Abstract
The morphological differences between orchid genera synonymized recently under the name Erycina, i.e., Psygmorchis, Stacyella and Erycina s.str. were evaluated. The results of the revision complemented with the available molecular data support the traditional recognition of three different genera. The morphological description of the taxa included in Erycina-complex and artificial keys to the subordinate taxa are provided. Two new species of Psygmorchis are described and illustrated based on Colombian material and their taxonomic affinities are briefly discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
A numerous theoretical (Maynard Smith 1962, 1966; Rice 1987) and empirical (Thoday and Gibson 1970; Howard and Harrison 1984; Hua and Wiens 2013) studies were incorporated into a composite model of speciation via habitat specialization. It was shown that the selection on a habitat preference always leads to the maintenance of an adaptive polymorphism and to the evolution of prezygotic reproductive isolation as a correlated character (Rice 1987; de Meeûs et al. 1993).
The habitat restrictions apply to the majority of orchid species and due to the cosmopolitan distribution of those plants, plenty of ecological groups may be distinguished within Orchidaceae. One of the most interesting of them, in aspects of evolution, ecology and taxonomy, consists of so-called “twig epiphytes”. The term refers to the plants growing on the smallest branches (<2.5 cm in diameter) of their phorophytes with weak attachment to them (Benzing 1990). This kind of habit is usually related to highest light levels and the greatest fluctuation in water availability (Gowland et al. 2011). The tolerance of twig epiphytes for the high humidity, that is common toward the trunk, is often low (Chase 1988; Zotz 2007). Plants are characterized by the short life cycle (often reaching maturity in one season), presence of hooks or projections on the seed testa (presumably for rapid water recruitment) and psygmoid (paedomorphic) habit (Neubig et al. 2012).
The majority of orchid twig epiphytes belong to Angraecinae, Aeridinae and Oncidiinae. As suggested by Dressler (1981), the adaptation to such specific habitat as the small branches has arisen several times within groups of genera. Among Neotropical oncidioid orchids the twig epiphytes were placed within different subtribes (Pabst and Dungs 1977) and as suggested by Chase the habitat variation and morphological adaptations to those different environment conditions do not reflect phylogenetic relationships (Chase and Palmer 1997). The same authors advocated that the radiation of orchid twig epiphytes started with vegetative and life history traits and later moved on to floral diversification in a “leapfrog” pattern. The term “leapfrog radiation” is used to describe a sequence of events starting with the invasion of an adaptive zone (twigs) and succeeded by a secondary radiation involving floral diversification (Stanley 1990; Sanderson 1998).
The recent molecular studies on oncidioid orchids resulted in synonymization of three twig-epiphytic genera under Erycina Lindl. (Williams et al. 2001). While this concept was accepted in the latest genetic research on Oncidiinae (Neubig et al. 2012), no comprehensive morphological evaluation of Erycina-complex has been done so far. The main goal of the research presented here was to evaluate the morphological basis for the generic delimitation of the three genera included in Erycina s.l., i.e., Erycina Lindl., Psygmorchis Dodson and Dressler and Stacyella Szlach.
The genus Erycina (Fig. 1a)
The Neotropical genus Erycina was described by John Lindley in 1853 based on Oncidium echinatum Kunth which the author recognized substantially distinct from other oncidioid orchids in its dwarf gynostemium, long sigmoid rostellum and prominent wing-like projections (“fleshy column arms”). Lindley noted similarity of O. echinatum to Zygostates Lindl. and Leochilus Knowles & Westc., but differing from the former in its conspicuously alate gynostemium, minute petals and the form of large lip. From the second genus it is easily distinguishable by its elongate, sigmoid rostellum as well as by the lip form.
The genus Psygmorchis (Fig. 1b)
In 1972 Dodson and Dressler, based on the distinctive, fan-shaped plant form, established genus Psygmorchis designating Epidendrum pusillum L. as the nominal species. The authors included in the new taxon species lacking pseudobulbs, producing ensiform leaves and characterized by the free perianth segments. Plants assigned to Psygmorchis were previously recognized by Kraenzlin (1920) as representatives of Oncidium sect. Aphanobulbia subsect. Iridifolia. In subsequent research, Dodson (1957) elevated this group to the sectional rank, but later Garay (1963, 1970) suggested that section Iridifolia of Oncidium should be united with Lockhartia. While the latter genus produces laterally flattened leaves like Psygmorchis, both differ in crucial characters. The inflorescence in Lockhartia representatives is often terminal (vs lateral in Psygmorchis), the tabula infrastigmatica is absent (vs present) and the tegula is bifid (vs entire).
The genus Stacyella (Fig. 1c)
During the studies on the Ecuadorian orchids, Dressler and Williams (2003) found one more species of Reichenbach’s Equitantia group of Oncidium which generic affinity should be reconsidered and they transferred O. crista-galli to Psygmorchis. This reassignment was later evaluated by Szlachetko (2006) who found this species substantially different from other Psygmorchis species in both vegetative (presence of unifoliate pseudobulbs) and generative (oblong clavate, distinctly cleft pollinia, convex tegula) characters.
Materials and methods
In total, over 70 dried herbarium specimens and liquid preserved plants deposited or borrowed from AMES, COL, CUVC, FMB, HUA, K, P, PSO, VALLE and W were examined according to the standard procedures. Every studied specimen was photographed and the data from the label were taken. The presence and form of the pseudobulbs as well as the leaves shape and arrangement were studied first. The details of the inflorescence, e.g., the form of the floral bracts and ovaries were observed under a stereoscopic microscope. The perianth parts were studied after softened flowers in the boiling water.
Taxonomic treatment
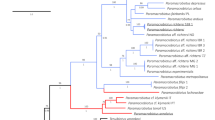
In the concept of Erycina proposed by Williams et al. (2001), the authors included Psygmorchis Dodson and Dressler and monotypic Stacyella Szlach. Surprisingly, despite the clear statement that all these three genera could be maintained, Neubig et al. (2012) decided to accept the broad concept of the genus because of their similar floral morphology and modified habit. However, the phylogenetic trees presented by Williams et al. (2001) and Neubig et al. (2012, Fig. 2) show that all three discussed genera are accommodated in the separated clades. The pair E. diaphana-E. echinata is sister to “E. pusilla”-“E. pumilio“ and those together are sister to “E. crista-galli”. Considering this tree structure and the significant differences in the vegetative and floral morphological characters between Erycina, Psygmorchis and Stacyella, we postulate to maintain them as separate genera.
Fragment of a single maximum likelihood tree presented by Neubig et al. (2012)
Key to the genera of Erycina-complex
1. | Pseudobulbs absent, leaves arranged in a fan | Psygmorchis |
1* | Pseudobulbs present, 1–2 foliate at the apex | 2 |
2. | Pseudobulbs unifoliate at the apex or leaf aborted, subtended by several foliaceous bracts, leaf without articulation | Stacyella |
2* | Pseudobulbs 1–2 foliate at the apex, subtended by several papyraceous sheaths, articulate at the base | Erycina |
Psygmorchis Dodson and Dressler
Phytologia 24(4): 288. 1972; Generitype: Psygmorchis pusilla (L.) Dodson and Dressler [≡ Epidendrum pusillum L.].
Pseudobulbs absent.Leaves numerous, distichous, imbricating, equitant, arranged in a fan. Inflorescence lateral, arching, 1–4 flowered. Flowers often proportionately large, yellow, showy, widely opened. Dorsal sepal and petals subsimilar, free, spreading; lateral sepals somewhat larger and basally more or less connate. Lip prominently larger than tepals, three lobed with prominent callus at the base, sometimes hairy; lateral lobes much smaller, spatulate to broadly obovate; middle lobe bilobulate. Gynostemium short, straight, rather delicate. Column part as long as anther or slightly longer, broadly winged near the stigma, wings flabellate and entire or irregularly dissected on margins. Column foot absent. Anther subventral, incumbent, operculate, oblong obtriangular, thin-walled, obscurely two chambered. Connective narrow, thin, apically elongate. Pollinia 2, clavate obovoid, hard, obscurely or shallowly cleft at the apex. Caudiculae sticky, amorphous. Apical clinandrium narrow. Stigma small, oblong elliptic, deeply concave, partially hidden by rostellum. Rostellum elongate, conical-cylindrical in the middle, blunt. Viscidium single, very small, thin. Tegula single, oblong to oblong deltoid, thin, lamellate, flat. Rostellum remnant bilobulate at the apex (Fig. 3).
Gynostemium of Psygmorchis glossomystax (Rchb. f.) Dodson and Dressler. a Gynostemium, bottom view. b Gynostemium, side view. c Gynostemium, front view. d Anther. e Pollinia, various views. f Tegula and viscidium, various views (Schulz 7285, U; Szlachetko & Mytnik-Ejsmont 2009)
Till present, the genus comprised five species distributed from Bolivia and Brazil to Mexico. During examination of the herbarium materials deposited in the Herbarium of National University in Bogotá (COL), we found specimens which, in our opinion, deserve to be described as new.
Key to the species
1. | Lip lateral lobes large, overlapping the middle lobe lobules | P. zamorensis |
1* | Lip lateral lobes small, not overlapping the middle lobe lobules | 2 |
2. | Callus margins entire | 3 |
2* | Callus margins crenate, digitate or fimbriate | 4 |
3. | Lateral sepals very shortly connate | P. pusilla |
3* | Lateral sepals connate to the middle | P. cuatrecasasi |
4. | Lip longer than broad, lateral lobes quadrate | P. pumilio |
4* | Lip as long or broader than long, lateral lobes suborbicular | 5 |
5. | Flowers entirely yellow, lip broader than long | P. gnomus |
5* | Flowers spotted brown, lip equally long and wide | 6 |
6. | Isthmus between the lobules prominent | P. glossomystax |
6* | Isthmus between the lobules very short | P. arevaloi |
Psygmorchis cuatrecasasi Kolan. & Szlach., sp. nov (Fig. 4)
Similar to P. pusilla (L.) Dodson and Dressler distinguished by the lateral sepals connate to the middle and the complicated lip callus.
Type: J
Idrobo 8924-Colombia, Amazonas, (COL!, holotype)
Leaves 7–8, up to 2.5 cm long, 0.5 cm wide, equitant, fleshy, oblanceolate-linear, subfalcate, subobtuse. Inflorescences 1.7 cm long, four flowered. Flowers yellow. Ovary and pedicel about 5 mm long. Floral bract 4 mm long. Dorsal sepal about 5 mm long, 2.8 mm wide, elliptic-ovate, acuminate, obtuse. Petals about 6 mm long, 3.5 mm wide, elliptic, acute. Lateral sepals 4 mm long, about 1 mm wide, linear-lanceolate, united for about half their length, free apices obtuse. Lip 12 mm long, 12 mm wide, three lobed; basal lobes 3.2 mm long, orbicular-rectangular, dolabriform; isthmus 2.5 mm long; middle lobe 9 mm wide, deeply divided at the apical third (2.8 mm), hence quadrilobulate, lobules suborbicular; callus a fleshy subpandurate pad, externally with two subtriangular, obtuse appendages. Gynostemium with a pair of conspicuous, dentate wings near the stigma.
Etymology
Dedicated to J. Cuatrecasas (1903–1996), an eminent collector of Colombian plants.
Distribution and ecology
So far this species is known from the Amazonian region of Colombia where it was found growing epiphytically at about 200 m a.s.l. It was found flowering in August and November (Fig. 5).
Representative specimens
J. Idrobo 8924-Colombia, Amazonas, Quebrada Aduche, alt. 200 m (5 Aug 1977), (COL!). Cuatrecasas 7384-Colombia., Vaupés, Selva entre Cano rande y San Jose del Guaviare, alt. 240 m (2 Nov 1939), (COL!).
Taxonomic notes
This species resembles P. pusilla (L.) Dodson and Dressler, from which it may be easily distinguished by the complicated callus (vs callus simple in P. pusilla), and lateral sepals connate to about the middle (vs. lateral sepals connate only near the base).
Psygmorchis arevaloi Kolan. & Szlach., sp. nov (Fig. 6)
Similar to P. glossomystax (Rchb.f.) Dodson and Dressler, but with the lip callus short, not fimbriate, the inconspicous isthmus between the lip lobules, the large, dolabriform lip basal lobes and the suborbicular lobules of the middle lobe.
Type: R
Arévalo & A. Tapasco 579-Colombia, Risaralda. (COL! holotype)
Leaves 10–12, 25 mm long, 3.5 mm wide, equitant, fleshy. Inflorescence 2.5 cm long, 2–3 flowered. Ovary and pedicel about 6 mm long. Floral bract 2 mm long. Dorsal sepal about 3.5 mm long, 2 mm wide, elliptic-ovate, acuminate, acute. Petals about 3.5 mm long, 2 mm wide, obliquely elliptic, obtuse. Lateral sepals 3.5 mm long, about 0.7 mm wide, linear-lanceolate, united at the base for 0.5 mm, apices obtuse. Lip 10 mm long, 9 mm wide, three lobed; basal lobes 4 mm long, orbicular-subrectangular, dolabriform; isthmus inconspicuous, about 0.5 mm long; middle lobe 5.5 mm long, 9 mm wide, divided at the apical fourth (1.4 mm), hence quadrilobulate, lobules suborbicular; callus a fleshy, 4-lobulate pad, dentate at the apices. Gynostemium with a pair of conspicuous, entire wings near the stigma.
Etymology
Dedicated to the co-collector of the type specimens
Distribution and ecology
So far the localities of this species are known exclusively from Colombia, however as it was found also near the Ecuadorian border, it is expected to be found also in this country. The populations were found growing epiphytically in rainforest as well as in the humid premontane and montane forest at the altitudes between 50 and 1,900 m. It was found flowering in January, April and October (Fig. 5).
Representative specimens
E.P.A. s.n.-Colombia, Caqueta. Florencia, alt. 420 m. (Dec 1930) (COL!); S. Diaz 3864-Colombia, Chocó, Mpio. San Jose del Palmar, Vereda La Badea, alt. 900 m (24 Jan 1983), (COL!); E. Forero, R. Jaramillo & J. McElroy 1383-Colombia, Chocó, Rio Serrano, afluente del Rio Atrato. 4–6 kms arriba de Guayabal, alt. 50 m (30 Apr 1973), (COL!); H. Garcia Barriga, Y. Hashimoto & M. Ishikawa 18699a-Colombia, Putumayo, Rio Putumayo, entre Puerto Asis y Puerto Leguizamo, fronteria Colombo-Ecuatoriana, alt. 400 m (14–15 Oct 1965), (COL!); R. Arevalo & A. Tapasco 579-Colombia, Risaralda, Mpio. Pueblo Rico, Vereda Monte Bello, PNN Tatama. Trocha sobre la casa de la finca de A. Tapasco, 5°12′46″N 76°05′08″W, alt. 1,600–1,900 m (28 Apr 2006), (COL!).
Notes
This species resembles P. glossomystax (Rchb.f.) Dodson and Dressler, from which is easily distinguished by the inconspicuously dentate lip callus (vs callus fimbriate in P. glossomystax), the isthmus between the lobules inconspicuous (vs isthmus long) and the orbicular-subquadrate, prominent lip basal lobes (vs basal lobes auriculate, small).
Stacyella szlach
Polish Bot. J. 51(1): 41. 2006; Generitype: Stacyella crista-galli (Rchb. f.) Szlach. [≡ Oncidium crista-galli Rchb. f.]
Pseudobulbs prominent, compressed, subtended by several foliaceous bracts, unifoliate at the apex or leaf aborted. Leaf thin, without articulation. Inflorescence lateral 1–4 flowered raceme, successively flowering. Sepals dissimilar, lateral sepals shortly connate. Petals similar to dorsal sepal. Lip deeply three lobed. Gynostemium short, straight. Column part as long as anther, broadly winged near the stigma, wings obliquely triangular, more or less dissected on margins. Column foot absent. Anther subventral, incumbent, operculate, oblong obtriangular, thin-walled, obscurely two chambered. Connective narrow, thin, apically elongate to form a rostrate projection. Pollinia 2, oblong clavate, hard, distinctly and unequally cleft at the apex. Caudiculae sticky, amorphous. Apical clinandrium narrow. Stigma rather large, oblong elliptic, deeply concave. Rostellum elongate, rostrate in the middle, blunt. Viscidium single, very small, elliptic, thin. Tegula single, oblong with oblong deltoid apical part, thin, lamellate, convex. Rostellum remnant bilobulate at the apex, canaliculate on the dorsal surface (Fig. 7).
Gynostemium of Stacyella crista-galli (Rchb. f.) Szlach. a. Gynostemium, bottom view. b. Gynostemium, side view. c. Rostellum remnant. d. Anther. E. Pollinarium, various views (Chase 84025, UGDA-DLSz; Szlachetko and Mytnik-Ejsmont 2009)
This is a monotypic genus known from Mesoamerica and northern South America.
Erycina Lindl.
Fol. Orchid. Erycina 2. 1853; Generitype: Erycina echinata (Kunth) Lindl. [≡ Oncidium echinatum Kunth]
Pseudobulbs aggregated, laterally compressed, 1–2 foliate, subtended by several papyraceous sheaths. Leaves thin, articulate at the base. Inflorescences 1 to several, from the base of pseudobulb, racemose, paniculate, with several to over 20 flowers per raceme, flowering simultaneously. Sepals dissimilar, lateral sepals connate at the base, petals similar to dorsal sepal. Lip three lobed. Gynostemium swollen just above the base, glabrous. Column part half as long as anther, with two wing-like projections on both sides of rostellum (E. echinata), or with large, thick projection pendent below stigma (E. diaphana). Column foot absent. Anther subapical, incumbent, operculate, dorsiventrally compressed, oblong, obscurely two chambered. Connective narrow, thin, papillate, forming prominent apical, roof-like projection, shortly apiculate at the apex. Pollinia 2, slightly almost elipsoid, shallowly cleft at the apex, hard. Caudiculae sticky, amorphous. Apical clinandrium obscure. Stigma small, elliptic, hidden by rostellum, deeply concave. Rostellum large, gently sigmoid, thick, narrowly cylindrical-conical, rounded at the apex. Viscidium very small, single, elliptic, thin. Tegula single, linear, slightly expanded at the apex, thin, lamellate. Rostellum remnant shallowly bilobed at the apex, canaliculate on the dorsal surface (Fig. 8).
Gynostemium of Erycina diaphana (Rchb. f.) Schltr. a. Gynostemium, side view. b. Gynostemium, front view. c. Rostellum remnant, apex. d. Anther, back view. e. Pollinia, various views. f and g. Tegula and viscidium, various views (Simmons 784, K; Szlachetko and Mytnik-Ejsmont 2009)
The genus contains two Mexican species.
Key to the species
1. | Pseudobulbs leafless, at anthesis enclothed basally by leafy sheaths, lip lateral lobes as large as the middle lobe | E. echinata |
1* | Pseudobulbs with apical leaf, without leaf or leafy sheaths at anthesis, lip lateral lobes smaller than the middle lobe | E. diaphana |
Conclusions
Phylogenetic relationships among major orchid clades have long been controversial, with no general consensus. The incoming molecular data are often incompatible with the results of the morphological and anatomical studies and the superiority of the molecular data is often implied, especially in the case of the twig epiphytes which has undergone an intensive adaptive radiation. The importance of the morphological data was, however, recalled by numerous authors (e.g., Wiens 2004; Hill 2005). The synergistic approach of combining morphological and molecular outcomes seems to be the most appropriate in resolving the actual relations between the taxa.
We believe that in the case of the studied taxa, the synonymization of the three genera was done without this comprehensive resolution. Moreover, it seems that the presence of the separated clades within Erycina-complex was ignored when both Psygmorchis and Stacyella were lumped in Erycina. The traditional recognition of the three genera is, in our opinion, supported by both morphological and genetic differences between them.
References
Benzing DH (1990) Vascular epiphytes: general biology and related biota. Cambridge University Press, Cambridge
Chase MW (1988) Obligate twig epiphytes: a distinct subset of Neotropical orchidaceous epiphytes. Selbyana 10:24–30
Chase MW, Palmer JD (1997) Leapfrog radiation in floral and vegetative traits among twig epiphytes in the orchid subtribe Oncidiinae. In: Givnish TJ, Sytsma KJ (eds) Molecular evolution and adaptive radiation. Cambridge University Press, Cambridge, pp 331–352
de Meeûs T, Michalakis Y, Renaud F, Olivieri I (1993) Polymorphism in heterogeneous environments, evolution of habitat selection and sympatric speciation: soft and hard selection models. Evol Ecol 7:175–198
Dodson CH (1957) Oncidium pusillum and its Allies. Amer Orchid Soc Bull 26:170–172
Dressler RL (1981) The orchids: natural history and classification. Harvard University Press, Cambridge
Dressler RL, Williams NH (2003) Psygmorchis crista-galli. In: Dodson CH, Esobar Restrepo R. (eds) Native Ecuadorian Orchids 4. Colina Livraria Editora Ltda, Rio de Janeiro, p 883
Garay LA (1963) Oliveriana and its position in the Oncidieae. Amer Orchid Soc Bull 32:18–24
Garay LA (1970) A reappraisal of the genus Oncidium Sw. Taxon 19:443–467
Gowland KM, Wood J, Clements MA, Nicotra AB (2011) Significant phorophyte (substrate) bias is not explained by fitness benefits in three epiphytic orchid species. Am J Bot 98:197–206
Hill RV (2005) Integration of morphological data sets for phylogenetic analysis of Amniota: the importance of integumentary characters and increased taxonomic sampling. Syst Biol 54:530–547
Howard DJ, Harrison RG (1984) Habitat segregation in ground crickets: the role of interspecific competition and habitat selection. Ecology 65:69–76
Hua X, Wiens JJ (2013) How does climate influence speciation? Amer Nat. doi:10.1086/670690
Kraenzlin F (1920) Orchidaceae-Monandrae, Tribus Oncidiinae-Odontoglosseae, pars II. Das Pflanzenreich IV. 50:1–344
Maynard Smith J (1962) Disruptive selection, polymorphism, and sympatric speciation. Nature 195:60–62
Maynard Smith J (1966) Sympatric speciation. Amer Natur 100:637–650
Neubig KM, Whitten WM, Williams NH, Blanco MA, Endara L, Burleigh JG, Silvera K, Cushman JC, Chase MW (2012) Generic recircumscriptions of Oncidiinae (Orchidaceae: Cymbidieae) based on maximum likelihood analysis of combined DNA datasets. Bot J Linean Soc 168:117–146
Pabst GFJ, Dungs F (1977) Orchidaceae Brasilienses V. 2. Brücke-Verlag Kurt Schmersow, Hildesheim
Rice WR (1987) Speciation via habitat specialization: the evolution of reproductive isolation as a correlated character. Evol Ecol 1:301–314
Sanderson MJ (1998) Reappraising adaptive radiation. Am J Bot 85(11):1650–1655
Stanley SM (1990) Adaptive radiation and macroevolution. In: Taylor PD, Larwood GP (eds) Major evolutionary radiations. Clarendon Press, Oxford, pp 1–15
Szlachetko DL (2006) Genera et species Orchidalium 11. Oncidieae. Polish Bot J 51:39–41
Szlachetko DL, Mytnik-Ejsmont J (2009) Gynostemia Orchidalium. vol. 4. Orchidaceae-Vandoideae. Acta Bot Fenn 180:1–313
Thoday JM, Gibson JB (1970) The probability of isolation by disruptive selection. Amer Natur 104:219–230
Wiens JJ (2004) The role of morphological data in phylogeny reconstruction. Syst Biol 53:653–661
Williams NH, Chase MW, Fulcher T, Whitten WM (2001) Molecular systematics of the Oncidiinae based on evidence from four DNA sequence regions: expanded circumscriptions of Cyrtochilum, Erycina, Otoglossum, and Trichocentrum and a new genus (Orchidaceae). Lindleyana 16:113–139
Zotz G (2007) Johansson revisited: the spatial structure of epiphyte assemblages. J Veg Sci 18:123
Acknowledgments
The curators and staff of the cited herbaria are thanked for their kind hospitality and assistance during visits and for making specimens available on loan. We are grateful and to J. Valdez Partida and J. M. Conejo Chaverri for providing photos. The research described here has been supported by the Polish Ministry of Science and Higher Education (research grant no. 3930/B/PO1/2007/33).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Kolanowska, M., Szlachetko, D.L. Notes on Erycina-complex with descriptions of new Colombian species. Plant Syst Evol 300, 527–534 (2014). https://doi.org/10.1007/s00606-013-0901-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-013-0901-z