrum disorder. J Dermatol Sci 61(1):67–69
Roderburg C et al (2011) Micro-RNA profiling reveals a role for miR-29 in human and murine liver fibrosis. Hepatology 53(1):209–218
Xuan J et al (2017) MiR-29a and miR-652 attenuate liver fibrosis by inhibiting the differentiation of CD4+ T cells. Cell Struct Funct 42(2):95–103
Xiao J et al (2012) miR-29 inhibits bleomycin-induced pulmonary fibrosis in mice. Mol Ther 20(6):1251–1260
He Y et al (2013) MicroRNA-29 family, a crucial therapeutic target for fibrosis diseases. Biochimie 95(7):1355–1359
Hsu YC et al (2016) Protective effects of miR-29a on diabetic glomerular dysfunction by modulation of DKK1/Wnt/beta-catenin signaling. Sci Rep 6:30575
Nguyen-Van-Tam JS et al (2010) Risk factors for hospitalisation and poor outcome with pandemic A/H1N1 influenza: United Kingdom first wave (May-September 2009). Thorax 65(7):645–651
Li P et al (2012) Serial evaluation of high-resolution CT findings in patients with pneumonia in novel swine-origin influenza A (H1N1) virus infection. Br J Radiol 85(1014):729–735
Singh V, Sharma BB, Patel V (2012) Pulmonary sequelae in a patient recovered from swine flu. Lung India 29(3):277–279
Kovner AV et al (2012) Structural and functional changes in pulmonary macrophages and lungs of mice infected with influenza virus A/H5N1 A/goose/Krasnoozerskoye/627/05. Bull Exp Biol Med 153(2):229–232
Qiao J et al (2009) Pulmonary fibrosis induced by H5N1 viral infection in mice. Respir Res 10:107
Acknowledgements
We thank Dr. Gillian Air (University of Oklahoma Health Sciences Center) for kindly providing A/Oklahoma/3052/09 H1N1, A/WSN/1933 H1N1 and A/Oklahoma/309/2006 H3N2. lentiCRISPR v2 was a gift from Dr. Feng Zhang (Addgene plasmid # 52961).
Funding
This work was supported by National Institutes of Health grants AI121591, GM103648 and HL135152, the Oklahoma Center for Advancement of Science and Technology HR20-050, the Oklahoma Center for Adult Stem Cell Research – A Program of the Tobacco Settlement Endowment Trust (TSET), and the Lundberg-Kienlen Endowment Fund (to LL).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants performed by any of the authors. The animal procedures were approved by the Institutional Animal Care and Use Committee at Oklahoma State University.
Additional information
Handling Editor: Ayato Takada.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, X., Liang, Y., Bamunuarachchi, G. et al. miR-29a is a negative regulator of influenza virus infection through targeting of the frizzled 5 receptor. Arch Virol 166, 363–373 (2021). https://doi.org/10.1007/s00705-020-04877-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00705-020-04877-z