Abstract
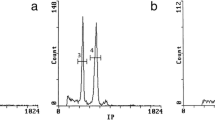
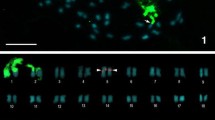
Karyotype and nuclear 2C-value data are considered important in taxonomic and evolutionary approaches in Coffea. Still, new methods are needed to further support such studies, especially to determine the progenitors of Coffea arabica. In this work, new cytogenetic and flow cytometry data were used to compare Coffea arabica, Coffea canephora and Coffea congensis. These data corroborate the hypothesis that C. canephora and C. congensis originated from a single ancestor, whose basic chromosome number was x = 11. In agreement with the observations of other authors, the karyotype and mean 2C-values confirm that C. arabica is a true allotetraploid originating from two diploid Coffea species with similar genomes. Although C. canephora and C. congensis have been considered potential progenitors of C. arabica, karyotype comparison revealed that only one of these species may be parental to C. arabica. These accurate cytogenetic and flow cytometry data contribute to expand our knowledge of the Coffea genome, as well as of possible progenitors of C. arabica.


Similar content being viewed by others
References
Barow M, Meister A (2002) Lack of correlation between AT frequency and genome size in higher plants and the effect of nonrandomness of base sequences on dye binding. Cytometry 47:1–7. doi:10.1002/cyto.10030
Bennett MD, Price HJ, Johnston S (2008) Anthocyanin inhibits propidium iodide DNA fluorescence in Euphorbia pulcherrima: implications for genome size variation and flow cytometry. Ann Bot 101:777–790. doi:10.1093/aob/mcm303
Berthouly M, Michaux-Ferriere NM (1996) High frequency somatic embryogenesis in Coffea canephora. Plant Cell Tissue Organ Cult 44:169–176. doi:10.1007/bf00048196
Bouharmont J (1963) Somatic chromosomes of Coffea species. Euphytica 12:254–257. doi:10.1007/BF00027458
Campa C, Doulbeau S, Dussert S, Hamon S, Noirot M (2005a) Diversity in bean caffeine content among wild Coffea species: evidence of a discontinuous distribution. Food Chem 91:633–637. doi:10.1016/j.foodchem.2004.06.032
Campa C, Doulbeau S, Dussert S, Hamon S, Noirot M (2005b) Qualitative relationship between caffeine and chlorogenic acid contents among wild Coffea species. Food Chem 93:135–139. doi:10.1016/j.foodchem.2004.10.015
Carvalho CR, Clarindo WR, Almeida PM (2007) Plant cytogenetics: still looking for the perfect mitotic chromosomes. Nucleus 50:453–462
Charrier A, Berthaud J (1985) Botanical classification of coffee. In: Clifford MN, Willson KC (eds) Coffee: botany, biochemistry and production of beans and beverage. Croom Helm, London, pp 13–47
Clarindo WR, Carvalho CR (2006) A high quality chromosome preparation from cell suspension aggregates culture of Coffea canephora. Cytologia 71:243–249. doi:10.1508/cytologia.71.243
Clarindo WR, Carvalho CR (2008) First Coffea arabica karyogram showing that this species is a true allotetraploid. Plant Syst Evol 247:237–241. doi:10.1007/s00606-008-0050-y
Clarindo WR, Carvalho CR (2009) Comparison of the Coffea canephora and Coffea arabica karyotype based on chromosomal DNA content. Plant Cell Rep 28:73–81. doi:10.1007/s00299-008-0621-y
Clarindo WR, Carvalho CR (2011) Flow cytometric analysis using SYBR Green I for genome size estimation in coffee. Acta Histochem 113(2):221–225. doi:10.1016/j.acthis.2009.10.005
Cros J, Gavalda MC, Chabrillange N, Recalt C, Duperray C, Hamon S (1994) Variations in the total nuclear DNA content in African Coffea species (Rubiaceae). Café Cacao The 38:3–10
Cros J, Combes MC, Chabrillange N, Duperray C, Angles AM, Hamon S (1995) Nuclear DNA content in the subgenus Coffea (Rubiaceae): inter- and intra-specific variation in African species. Can J Bot 73:14–20
Cruz CD (1997) Programa GENES- Aplicativo Computacional em Genética e Estatística. Editora UFV, Viçosa
Doležel J, Bartoš J (2005) Plant DNA flow cytometry and estimation of nuclear genome size. Ann Bot 95:99–110. doi:10.1093/aob/mci005
Doležel J, Göhde W (1995) Sex determination in dioecious plants Melandrium album and M. rubrum using high-resolution flow cytometry. Cytometry 19:103–106. doi:10.1002/cyto.990190203
Doležel J, Greilhuber J (2010) Nuclear genome size: are we getting closer? Cytometry 77A:635–642. doi:10.1002/cyto.a.20915
Doležel J, Sgorbati S, Lucretti S (1992) Comparation of three DNA fluorochromes for flow cytometric estimation of nuclear DNA content in plants. Physiol Plant 85:625–631. doi:10.1111/j.1399-3054.1992.tb04764.x
Doležel J, Greilhuber J, Lucretti S, Meister A, Lysák MA, Nardi L, Obermayer R (1998) Plant genome size estimation by flow cytometry: inter-laboratory comparation. Ann Bot 82:17–26. doi:10.1006/anbo.1998.0730
Doležel J, Bartoš J, Voglmayr H, Greilhuber J (2003) Nuclear DNA and genome size of trout and human. Cytometry 51:127–128. doi:10.1002/cyto.a.10013
Ducos JP, Alenton R, Reano JF, Kanchanomai C, Deshayes A, Pétiard V (2003) Agronomic performance of Coffea canephora P. trees derived from large-scale somatic embryo production in liquid medium. Euphytica 131:215–223. doi:10.1023/A:1023915613158
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228. doi:10.1023/A:1024033216561
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220:1049–1051. doi:10.1126/science.220.4601.1049
Godelle B, Cartier D, Marie D, Brown CS, Siljak-Yakovlev S (1993) Heterochromatin study demonstrating the non-linearity of fluorometry useful for calculations genomic base composition. Cytometry 14:618–626. doi:10.1002/cyto.990140606
Greilhuber J (1998) Intraspecific variation in genome size: a critical reassessment. Ann Bot 82:27–35
Greilhuber J (2005) Intraspecific variation in genome size in angiosperms: identifying its existence. Ann Bot 95:91–98. doi:10.1093/aob/mci004
Hamon P, Siljak-Yakovlev S, Srisuwan S, Robin O, Poncet V, Hamon S, de Kochko A (2009) Physical mapping of rDNA and heterochromatin in chromosomes of 16 Coffea species: a revised view of species differentiation. Chromosome Res 17(3):291–304. doi:10.1007/s10577-009-9033-2
Jain SM, de Klerk GJ (1998) Somaclonal variation in breeding and propagation of ornamental crops. Plant Tissue Cult Biotechnol 4:63–75
Jensen RH (1977) Chromomycin A3, as a fluorescent probe for flow cytometry of human gynecologic samples. J Histochem Cytochem 25:573–579. doi:10.1177/25.7.70448
Krug CA (1934) Variações somáticas em Coffea arabica L. Boletim Técnico Instituto Agronômico de Campinas 12:1–11
Ky CL, Louarn S, Dussert S, Guyot B, Hamon S, Noirot M (2001) Caffeine, trigonelline, chlorogenic acids and sucrose diversity in wild Coffea arabica L. and Coffea canephora P. accessions. Food Chem 75:223–230. doi:10.1016/S0308-8146(01)00204-7
Lashermes P, Combes MC, Trouslot P, Charrier A (1997) Phylogenetic relationships of coffee-tree species (Coffea L.) as inferred from ITS sequences of nuclear ribosomal DNA. Theor Appl Genet 94:947–955. doi:10.1007/s001220050500
Lashermes P, Combes MC, Robert J, Trouslot P, D’Hont A, Anthony F, Charrier A (1999) Molecular characterization and origin of the Coffea arabica L. genome. Mol Gen Genet 261:259–266. doi:10.1007/s004380050965
Loureiro J, Rodriguez E, Doležel J, Santos C (2006a) Comparison of four nuclear isolation buffers for plant DNA flow cytometry. Ann Bot 98:679–689. doi:10.1093/aob/mcl141
Loureiro J, Rodriguez E, Doležel J, Santos C (2006b) Flow cytometric and microscopic analysis of the effect of tannic acid on plant nuclei and estimation of DNA content. Ann Bot 98:515–527. doi:10.1093/aob/mcl140
Mahé L, Combes M, Lashermes P (2007) Comparison between a coffee single copy chromosomal region and Arabidopsis duplicated counterparts evidenced high level synteny between the coffee genome and the ancestral Arabidopsis genome. Plant Mol Biol 64:699–711. doi:10.1007/s11103-007-9191-6
Meister A (2005) Calculation of binding length of base-specific DNA dyes by comparison of sequence and flow cytometric data. Application to Oryza sativa and Arabidopsis thaliana. J Theor Biol 232:93–97. doi:10.1016/j.jtbi.2004.07.022
Meister A, Barow M (2007) DNA base composition of plant genomes. In: Doležel J, Greilhuber J, Suda J (eds) Flow cytometry with plant cells. Wiley-VCH, Weinheim, pp 177–215
Mendes AJT (1938a) Morfologia dos cromossomos de Coffea excelsa Chev. Boletim Técnico Instituto Agronômico de Campinas 56:1–8
Mendes AJT (1938b) Os cromossomos das Rubiáceas. Boletim Técnico Instituto Agronômico de Campinas 56:1–11
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Noirot M, Barre P, Louarn J, Duperray C, Hamon S (2000) Nucleus-cytosol interactions–a source of stoichiometric error in flow cytometric estimation of nuclear DNA content in plants. Ann Bot 86:309–316. doi:10.1006/anbo.2000.1187
Noirot M, Barre P, Louarn J, Deperray C, Hamoin S (2002) Consequences of stoichiometric error on nuclear DNA content evaluation in Coffea liberica var. dewevrei using DAPI and propidium iodide. Ann Bot 89:385–389. doi:10.1093/aob/mcf056
Noirot M, Barre P, Duperray C, Louarn J, Hamon S (2003a) Effects of caffeine and chlorogenic acid on propidium iodide accessibility to DNA: consequences on genome size evaluation in coffee tree. Ann Bot 92:259–264. doi:10.1093/aob/mcg139
Noirot M, Poncet V, Barre P, Hamon P, Hamon S, Kochko A (2003b) Genome size variations in diploid African Coffea species. Ann Bot 92:709–714. doi:10.1093/aob/mcg183
Noirot M, Barre P, Christiphe D, Hamon S, Kochko A (2005) Investigation on the causes of stoichiometric error in genome size estimation using heat experiments: consequences on data interpretation. Ann Bot 95:111–118. doi:10.1093/aob/mci006
Otto FJ (1990) DAPI staining of fixed cells for high-resolution flow cytometry of nuclear DNA. In: Darzynkiewiez Z, Crissman HA, Robinson JP (eds) Methods in cell biology, vol 33. Academic, San Diego, pp 105–110
Pasternak T, Prinsen E, Ayaydin F, Miskolczi P, Potters G, Asard H, van Onckelen H, Dudits D, Fehér A (2002) The role of auxin, pH and stress in the activation of embryogenic cell division in leaf protoplast-derived cells of alfalfa (Medicago sativa L.). Plant Physiol 129:1807–1819. doi:10.1104/pp.000810
Pinto G, Loureiro J, Lopes T, Santos C (2004) Analysis of the genetic stability of Eucalyptus globulus Labill. somatic embryos by flow cytometry. Theor App Genet 109:580–587. doi:10.1007/s00122-004-1655-3
Praça-Fontes MM, Carvalho CR, Clarindo WR (2011a) C-value reassessment of plant standards: an image cytometry approach. Plant Cell Rep 30:2303–2312. doi:10.1007/s00299-011-1135-6
Praça-Fontes MM, Carvalho CR, Clarindo WR, Cruz CD (2011b) Revisiting the DNA C-values of the genome size-standards used in plant flow cytometry to choose the ‘‘best primary standards’’. Plant Cell Rep 30:1183–1191. doi:10.1007/s00299-011-1026-x
Price HJ, Hodnett G, Jhonston S (2000) Sunflower (Helianthus annus) leaves contain compounds that reduce nuclear propidium iodide fluorescence. Ann Bot 86:929–934. doi:10.1006/anbo.2000.1255
Quiroz-Figueroa F, Méndez-Zeel M, Sánchez-Teyer F, Rojas-Herrera R, Loyola-Vargas VM (2002) Differential gene expression in embryogenic and non-embryogenic clusters from cell suspension cultures of Coffea arabica. J Plant Physiol 159:1267–1270. doi:10.1078/0176-1617-00878
Raina SN, Mukai Y, Yamamoto M (1998) In situ hybridization identifies the diploid progenitor species of Coffea arabica (Rubiaceae). Theor App Genet 97:1204–1209. doi:10.1007/s001220051011
Santos-Briones C, Hernández-Sotomayor SMT (2006) Coffee biotechnology. Braz J Plant Physiol 18:217–227. doi:10.1590/S1677-04202006000100015
Schwencke J, Bureau JM, Crosnier MT, Brown S (1998) Cytometric determination of genome size and base composition of tree species of three genera of Casuarinaceae. Plant Cell Rep 18:346–349. doi:10.1007/s002990050584
Shapiro HM (2003) Practical flow cytometry. Wiley-Liss, New Jersey
Singh RJ (2003) Plant cytogenetics, 2nd edn. CRC, Boca Raton
Suda J, Leitch IJ (2010) The quest for suitable reference standards in genome size research. Cytometry 77A:717–720. doi:10.1002/cyto.a.20907
van Boxtel J, Berthouly M (1996) High frequency somatic embryogenesis from coffee leaves. Factors influencing embryogenesis and subsequent proliferation and regeneration in liquid medium. Plant Cell Tissue Organ Cult 44:4–17. doi:10.1007/bf00045907
Yu Q, Guyo R, Kochko A, Byers A, Navajas-Pérez R, Langston BJ, Dubreuil-Tranchant C, Paterson AH, Poncet V, Nagai C, Ming R (2011) Micro-collinearity and genome evolution in the vicinity of an ethylene receptor gene of cultivated diploid and allotetraploid coffee species (Coffea). Plant J 67:305–317. doi:10.1111/j.1365-313X.2011.04590.x
Acknowledgments
We thank FAPES (Fundação de Amparo à Pesquisa do Espírito Santo), FAPEMIG (Fundação de Amparo à Pesquisa de Minas Gerais), CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), CBP&D/Café (Consórcio Brasileiro de Pesquisa e Desenvolvimento do Café) and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior), Brazil, for their financial support. We also thank BioCafé/BIOAGRO (Viçosa-MG, Brazil) for their support, especially to Dr. Eveline Teixeira Caixeta for her technical assistance and advice.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Clarindo, W.R., Carvalho, C.R. & Mendonça, M.A.C. Cytogenetic and flow cytometry data expand knowledge of genome evolution in three Coffea species. Plant Syst Evol 298, 835–844 (2012). https://doi.org/10.1007/s00606-012-0595-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-012-0595-7