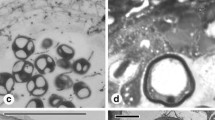
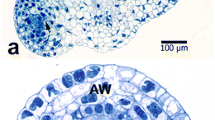
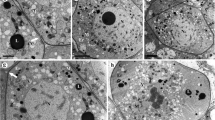
Abstract
Despite that there is some literature on pollen morphology of Rhamnaceae, studies addressing general aspects of the microsporogenesis, microgametogenesis, and anther development are rare. The aim of this paper is to describe the ultrastructure of pollen grain ontogeny with special attention to tapetum cytology in Hovenia dulcis. Anthers at different stages of development were processed for transmission and scanning electron microscopy, bright-field microscopy, and fluorescence microscopy. Different histochemical reactions were carried out. The ultrastructural changes observed during the development of the tapetal cells and pollen grains are described. Large vesicles containing carbohydrates occur in the tapetal cell cytoplasm during the early stages of pollen development. Its origin and composition are described and discussed. This is the first report on the ontogeny and ultrastructure of the pollen grain and related sporophytic structures of H. dulcis.







Similar content being viewed by others
References
Bieleski RL, Redgwell RJ (1980) Sorbitol metabolism in nectaries from flowers of Roseaceae. Aust J Plant Physiol 7:15–25
Cho JY, Moon JH, Eun JB, Chung SJ, Park KH (2004) Isolation and characterization of 3(Z)-dodecenedioic acid as an antibacterial substance from Hovenia dulcis THUNB. Food Sci Biotechnol 13:46–50
Driouich A, Faye L, Staehelin LA (1993) The plant Golgi apparatus: a factory for complex polysaccharides and glycoproteins. Trends Biochem Sci 18:210–214
Driouich A, Cannesan MA, Dardelle F et al (2012) Unity is strength: the power of border cells and border like cells in relation with plant defense. In: Vivanco JM, Baluska F (eds) Secretions and exudates in biological systems. Springer, Heidelberg, pp 91–108
Galati BG (2003) Ubisch bodies in Angiosperms. In: Pandey K, Dhakal MR (eds) Advances in plant reproductive biology, vol II. Narendra Publishing House, Delhi, pp 1–20
Galati BG, Gotelli MM, Rosenfeldt S, Torretta JP, Zarlavsky G (2010) Orbicules in relation to the pollination modes. In: Kaiser BJ (ed) Pollen: structure, types and effects. Nova, New York, pp 1–15
Gotelli M, Galati B, Medan D (2012) Pollen, Tapetum, and Orbicule Development in Colletia paradoxa and Discaria americana (Rhamnaceae). ScientificWorldJournal: Article ID 948469. doi:10.1100/2012/948469
Hernández-Pinzón I, Ross JHE, Barnes KA, Damant AP, Murphy DJ (1999) Composition and role of tapetal lipid bodies in the biogenesis of the pollen coat of Brassica napus. Planta 208:588–598
Hsieh K, Huang AHC (2005) Lipid-rich tapetosomes in Brassica tapetum are composed of oleosin-coated oil droplets and vesicles, both assembled in and then detached from the endoplasmic reticulum. Plant J 43:889–899
Hsieh K, Huang AHC (2007) Tapetosomes in Brassica tapetum accumulate endoplasmic reticulum-derived flavonoids and alkanes for delivery to the pollen surface. Plant Cell 19:582–596
Huysmans S, El-Ghazaly G, Smets E (1998) Orbicules in Angiosperms: morphology, function, distribution, and relation with tapetum types. Bot Rev 64:240–272
Jensen WA (1962) Botanical Hystochemistry. Freeman, San Francisco
Johri BM, Ambegaokar KB, Srivastava PS (1992) Comparative embryology of angiosperms, vol 1–2. Springer, Berlin
Lerouxel O, Cavalier DM, Liepman AH, Keegstra K (2006) Biosynthesis of plant cell wall polysaccharides—a complex process. Curr Opin Plant Biol 9:621–630
Maheshwari P (1950) An introduction to the embryology of angiosperms. McGraw-Hill, New York
McCue AD, Cresti M, Feijo JA, Slotkin RK (2011) Cytoplasmic connection of sperm cells to the pollen vegetative cell nucleus: potential roles of the male germ unit revisited. J Exp Bot 62:1621–1631
Medan D, Schirarend C (2004) Rhamnaceae. In: Kubitzki K (ed) The families and genera of vascular plants VI. Flowering plants – dicotyledons: Celastrales, Oxalidales, Rosales, Cornales, Ericales. Springer Verlag, Heidelberg
Mollenhauer HH, Morré DJ (1966) Tubular connections between dictyosomes and forming secretory vesicles in plant Golgi apparatus. J Cell Biol 29:373–376
Murphy DJ, Ross JHE (1998) Biosynthesis, targeting and processing of oleosin-like proteins, which are major pollen coat components in Brassica napus. Plant J 12:1–16
Okuma Y, Ishikawa H, Ito Y, Hayashi Y, Endo A, Watanabe T (1995) Effects of extracts from Hovenia dulcis Thunb. on alcohol concentration in rats and men administered alcohol. J Jpn Soc Food Sci Technol 48:167–172
Papagiannes E (1974) Pollen studies of selected genera of Rhamnaceae, Master Thesis. University of Illinois at the Chicago Circle, Chicago, Illinois
Pearse AGE (1961) Histochemistry, theoretical and applied, 2nd edn. Little Brown, Boston
Perveen A, Qaiser M (2005) Pollen Flora of Pakistan –XLIV. Rhamnaceae. Pakistan J Bot 37:195–202
Radice S, Galati BG (2003) Floral nectary ultrastructure of Prunus persica (L.) Batch cv. Forastero (Newcomer), an Argentine peach. Plant Syst Evol 238:23–32
Raghavan V (1997) Molecular embryology of flowering plants. Cambridge University Press, UK, pp 357–393
Ross JHE, Murphy DJ (1996) Characterization of antherexpressed genes encoding a major class of extracellular oleosinlike proteins in the pollen coat of Brassicaceae. Plant J 9:625–637
Ruiter RK, van Eldik GJ, van Herpen RMA, Schrauwen JAM (1997) Characterization of oleosins in the pollen coat of Brassica oleracea. Plant Cell 9:1621–1631
Schirarend C (1996) Pollen morphology of the genus Paliurus (Rhamnaceae). Grana 35:347–356
Schirarend C, Köhler E (1993) Rhamnaceae Juss. World Pollen and Spore Flora 17(18):1–53
Stpiczynska M (2002) Nectar resorption in the spur of Platanthera chloranta Custer (Rchb.) Orchidaceae. Nectar and Nectary: from biology to biotechnology. Montalcino, Siena, pp 28–31
Suzuki T, Tsunekawa S, Koizuka C et al (2013) Development and disintegration of tapetum-specific lipid-accumulating organelles, elaioplasts and tapetosomes, in Arabidopsis thaliana and Brassica napus. Plant Sci 207:25–36
Ubisch G (1927) Kurze mitteilungen zur entwicklungsgeschiehte der antheren. Planta 3:490–495
Wu SSH, Platt KA, Ratnayake C, Wang TW, Ting JTL, Huang AHC (1997) Isolation and characterization of novel neutrallipid- containing organelles and globuli-filled plastids from Brassicanapus tapetum. Proc Natl Acad Sci U S A 94:12711–12716
Xiang J, Zhu W, Han J, Li Z, Ge H, Lin D (2012) Analysis of organic acids in Chinese raisin tree (Hovenia dulcis) peduncle and their changes in liquid fermentation process. Food Sci Biotechnol 21:1119–1127
Yu HS, Hu SY, Zhu C (1989) Ultrastructure of sperm cells and the male germ unit in pollen tubes of Nicotiana tabacum. Protoplasma 152:29–36
Yun CW, Lee BC (2002) Vegetation structure of Hovenia dulcis community in South Korea. Korean J Ecol 25:93–99
Zarlavsky GE (2014) Histología Vegetal: Técnicas Simples y Complejas. Sociedad Argentina de Botánica, Buenos Aires
Funding
This research was financially supported by a grant (UBACyT 2013–2016 GC 20020120100056BA) to B. Galati. M. Gotelli and D. Medan are affiliated with CONICET, Argentina.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling Editor: Alexander Schulz
Rights and permissions
About this article
Cite this article
Gotelli, M.M., Galati, B.G., Zarlavsky, G. et al. Pollen and microsporangium development in Hovenia dulcis (Rhamnaceae): a different type of tapetal cell ultrastructure. Protoplasma 253, 1125–1133 (2016). https://doi.org/10.1007/s00709-015-0870-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-015-0870-x