Abstract
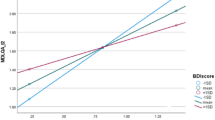
To determine the association of daily physical activity with cognition, mood disorders, and olfactory function in treatment-naive patients with early-stage Parkinson’s disease (PD). The study subjects were 52 treatment-naive patients with early-stage PD (< 80 years). Daily physical activity was measured using a wearable sensor with a built-in triaxial accelerometer, and its association with cognition [mini-mental state examination (MMSE), clock-drawing test (CDT), frontal assessment battery (FAB), and behavioral assessment of the dysexecutive syndrome (BADS)], depressive symptoms [Beck Depression Inventory-Second Edition (BDI-II)], apathy [Starkstein Apathy Scale (AS)], and olfactory function [Odor Stick Identification Test for the Japanese (OSIT-J)] was analyzed using multiple linear regression after adjustment for age, sex, and education status. The daily physical activity (0.42 ± 0.11 m/s2) of the PD group was significantly lower than that of healthy controls (p < 0.001). Moreover, the daily physical activity of the PD group was significantly associated with FAB (β = 0.337, p = 0.027) and BADS (β = 0.374, p = 0.017) scores, but not with MMSE, CDT, BDI-II, AS, and OSIT-J scores. The daily physical activity is significantly reduced in treatment-naive patients with early-stage PD, and the low activity correlates with frontal/executive function.

Similar content being viewed by others
References
Aarsland D, Brønnick K, Alves G, Tysnes OB, Pedersen KF, Ehrt U, Larsen JP (2009) The spectrum of neuropsychiatric symptoms in patients with early untreated Parkinson’s disease. J Neurol Neurosurg Psychiatry 80:928–930. https://doi.org/10.1136/jnnp.2008.166959
Amara AW, Chahine L, Seedorff N, Caspell-Garcia CJ, Coffey C, Simuni T, Initiative Parkinson’s Progression Markers (2019) Self-reported physical activity levels and clinical progression in early Parkinson’s disease. Parkinsonism Relat Disord 61:118–125. https://doi.org/10.1016/j.parkreldis.2018.11.006
Baba T, Takeda A, Kikuchi A, Nishio Y, Hosokai Y, Hirayama K, Hasegawa T, Sugeno N, Suzuki K, Mori E, Takahashi S, Fukuda H, Itoyama Y (2011) Association of olfactory dysfunction and brain. Metabolism in Parkinson’s disease. Mov Disord 26:621–628. https://doi.org/10.1002/mds.23602
Baba T, Kikuchi A, Hirayama K, Nishio Y, Hosokai Y, Kanno S, Hasegawa T, Sugeno N, Konno M, Suzuki K, Takahashi S, Fukuda H, Aoki M, Itoyama Y, Mori E, Takeda A (2012) Severe olfactory dysfunction is a prodromal symptom of dementia associated with Parkinson’s disease: a 3 year longitudinal study. Brain 135:161–169. https://doi.org/10.1093/brain/awr321
Barone P, Antonini A, Colosimo C, Marconi R, Morgante L, Avarello TP, Bottacchi E, Cannas A, Ceravolo G, Ceravolo R, Cicarelli G, Gaglio RM, Giglia RM, Iemolo F, Manfredi M, Meco G, Nicoletti A, Pederzoli M, Petrone A, Pisani A, Pontieri FE, Quatrale R, Ramat S, Scala R, Volpe G, Zappulla S, Bentivoglio AR, Stocchi F, Trianni G, Dotto PD, PRIAMO study group (2009) The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov Disord 24:1641–1649. https://doi.org/10.1002/mds.22643
Berendse HW, Roos DS, Raijmakers P, Doty RL (2011) Motor and non-motor correlates of olfactory dysfunction in Parkinson’s disease. J Neurol Sci 310:21–24. https://doi.org/10.1016/j.jns.2011.06.020
de la Riva P, Smith K, Xie SX, Weintraub D (2014) Course of psychiatric symptoms and global cognition in early Parkinson disease. Neurology 83:1096–1103. https://doi.org/10.1212/WNL.0000000000000801
Del Din S, Godfrey A, Mazzà C, Lord S, Rochester L (2016) Free-living monitoring of Parkinson’s disease: lessons from the field. Mov Disord 31:1293–1313. https://doi.org/10.1002/mds.26718
Dirnberger G, Jahanshahi M (2013) Executive dysfunction in Parkinson’s disease: a review. J Neuropsychol 7:193–224. https://doi.org/10.1111/jnp.12028
Dontje ML, de Greef MH, Speelman AD, van Nimwegen M, Krijnen WP, Stolk RP, Kamsma YP, Bloem BR, Munneke M, van der Schans CP (2013) Quantifying daily physical activity and determinants in sedentary patients with Parkinson’s disease. Parkinsonism Relat Disord 19:878–882. https://doi.org/10.1016/j.parkreldis.2013.05.014
Dubois B, Slachevsky A, Litvan I, Pillon B (2000) The FAB: a frontal assessment battery at bedside. Neurology 55:1621–1626. https://doi.org/10.1212/WNL.55.11.1621
Dujardin K, Langlois C, Plomhause L, Carette AS, Delliaux M, Duhamel A, Defebvre L (2014) Apathy in untreated early-stage Parkinson disease: relationship with other non-motor symptoms. Mov Disord 29:1796–1801. https://doi.org/10.1002/mds.26058
Duncan GW, Khoo TK, Yarnall AJ, O’Brien JT, Coleman SY, Brooks DJ, Barker RA, Burn DJ (2014) Health-related quality of life in early Parkinson’s disease: the impact of nonmotor symptoms. Mov Disord 29:195–202. https://doi.org/10.1002/mds.25664
Fullard ME, Tran B, Xie SX, Toledo JB, Scordia C, Linder C, Purri R, Weintraub D, Duda JE, Chahine LM, Morley JF (2016) Olfactory impairment predicts cognitive decline in early Parkinson’s disease. Parkinsonism Relat Disord 25:45–251. https://doi.org/10.1016/j.parkreldis.2016.02.013
Gibb WR, Lees AJ (1988) The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson’s disease. J Neurol Neurosurg Psychiatry 51:745–752. https://doi.org/10.1136/jnnp.51.6.745
Hatanaka N, Sato K, Hishikawa N, Takemoto M, Ohta Y, Yamashita T, Abe K (2016) Comparative gait analysis in progressive supranuclear palsy and Parkinson’s Disease. Eur Neurol 75:282–289. https://doi.org/10.1159/000445111
Hu MT, Szewczyk-Królikowski K, Tomlinson P, Nithi K, Rolinski M, Murray C, Talbot K, Ebmeier KP, Mackay CE, Ben-Shlomo Y (2014) Predictors of cognitive impairment in an early stage Parkinson’s disease cohort. Mov Disord 29:351–359. https://doi.org/10.1002/mds.25748
Iijima M, Kobayakawa T, Saito S, Osawa M, Tsutsumi Y, Hashimoto S, Iwata M (2008) Smell identification in Japanese Parkinson’s disease patients: using the odor stick identification test for Japanese subjects. Intern Med 47:1887–1892. https://doi.org/10.2169/internalmedicine.47.1345
Iijima M, Mitoma H, Uchiyama S, Kitagawa K (2017) Long-term monitoring gait analysis using a wearable device in daily lives of patients with Parkinson’s Disease: the efficacy of selegiline hydrochloride for gait disturbance. Front Neurol 8:542. https://doi.org/10.3389/fneur.2017.00542
Kamei S, Hara M, Serizawa K, Murakami M, Mizutani T, Ishiburo M, Kawahara R, Takagi Y, Ogawa K, Yoshihashi H, Shinbo S, Suzuki Y, Yamaguchi M, Morita A, Takeshita J, Hirayanagi K (2008) Executive dysfunction using behavioral assessment of the dysexecutive syndrome in Parkinson’s disease. Mov Disord 23:566–573. https://doi.org/10.1002/mds.21890
Khoo TK, Yarnall AJ, Duncan GW, Coleman S, O’Brien JT, Brooks DJ, Barker RA, Burn DJ (2013) The spectrum of nonmotor symptoms in early Parkinson disease. Neurology 80:276–281. https://doi.org/10.1212/WNL.0b013e31827deb74
Litvan I, Goldman JG, Tröster AI, Schmand BA, Weintraub D, Petersen RC, Mollenhauer B, Adler CH, Marder K, Williams-Gray CH, Aarsland D, Kulisevsky J, Rodriguez-Oroz MC, Burn DJ, Barker RA, Emre M (2012) Diagnostic criteria for mild cognitive impairment in Parkinson’s disease: movement Disorder Society Task Force guidelines. Mov Disord 27:349–356. https://doi.org/10.1002/mds.24893
Liu H, Ou R, Wei Q, Hou Y, Zhang L, Cao B, Zhao B, Song W, Shang H (2017) Apathy in drug-naïve patients with Parkinson’s disease. Parkinsonism Relat Disord 44:28–32. https://doi.org/10.1016/j.parkreldis.2017.08.008
Loprinzi PD, Danzl MM, Ulanowski E, Paydo C (2018) A pilot study evaluating the association between physical activity and cognition among individuals with Parkinson’s disease. Disabil Health J 11:165–168. https://doi.org/10.1016/j.dhjo.2017.05.004
Lord S, Galna B, Coleman S, Burn D, Rochester L (2013) Mild depressive symptoms are associated with gait impairment in early Parkinson’s disease. Mov Disord 28:634–639. https://doi.org/10.1002/mds.25338
Marinus J, Zhu K, Marras C, Aarsland D, van Hilten JJ (2018) Risk factors for non-motor symptoms in Parkinson’s disease. Lancet Neurol 17:559–568. https://doi.org/10.1016/S1474-4422(18)30127-3
Masala C, Solla P, Liscia A, Defazio G, Saba L, Cannas A, Cavazzana A, Hummel T, Haehner A (2018) Correlation among olfactory function, motors’ symptoms, cognitive impairment, apathy, and fatigue in patients with Parkinson’s disease. J Neurol 265:1764–1771. https://doi.org/10.1007/s00415-018-8913-9
Matsui H, Udaka F, Miyoshi T, Hara N, Tamura A, Oda M, Kubori T, Nishinaka K, Kameyama M (2006) Frontal assessment battery and brain perfusion image in Parkinson’s disease. J Geriatr Psychiatry Neurol 19:41–45. https://doi.org/10.1177/0891988705284714
Mitoma H, Yoneyama M, Orimo S (2010) 24-hour recording of parkinsonian gait using a portable gait rhythmogram. Intern Med 49:2401–2408. https://doi.org/10.2169/internalmedicine.49.3511
Okada K, Kobayashi S, Yamagata S, Takahashi K, Yamaguchi S (1997) Poststroke apathy and regional cerebral blood flow. Stroke 28:2437–2441. https://doi.org/10.1161/01.STR.28.12.2437
Pedersen KF, Alves G, Brønnick K, Aarsland D, Tysnes OB, Larsen JP (2010) Apathy in drug-naïve patients with incident Parkinson’s disease: the Norwegian ParkWest study. J Neurol 257:217–223. https://doi.org/10.1007/s00415-009-5297-x
Poewe W, Seppi K, Tanner CM, Halliday GM, Brundin P, Volkmann J, Schrag AE, Lang AE (2017) Parkinson disease. Nat Rev Dis Primers 3:17013. https://doi.org/10.1038/nrdp.2017.13
Rouleau I, Salmon DP, Butters N, Kennedy C, McGuire K (1992) Quantitative and qualitative analyses of clock drawings in Alzheimer’s and Huntington’s disease. Brain Cogn 18:70–87. https://doi.org/10.1016/0278-2626(92)90112-Y
Rovini E, Maremmani C, Cavallo F (2017) How wearable sensors can support Parkinson’s disease diagnosis and treatment: a systematic review. Front Neurosci. 11:555. https://doi.org/10.3389/fnins.2017.00555
Saito S, Ayabe-Kanamura S, Takashima Y, Gotow N, Naito N, Nozawa T, Mise M, Deguchi Y, Kobayakawa T (2006) Development of a smell identification test using a novel stick-type odor presentation kit. Chem Senses 31:379–391. https://doi.org/10.1093/chemse/bjj042
Schapira AHV, Chaudhuri KR, Jenner P (2017) Non-motor features of Parkinson disease. Nat Rev Neurosci 18:435–450. https://doi.org/10.1038/nrn.2017.62
Speelman AD, van de Warrenburg BP, van Nimwegen M, Petzinger GM, Munneke M, Bloem BR (2011) How might physical activity benefit patients with Parkinson disease? Nat Rev Neurol 7:528–534. https://doi.org/10.1038/nrneurol.2011.107
Starkstein SE, Mayberg HS, Preziosi TJ, Andrezejewski P, Leiguarda R, Robinson RG (1992) Reliability, validity, and clinical correlates of apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci 4:134–139. https://doi.org/10.1176/jnp.4.2.134
Terashi H, Utsumi H, Ishimura Y, Mitoma H (2013) Independent regulation of the cycle and acceleration in parkinsonian gait analyzed by a long-term daily monitoring system. Eur Neurol 69:134–141. https://doi.org/10.1159/000345266
Terashi H, Utsumi H, Ishimura Y, Aizawa H, Yoneyama M, Mitoma H (2015) Kinematic analysis of 24-hour recording of walking pattern in patients with vascular parkinsonism. Int J Neurosci 125:733–741. https://doi.org/10.3109/00207454.2014.967350
Terashi H, Mitoma H, Yoneyama M, Aizawa H (2017) Relationship between amount of daily movement measured by a triaxial accelerometer and motor symptoms in patients with Parkinson’s disease. Appl. Sci. 7:486. https://doi.org/10.3390/app7050486
Utsumi H, Terashi H, Ishimura Y, Takazawa T, Hayashi A, Mochizuki H, Okuma Y, Orimo S, Takahashi K, Yoneyama M, Mitoma H (2012) Quantitative assessment of gait bradykinesia in Parkinson’s disease using a portable gait rhythmogram. Acta Med Okayama 66:31–40. https://doi.org/10.18926/AMO/48079
van Nimwegen M, Speelman AD, Hofman-van Rossum EJ, Overeem S, Deeg DJ, Borm GF, van der Horst MH, Bloem BR, Munneke M (2011) Physical inactivity in Parkinson’s disease. J Neurol 258:2214–2221. https://doi.org/10.1007/s00415-011-6097-7
Visser M, Leentjens AF, Marinus J, Stiggelbout AM, van Hilten JJ (2006) Reliability and validity of the Beck depression inventory in patients with Parkinson’s disease. Mov Disord 21:668–672. https://doi.org/10.1002/mds.20792
Weintraub D, Simuni T, Caspell-Garcia C, Coffey C, Lasch S, Siderowf A, Aarsland D, Barone P, Burn D, Chahine LM, Eberling J, Espay AJ, Foster ED, Leverenz JB, Litvan I, Richard I, Troyer MD, Hawkins KA (2015) Parkinson’s progression markers initiative Cognitive performance and neuropsychiatric symptoms in early, untreated Parkinson’s disease. Mov Disord 30:919–927. https://doi.org/10.1002/mds.26170
Wilson BA, Alderman N, Burguess PW, Emslie H, Evans JJ (1996) Behavioural assessment of the dysexecutive syndrome. Thames Valley Test Company, Bury St. Edmunds
Yamamoto S, Mogi N, Umegaki H, Suzuki Y, Ando F, Shimokata H, Iguchi A (2004) The clock drawing test as a valid screening method for mild cognitive impairment. Dement Geriatr Cogn Disord 18:172–179. https://doi.org/10.1159/000079198
Yoneyama M, Kurihara Y, Watanabe K, Mitoma H (2013a) Accelerometry-based gait analysis and its application to Parkinson’s disease assessment-Part 2: a new measure for quantifying walking behavior. IEEE Trans Neural Syst Rehabil Eng 21:999–1005. https://doi.org/10.1109/TNSRE.2013.2268251
Yoneyama M, Mitoma H, Okuma Y (2013b) Accelerometry-based long-term monitoring of movement disorders: from diurnal gait behavior to nocturnal bed mobility. J Mech Med Biol 13:1350041. https://doi.org/10.1142/S0219519413500413
Yoneyama M, Kurihara Y, Watanabe K, Mitoma H (2014) Accelerometry-based gait analysis and its application to Parkinson’s disease assessment-Part 1: detection of stride event. IEEE Trans Neural Syst Rehabil Eng 22:613–622. https://doi.org/10.1109/TNSRE.2013.2260561
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Terashi, H., Taguchi, T., Ueta, Y. et al. Association of daily physical activity with cognition and mood disorders in treatment-naive patients with early-stage Parkinson’s disease. J Neural Transm 126, 1617–1624 (2019). https://doi.org/10.1007/s00702-019-02085-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-019-02085-x