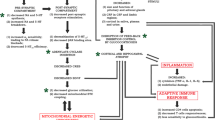
Abstract
Hypoxia-inducible factor 1 has been identified as an important therapeutic target in psychiatric illnesses. Hypoxia is a condition in which tissues do not receive enough oxygen, resulting in less oxidative energy production. HIF-1, the master regulator of molecular response to hypoxia, is destabilized when oxygen levels fall. HIF-1, when activated, increases the gene transcription factors that promote adaptive response and longevity in hypoxia. HIF-regulated genes encode proteins involved in cell survival, energy metabolism, angiogenesis, erythropoiesis, and vasomotor control. Multiple genetic and environmental variables contribute to the pathophysiology of psychiatric disease. This review focuses on the most recent findings indicating the role of oxygen deprivation in CNS damage, with strong attention on HIF-mediated pathways. Several pieces of evidence suggested that, in the case of hypoxia, induction and maintenance of HIF-1 target genes may help reduce nerve damage. Major new insights into the molecular mechanisms that control HIF's sensitivity to oxygen are used to make drugs that can change the way HIF works as a therapeutic target for some CNS diseases.



Similar content being viewed by others
Data availability
Not applicable.
References
Amodeo G, Trusso MA, Fagiolini A (2017) Depression and inflammation: disentangling a clear yet complex and multifaceted link. Neuropsychiatry 7(4):448–457
Arora B, Khan H, Grewal AK, Singh TG (2023) Mechanistic insights on the role of nitric oxide in ischemia-reperfusion injury. InNitric oxide in health and disease. Academic Press, pp 275–285. https://doi.org/10.1016/B978-0-443-13342-8.00004-1
Bauer ME, Teixeira AL (2019) Inflammation in psychiatric disorders: what comes first? Ann N Y Acad Sci 1437(1):57–67. https://doi.org/10.1111/nyas.13712
Behl T, Kaur D, Sehgal A, Singh S, Sharma N, Zengin G, Andronie-Cioara FL et al (2021a) Role of monoamine oxidase activity in Alzheimer’s disease: an insight into the therapeutic potential of inhibitors. Molecules 26(12):3724. https://doi.org/10.3390/molecules26123724
Behl T, Kaur G, Sehgal A, Bhardwaj S, Singh S, Buhas C, Judea-Pusta C et al (2021b) Multifaceted role of matrix metalloproteinases in neurodegenerative diseases: pathophysiological and therapeutic perspectives. Int J Mol Sci 22(3):1413. https://doi.org/10.3390/ijms22031413
Bray NJ, O’Donovan MC (2018) The genetics of neuropsychiatric disorders. Brain Neurosci Adv. https://doi.org/10.1177/2398212818799271
Brocker C, Thompson D, Matsumoto A, Nebert DW, Vasiliou V (2010) Evolutionary divergence and functions of the human interleukin (IL) gene family. Hum Genomics 5(1):1–26. https://doi.org/10.1186/1479-7364-5-1-30
Brockmann MD, Kukovic M, Schönfeld M, Sedlacik J, Hanganu-Opatz IL (2013) Hypoxia-ischemia disrupts directed interactions within neonatal prefrontal-hippocampal networks. PLoS ONE 8(12):e83074. https://doi.org/10.1371/journal.pone.0083074
Deng Q, Chang Y, Cheng X, Luo X, Zhang J, Tang X (2018) Postconditioning with repeated mild hypoxia protects neonatal hypoxia-ischemic rats against brain damage and promotes rehabilitation of brain function. Brain Res Bull 139:31–37. https://doi.org/10.1016/j.brainresbull.2018.02.006
Depression WHO (2017) Other common mental disorders: global health estimates. World Health Organization, Geneva, p 24
Duelli R, Enerson BE, Gerhart DZ, Drewes LR (2000) Expression of large amino acid transporter LAT1 in rat brain endothelium. J Cereb Blood Flow Metab 20(11):1557–1562. https://doi.org/10.1097/00004647-200011000-00005
Duman RS, Monteggia LM (2006) A neurotrophic model for stress-related mood disorders. Biol Psychiatry 59(12):1116–1127. https://doi.org/10.1016/j.biopsych.2006.02.013
Edition F (2013) Diagnostic and statistical manual of mental disorders. Am Psychiatric Assoc 21(21):591–643
Eftekharian MM, Ghafouri-Fard S, Noroozi R, Omrani MD, Arsang-Jang S, Ganji M et al (2018) Cytokine profile in autistic patients. Cytokine 108:120–126. https://doi.org/10.1016/j.cyto.2018.03.034
Ehrenreich H, Degner D, Meller J, Brines M, Behe M, Hasselblatt M et al (2004) Erythropoietin: a candidate compound for neuroprotection in schizophrenia. Mol Psychiatry 9(1):42–54. https://doi.org/10.1038/sj.mp.4001442
Eissa N, Sadeq A, Sasse A, Sadek B (2020) Role of neuroinflammation in autism spectrum disorder and the emergence of brain histaminergic system. Lessons also for BPSD. Front Pharmacol 11:886. https://doi.org/10.3389/fphar.2020.00886
Fan T, Hu Y, Xin J, Zhao M, Wang J (2020) Analyzing the genes and pathways related to major depressive disorder via a systems biology approach. Brain Behav 10(2):e01502. https://doi.org/10.1002/brb3.1502
Flügel D, Görlach A, Michiels C, Kietzmann T (2007) Glycogen synthase kinase-3 phosphorylates hypoxia-inducible factor 1alpha and mediates its destabilization in a VHL-independent manner. Mol Cell Biol 27:3253–3265. https://doi.org/10.1128/MCB.00015-07
Gu YZ, Hogenesch JB, Bradfield CA (2000) The PAS superfamily: sensors of environmental and developmental signals. Annu Rev Pharmacol Toxicol 40(1):519–561
Guo S, Miyake M, Liu KJ, Shi H (2009) Specific inhibition of hypoxia inducible factor 1 exaggerates cell injury induced by in vitro ischemia through deteriorating cellular redox environment. J Neurochem 108(5):1309–1321. https://doi.org/10.1111/j.1471-4159.2009.05877.x
Gupta A, Khan H, Kaur A, Singh TG (2021) Novel targets explored in the treatment of alcohol withdrawal syndrome. CNS Neurol Disord Drug Targets 20(2):158–173. https://doi.org/10.2174/1871527319999201118155721
Henderson M, Harvey SB, Øverland S, Mykletun A, Hotopf M (2011) Work and common psychiatric disorders. J R Soc Med 104(5):198–207. https://doi.org/10.1258/jrsm.2011.100231
Hollander JA, Cory-Slechta DA, Jacka FN, Szabo ST, Guilarte TR, Bilbo SD et al (2020) Beyond the looking glass: recent advances in understanding the impact of environmental exposures on neuropsychiatric disease. Neuropsychopharmacol 45(7):1086–1096. https://doi.org/10.1038/s41386-020-0648-5
Hollocks MJ, Lerh JW, Magiati I, Meiser-Stedman R, Brugha TS (2018) Anxiety and depression in adults with autism spectrum disorder: a systematic review and meta-analysis. Psychol Med. https://doi.org/10.1017/s0033291718002283
Howlin P (2000) Outcome in adult life for more able individuals with autism or Asperger syndrome. Autism 4(1):63–83. https://doi.org/10.1177/1362361300004001005
Hu CJ, Wang LY, Chodosh LA, Keith B, Simon MC (2003) Differential roles of hypoxia-inducible factor 1α (HIF-1α) and HIF-2α in hypoxic gene regulation. Mol Cell Biol 23(24):9361–9374. https://doi.org/10.1128/MCB.23.24.9361-9374.2003
Isung J, Aeinehband S, Mobarrez F, Mårtensson B, Nordström P, Åsberg M, Piehl F et al (2012) Low vascular endothelial growth factor and interleukin-8 in cerebrospinal fluid of suicide attempters. Transl Psychiatry 2(11):e196–e196. https://doi.org/10.1038/tp.2012.123
Kaelin WG, Ratcliffe PJ (2008) Oxygen sensing by metazoans: the central role of the HIF hydroxylase pathway. Mol Cell 30(4):393–402. https://doi.org/10.1016/j.molcel.2008.04.009
Kalra P, Khan H, Kaur A, Singh TG (2022) Mechanistic insight on autophagy modulated molecular pathways in cerebral ischemic injury: from preclinical to clinical perspective. Neurochem Res. https://doi.org/10.1007/s11064-021-03500-0
Kamura T, Sato S, Iwai K, Czyzyk-Krzeska M, Conaway RC, Conaway JW (2000) Activation of HIF1α ubiquitination by a reconstituted von Hippel-Lindau (VHL) tumor suppressor complex. Proc Natl Acad Sci 97(19):10430–10435. https://doi.org/10.1073/pnas.190332597
Kang I, Kondo D, Kim J, Lyoo IK, Yurgelun-Todd D, Hwang J et al (2021) Elevating the level of hypoxia inducible factor may be a new potential target for the treatment of depression. Med Hypotheses 146:110398. https://doi.org/10.1016/j.mehy.2020.110398
Kast RE (2008) How lithium treatment generates neutrophilia by enhancing phosphorylation of GSK-3, increasing HIF-1 levels and how this path is important during engraftment. Bone Marrow Transplant 41:23–26. https://doi.org/10.1038/sj.bmt.1705872
Kaur A, Singh TG, Khan H, Kumar M, Singh N, Abdel-Daim MM (2022) Neuroprotective effect of piclamilast-induced post-ischemia pharmacological treatment in mice. Neurochem Res. https://doi.org/10.1007/s11064-022-03609-w
Ke Q, Costa M (2006) Hypoxia-inducible factor-1 (HIF-1). Mol Pharmacol 70(5):1469–1480. https://doi.org/10.1124/mol.106.027029
Kewley RJ, Whitelaw ML, Chapman-Smith A (2004) The mammalian basic helix–loop–helix/PAS family of transcriptional regulators. Int J Biochem Cell Biol 36(2):189–204. https://doi.org/10.1016/S1357-2725(03)00211-5
Khan H, Gupta A, Singh TG, Kaur A (2021a) Mechanistic insight on the role of leukotriene receptors in ischemic–reperfusion injury. Pharmacol Rep 73:1240–1254. https://doi.org/10.1007/s43440-021-00258-8
Khan H, Singh A, Thapa K, Garg N, Grewal AK, Singh TG (2021b) Therapeutic modulation of the phosphatidylinositol 3-kinases (PI3K) pathway in cerebral ischemic injury. Brain Res J 1761:147399. https://doi.org/10.1016/j.brainres.2021.147399
Khan H, Bangar A, Grewal AK, Bansal P, Singh TG (2022a) Caspase-mediated regulation of the distinct signaling pathways and mechanisms in neuronal survival. Int Immunopharmacol 110:108951. https://doi.org/10.1016/j.intimp.2022.108951
Khan H, Garg N, Singh TG, Kaur A, Thapa K (2022b) Calpain inhibitors as potential therapeutic modulators in neurodegenerative diseases. Neurochem Res 47(5):1125–1149. https://doi.org/10.1007/s11064-021-03521-9
Khan H, Grewal AK, Singh TG (2022c) Mitochondrial dynamics related neurovascular approaches in cerebral ischemic injury. Mitochondrion 66:54–66. https://doi.org/10.1016/j.mito.2022.08.001
Khan H, Grewal AK, Singh TG (2022d) Pharmacological postconditioning by protocatechuic acid attenuates brain injury in ischemia-reperfusion (I/R) mice model: implications of nuclear factor erythroid-2-related factor pathway. Neuroscience 491:23–31. https://doi.org/10.1016/j.neuroscience.2022.03.016
Khan H, Singh TG, Dahiya RS, Abdel-Daim MM (2022e) α-Lipoic acid, an organosulfur biomolecule a novel therapeutic agent for neurodegenerative disorders: an mechanistic perspective. Neurochem Res 47(7):1853–1864. https://doi.org/10.1007/s11064-022-03598-w
Leconte C, Léger M, Boulouard M, Tixier E, Fréret T, Bernaudin M et al (2012) Repeated mild hypoxic exposures decrease anxiety-like behavior in the adult mouse together with an increased brain adrenomedullin gene expression. Behav Brain Res 230(1):78–84. https://doi.org/10.1016/j.bbr.2012.01.054
Li Y, Zhong W, Jiang Z, Tang X (2019) New progress in the approaches for blood–brain barrier protection in acute ischemic stroke. Brain Res Bull 144:46–57. https://doi.org/10.1016/j.brainresbull.2018.11.006
Li G, Zhao M, Cheng X, Zhao T, Feng Z, Zhao Y et al (2020) FG-4592 improves depressive-like behaviors through HIF-1-mediated neurogenesis and synapse plasticity in rats. Neurotherapeutics 17(2):664–675. https://doi.org/10.1007/s13311-019-00807-3
Li Y, Wang ML, Zhang B, Fan XX, Tang Q, Yu X et al (2022) Antidepressant-like effect and mechanism of ginsenoside Rd on rodent models of depression. Drug Des Devel Ther 16:843. https://doi.org/10.2147/DDDT.S351421
Lis K (2008) Insulin-like growth factor 1 (IGF-1) and growth hormone (hGH) as the markers of osteoarthritis. Pol Orthop Traumatol 73(1):49–52
Liu W, Ge T, Leng Y, Pan Z, Fan J, Yang W, Cui R (2017) The role of neural plasticity in depression: from hippocampus to prefrontal cortex. J Neural Transplant Plast. https://doi.org/10.1155/2017/6871089
LLorens-Martin M, Torres-Aleman I, Trejo JL (2009) Reviews: mechanisms mediating brain plasticity: IGF1 and adult hippocampal neurogenesis. Neurosci 15(2):134–148. https://doi.org/10.1177/1073858408331371
Lugo-Marin J, Magan-Maganto M, Rivero-Santana A, Cuellar-Pompa L, Alviani M, Jenaro-Rio C, Diez E et al (2019) Prevalence of psychiatric disorders in adults with autism spectrum disorder: A SYSTEMATIC review and meta-analysisRes. Autism Spectr Disord 59:22–33. https://doi.org/10.1016/j.rasd.2018.12.004
McCutcheon RA, Marques TR, Howes OD (2020) Schizophrenia—an overview. JAMA Psychiat 77(2):201–210. https://doi.org/10.1001/jamapsychiatry.2019.3360
Miguel PM, Schuch CP, Rojas JJ, Carletti JV, Deckmann I, Martinato LH et al (2015) Neonatal hypoxia-ischemia induces attention-deficit hyperactivity disorder-like behavior in rats. Behav Neurosci 129(3):309. https://doi.org/10.1037/bne0000063
Miller DL, Papayannopoulos IA, Styles J, Bobin SA, Lin YY, Biemann K, Iqbal KH (1993) Peptide compositions of the cerebrovascular and senile plaque core amyloid deposits of Alzheimer’ s disease. Arch Biochem Biophys 301(1):41–52. https://doi.org/10.1006/abbi.1993.1112
Mitroshina EV, Savyuk MO, Ponimaskin E, Vedunova MV (2021) Hypoxia-inducible factor (HIF) in ischemic stroke and neurodegenerative disease. Front Cell Dev Biol. https://doi.org/10.3389/fcell.2021.703084
Modabbernia A, Velthorst E, Reichenberg A (2017) Environmental risk factors for autism: an evidence-based review of systematic reviews and meta-analyses. Mol Autism 8(1):1–16. https://doi.org/10.1186/s13229-017-0121-4
Mudgil M, Gupta N, Nagpal M, Pawar P (2012) Nanotechnology: a new approach for ocular drug delivery system. Int J Pharm Pharm Sci 4(2):105–112
Nagu P, Parashar A, Behl T, Mehta V (2021) CNS implications of COVID-19: a comprehensive review. Rev Neurosci 32(2):219–234. https://doi.org/10.1515/revneuro-2020-0070
Nanduri J, Vaddi DR, Khan SA, Wang N, Makarenko V, Semenza GL et al (2015) HIF-1α activation by intermittent hypoxia requires NADPH oxidase stimulation by xanthine oxidase. PLoS ONE 10(3):e0119762. https://doi.org/10.1371/journal.pone.0119762
Ng F, Berk M, Dean O, Bush AI (2008) Oxidative stress in psychiatric disorders: evidence base and therapeutic implications. Int J Neuropsychopharmacol 11(6):851–876. https://doi.org/10.1017/s1461145707008401
Nzou G, Wicks RT, VanOstrand NR, Mekky GA, Seale SA, El-Taibany A et al (2020) Multicellular 3D neurovascular unit model for assessing hypoxia and neuroinflammation induced blood-brain barrier dysfunction. Sci Rep 10(1):1–15. https://doi.org/10.1038/s41598-020-66487-8
Okazaki S, Boku S, Watanabe Y, Otsuka I, Horai T, Morikawa R et al (2022) Polymorphisms in the hypoxia inducible factor binding site of the macrophage migration inhibitory factor gene promoter in schizophrenia. PLoS ONE 17(3):e0265738. https://doi.org/10.1371/journal.pone.0265738
Oman J, Ubah D, Rivas D, Triyasakorn K, Bhushan A, Lai J, Awale P (2021) Valproic acid alters the expression of HIF-1α in embryonic brains in a valproic acid rodent model of autism. FASEB J. https://doi.org/10.1096/fasebj.2021.35.S1.03260
Onishi Y, Hiraiwa M, Kamada H, Iezaki T, Yamada T, Kaneda K, Hinoi E (2019) Hypoxia affects Slc7a5 expression through HIF-2α in differentiated neuronal cells. FEBS Open Bio 9(2):241–247
Prabhakar NK, Khan H, Grewal AK, Singh TG (2022) Intervention of neuroinflammation in the traumatic brain injury trajectory: in vivo and clinical approaches. Int Immunopharmacol 108:108902. https://doi.org/10.1016/j.intimp.2022.108902
Rani R, Kumar A, Jaggi AS, Singh N (2021) Pharmacological investigations on efficacy of Phlorizin asodium-glucose co-transporter (SGLT) inhibitor in mouse model ofintracerebroventricular streptozotocin induced dementia of AD type. J Basic Clin Physiol Pharmacol 32(6):1057–1064. https://doi.org/10.1515/jbcpp-2020-0330
Rihal V, Kaur A, Singh TG, Abdel-Daim MM (2022) Therapeutic and mechanistic intervention of vitamin D in neuropsychiatric disorders. Psychiat Res 14:114782. https://doi.org/10.1016/j.psychres.2022.114782
Roman FS, Truchet B, Marchetti E, Chaillan FA, Soumireu-Mourat B (1999) Correlations between electrophysiological observations of synaptic plasticity modifications and behavioral performance in mammals. Prog Neurobiol 58(1):61–87. https://doi.org/10.1016/S0301-0082(98)00076-8
Rosenstein JM, Krum JM, Ruhrberg C (2008) VEGF in the nervous system. VEGF Dev. https://doi.org/10.1007/978-0-387-78632-2_8
Russo VC, Gluckman PD, Feldman EL, Werther G (2005) The insulin-like growth factor system and its pleiotropic functions in brain. Endocr Rev 26(7):916–943. https://doi.org/10.1210/er.2004-0024
Sayal K, Prasad V, Daley D, Ford T, Coghill D (2018) ADHD in children and young people: prevalence, care pathways, and service provision. Lancet Psychiatry 5(2):75–186. https://doi.org/10.1016/s2215-0366(17)30167-0
Semenza GL (1998) Hypoxia-inducible factor 1: master regulator of O2 homeostasis. Curr Opin Genet Dev 8(5):588–594. https://doi.org/10.1016/s0959-437x(98)80016-6
Semenza GL (2007) Hypoxia-inducible factor 1 (HIF-1) pathway. Sci STKE. https://doi.org/10.1126/stke.4072007cm8
Sharma B, Sharma A (2012) Future prospect of nanotechnology in development of anti-ageing formulations. Int J Pharm Pharm Sci 4(3):57–66
Shi LZ, Wang R, Huang G, Vogel P, Neale G, Green DR, Chi H (2011) HIF1α–dependent glycolytic pathway orchestrates a metabolic checkpoint for the differentiation of TH17 and Treg cells. J Exp Med 208(7):1367–1376. https://doi.org/10.1084/jem.20110278
Shibata T, Yamagata H, Uchida S, Otsuki K, Hobara T, Higuchi F et al (2013) The alteration of hypoxia inducible factor-1 (HIF-1) and its target genes in mood disorder patients. Prog Neuropsychopharmacol Biol Psychiatry 43:222–229. https://doi.org/10.1016/j.pnpbp.2013.01.003
Shrivastav S, Sindhu R, Kumar S, Kumar P (2009) Anti-psoriatic and phytochemical evaluation of Thespesia populnea bark extracts. Int J Pharm Pharm Sci 1(1):76–85
Şimşek F, Işık Ü, Aktepe E, Kılıç F, Şirin FB, Bozkurt M (2021) Comparison of serum VEGF, IGF-1, and HIF-1α levels in children with autism spectrum disorder and healthy controls. CcJ Autism Dev Disord 51(10):3564–3574. https://doi.org/10.1007/s10803-020-04820-w
Singh V, Arora S, Kaur R, Shri R (2021) Memory improvement effects of Ganoderma mediosinense in mice via its anticholinergic and antioxidant activities. Alzheimer’s Dement 17:e050707. https://doi.org/10.1002/alz.050707
Siniscalco D, Schultz S, Brigida AL, Antonucci N (2018) Inflammation and neuro-immune dysregulations in autism spectrum disorders. Pharmaceuticals 11(2):56. https://doi.org/10.3390/ph11020056
Skogstrand K, Hagen CM, Borbye-Lorenzen N, Christiansen M, Bybjerg-Grauholm J, Bækvad-Hansen M (2019) Reduced neonatal brain-derived neurotrophic factor is associated with autism spectrum disorders. Transl Psychiatry 9(1):1–9. https://doi.org/10.1038/s41398-019-0587-2
Strowitzki MJ, Cummins EP, Taylor CT (2019) Protein hydroxylation by hypoxia-inducible factor (HIF) hydroxylases: unique or ubiquitous? Cells 8(5):384. https://doi.org/10.3390/cells8050384
Tărlungeanu DC, Deliu E, Dotter CP, Kara M, Janiesch PC, Scalise M (2016) Impaired amino acid transport at the blood brain barrier is a cause of autism spectrum disorder. Cell 167(6):1481–1494
Thakur AK, Chellappan DK, Dua K, Mehta M, Satija S, Singh I (2020) Patented therapeutic drug delivery strategies for targeting pulmonary diseases. Expert Opin Ther Pat 30(5):375–387. https://doi.org/10.1080/13543776.2020.1741547
Tiwari P, Khan H, Singh TG, Grewal AK (2022) Poly (ADP-ribose) polymerase: an overview of mechanistic approaches and therapeutic opportunities in the management of stroke. Neurochem Res 47:1830–1852. https://doi.org/10.1007/s11064-022-03595-z
Vargas DL, Nascimbene C, Krishnan C, Zimmerman AW, Pardo CA (2005) Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann Neurol 57(1):67–81
Vengellur A, Phillips JM, Hogenesch JB, LaPres JJ (2005) Gene expression profiling of hypoxia signaling in human hepatocellular carcinoma cells. Physiol Genomics 22(3):308–318. https://doi.org/10.1152/physiolgenomics.00045.2004
Vogelzangs N, Beekman A, De Jonge P, Penninx BJTp (2013) Anxiety disorders and inflammation in a large adult cohort. Transl Psychiatry 3(4):e249–e249. https://doi.org/10.1038/tp.2013.27
Wang XX, Zha YY, Yang B, Chen L, Wang M (2016) Suppression of synaptic plasticity by fullerenol in rat hippocampus in vitro. Int J Nanomed. https://doi.org/10.2147/IJN.S104856
Wright M, Di Ciano P, Brands B (2020) Use of cannabidiol for the treatment of anxiety: a short synthesis of pre-clinical and clinical evidence. Cannabis Cannabinoid Res 5(3):191–196. https://doi.org/10.1089/can.2019.0052
Xia Y, Choi HK, Lee K (2012) Recent advances in hypoxia-inducible factor (HIF)-1 inhibitors. Eur J Med Chem 49:24–40. https://doi.org/10.1016/j.ejmech.2012.01.033
Xiao Q, Qin X (2017) Expression of hypoxia-inducible factor-1 α in children with attention deficit hyperactivity disorder and its relationship with respiratory function. Chin J Prim Med Pharm 12:1390–1393
Xu LH, Xie H, Shi ZH, Du LD, Wing YK, Li AM, Ke Y et al (2015) Critical role of endoplasmic reticulum stress in chronic intermittent hypoxia-induced deficits in synaptic plasticity and long-term memory. Antioxid Redox Signal 23(9):695–710. https://doi.org/10.1089/ars.2014.6122
Ye Z, Kappelmann N, Moser S, Smith GD, Burgess S, Jones PB, Khandaker GM (2021) Role of inflammation in depression and anxiety: Tests for disorder specificity, linearity and potential causality of association in the UK Biobank. EClinicalMedicine 38:100992
Yuan G, Nanduri J, Khan S, Semenza GL, Prabhakar NR (2008) Induction of HIF-1α expression by intermittent hypoxia: involvement of NADPH oxidase, Ca2+ signaling, prolyl hydroxylases, and mTOR. J Cell Physiol 217(3):674–685. https://doi.org/10.1002/jcp.21537
Zhang Z, Yan J, Chang Y, ShiDu Yan S, Shi H (2011) Hypoxia inducible factor-1 as a target for neurodegenerative diseases. Curr Med Chem 18(28):4335–4343. https://doi.org/10.2174/092986711797200426
Zimna A, Kurpisz M (2015) Hypoxia-inducible factor-1 in physiological and pathophysiological angiogenesis: applications and therapies. Biomed Res Int. https://doi.org/10.1155/2015/549412
Acknowledgements
The authors are grateful to the Chitkara College of Pharmacy, Chitkara University, Rajpura, Patiala, Punjab, India for providing the necessary facilities to carry out the research work.
Funding
Nil.
Author information
Authors and Affiliations
Contributions
Conceptualization: conceived and designed the experiments: TGS. Analyzed the data: HK, TGS. Wrote the manuscript: DS, HK Editing of the Manuscript: AK, AKG. Critically reviewed the article: TGS and KD.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Ethical approval and consent of participation
Not applicable.
Consent for publication
All authors read and given their consent for the final manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, D., Khan, H., Kumar, A. et al. Pharmacological modulation of HIF-1 in the treatment of neuropsychiatric disorders. J Neural Transm 130, 1523–1535 (2023). https://doi.org/10.1007/s00702-023-02698-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-023-02698-3