Abstract
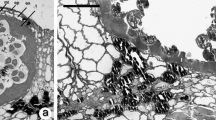
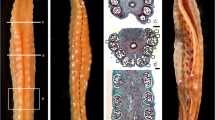
We investigated the mechanism of antheridial dehiscence in ferns for the first time using fluorescence microscopy as well as scanning and transmission electron microscopy. The mechanism leading to antheridial dehiscence in Polystichum setiferum, Asplenium trichomanes and A. onopteris was found to depend on the different cellulose contents of the inner and outer walls of the ring cells detected with calcofluor white stain and the Thiéry test. The extremely low cellulose content of the ring cell walls facing spermatozoids made them less mechanically resilient than external wall cells. When the ring cells absorbed water they expanded only into the antheridial cavity, pushing the gametes against the cap cell, which detached from the ring cell below and enabled spermatozoid release. The newly released spermatozoids were spherical bodies covered in cellulose fibrils. The significance of cellulose fibrils could be to isolate the gametes from each other, to reinforce the electron transparent material and to protect the gamete from pressure created by the ring cells during release.



Similar content being viewed by others
References
Atkinson LR, Stokey AG (1973) The gametophytes of some Jamaican thelypteroid ferns. J Linn Soc Bot 66:23–36
Cave CF, Bell PR (1973) The cytochemistry of the walls of the spermatocytes of Ceratopteris thalictroides. Planta 109:99–104
Elmore MW, Adams RJ (1976) Scanning electron microscopic observations of the gametophytes and sperm of the bracken fern, Pteridium aquilinum (L.) Kuhn. New Phytol 76:519–522
Ferrarini E, Ciampolini F, Pichi Sermolli REG, Marchetti D (1986) Iconographya palynologica pteridophytorum Italiane. Webbia 40:1–202
Godwin H (1968) The origin of the exine. New Phytol 67:667–676
Gori P (1982) Accumulation of polysaccharides in the anther cavity of Allium sativum, cl Piemonte. J Ultrastruct Res 81:158–162
Gori P, Ferri S (1982) Ultrastructural study of the microspore development in Allium sativum, cl Piemonte. J Ultrastruct Res 79:341–349
Gori P, Muccifora S, Woo S, Bellani L (1997) An ultrastructural study of the mature spermatozoid of the fern Asplenium trichomanes L. subsp. trichomanes. Sex Plant Reprod 10:142–148
Hartman ME (1931) Antheridial dehiscence in the Polypodiaceae. Bot Gaz (London) 91:252–276
Heslop-Harrison J (1966) Cytoplasmic continuities during spore formation in flowering plants. Endeavour 25:65–72
Hughes J, McCully M (1975) The use of an optical brightener in the study of plant structure. Stain Technol 50:319–329
Kotenko JL (1990) Spermatogenesis in a homosporous fern, Onoclea sensibilis. Am J Bot 77:809–825
Muccifora S, Gori P (2005) Ultrastructure of mature spermatozoids in the fern Asplenium onopteris L. Micron 36:539–544
Muccifora S, Bellani LM, Gori P (1996) Spermatogenesis in Asplenium trichomanes ssp. trichomanes. Giorn Bot Ital 130:945–947
Muccifora S, Woo S, Gori P (2000) Ultrastructural features of spermatocytes and spermatozoids in the fern Phyllitis scolopendrium (L.) Newm. subsp. scolopendrium. Sex Plant Reprod 12:323–331
Muñiz-Díaz de Leon ME, Pérez-García B, Márquez-Guzmán J, Mendoza-Ruiz A (2008) Developmental gametophyte morphology of seven species of Thelypteris subg. Cyclosorus (Thelypteridiaceae). Micron 39:1351–1362
Nester JE (1985) Scanning electron microscopy of antheridia and archegonia of Aneimia mexicana Klotzsh. Am J Bot 72:777–780
O’Brien K, McCully ME (1981) The study of plant structure – principles and selected methods. Thermacarphi, Melbourne
Pacini E (2010) Relationships between tapetum, loculus, and pollen during development. Int J Plant Sci 171:1–11
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Ruzin SE (1999) Plant microtechnique and microscopy. Oxford University Press, New York
Smith AR, Pryer KM, Schuettpelz E, Korall P, Schneider H, Wolf PG (2006) A classification for extant ferns. Taxon 55:705–731
Teng NJ, Huang ZH, Mu XJ, Jin B, Hu YX, Lin JX (2005) Microsporogenesis and pollen development in Leymus chinensis with emphasis on dynamic changes in callose deposition. Flora 200:256–263
Thiéry J (1967) Mise en évidence des polysaccharides sur coupes fines en microscopie électronique. J Microsc 6:987–1018
Tigerschiöld E (1989) Scanning electron microscopy of gametophyte characters and antheridial opening in some Ceylonese species of Thelypteridaceae. Nordic J Bot 8:639–648
Tigerschiöld E (2008) Dehiscence of antheridia in thelypteroid ferns. Nordic J Bot 9:407–412
Tutin TG, Burges NA, Schater AO, Edmonson JR, Heywood WH, Moore DH, Walters SM, Webb DA (1993) Flora europea psilotaceae to platanaceae, vol. I, 2nd edn. Cambridge University Press, Cambridge
Watson ML (1958) Staining of tissue sections for electron microscopy with heavy metals. J Biophys Biochem Cytol 4:475–478
Acknowledgments
The authors thank Prof. Ettore Pacini for revision of the manuscript and help with line drawings in Fig. 3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Muccifora, S., Bellani, L.M. Antheridial dehiscence in ferns. Plant Syst Evol 297, 51 (2011). https://doi.org/10.1007/s00606-011-0498-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-011-0498-z