Abstract
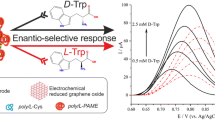
Chiral materials have drawn the widespread attention for their its chiral recognition ability. The design and synthesis of chiral material are of importance owing to the unpredictability in controlling chirality during the synthesis process. To circumvent problems, a chiral MOF (D-His-ZIF-8) was synthesized by ligand exchange of 2-methylimidazole (Hmim) on ZIF-8 by D-histidine (D-His), which can be treated as chiral host to distinguish amino acid enantiomers. The obtained D-His-ZIF-8 can provide chiral nanochannels for amino acid guests. Meanwhile, polynary transition-metal ion (Co2+ and Fe3+) coordinating with polydopamine (PDA) wrapped on the surface of D-His-ZIF-8 can increase the active sites. The electrochemical chiral recognition behavior showed that D-His-ZIF-8@CoFe-PDA exhibited good recognition of the tryptophan enantiomer (L/D-Trp) (working potential of −0.2 V vs. Hg/HgCl2). The LOD and LOQ of L-Trp were 0.066 mM and 0.22 mM, respectively, while the LOD and LOQ of D-Trp were 0.15 mM and 0.50 mM, respectively. Finally, the usefulness of D-His-ZIF-8@CoFe-PDA/GCE was evaluated with a recovery of 94.4–103%. The analysis of real samples shows that D-His-ZIF-8@CoFe-PDA/GCE is a feasible sensing platform for the detection of L-Trp and D-Trp.
Graphical abstract






Similar content being viewed by others
References
Dong B, Liu J, Xue M, Ni Z, Guo Y, Huang Z, Zhang Z (2021) One-fold anisotropy of silver chiral nanoparticles studied by second-harmonic generation. ACS Sens 6:454–460. https://doi.org/10.1021/acssensors.0c02031
Bromfield S, Smith D (2015) Heparin versus DNA: chiral preferences in polyanion binding to self-assembled multivalent (SAMul) nanostructures. J Am Chem Soc 137:10056–10059. https://doi.org/10.1021/jacs.5b04344
Huang Y, Nguyen M, Natarajan A, Nguyen V, Kuzyk A (2018) A DNA Origami-based chiral plasmonic sensing device. ACS Appl Mater Interfaces 10:44221–44225. https://doi.org/10.1021/acsami.8b19153
Sholl D, Gellman A (2009) Developing chiral surfaces for enantioselective chemical processing. AIChE J 55:2484–2490. https://doi.org/10.1002/aic.12036
Chen Y, Deng K, Lei S, Yang R, Li T, Gu Y, Yang Y, Qiu X, Wang C (2018) Single-molecule insights into surface-mediated homochirality in hierarchical peptide assembly. Nat Commun 9:2711. https://doi.org/10.1038/s41467-018-05218-0
He S, Shang X, Lu W, Tian Y, Xu Z, Zhang W (2021) Electrochemical enantioselective sensor for effective recognition of tryptophan isomers based on chiral polyaniline twisted nanoribbon. Anal Chim Acta 1147:155–164. https://doi.org/10.1016/j.aca.2020.12.058
Yang X, Li Z, Polyakova T, Dejneka A, Zablotskii V, Zhang X (2020) Effect of static magnetic field on DNA synthesis: the interplay between DNA chirality and magnetic field left-right asymmetry. FASEB Bioadv 2:254–263. https://doi.org/10.1096/fba.2019-00045
Li Z, Xu H, Wu D, Zhang J, Liu X, Gao S, Kong Y (2019) Electrochemical chiral recognition of tryptophan isomers based on nonionic surfactants assisted molecular imprinting sol-gel silica. ACS Appl Mater Interfaces 11:2840–2848. https://doi.org/10.1021/acsami.8b19399
Niu Q, Jin P, Huang Y, Fan L, Zhang C, Yang C, Dong C, Liang W, Shuang S (2022) A selective electrochemical chiral interface based on a carboxymethyl-β-cyclodextrin/Pd@Au nanoparticles/3D reduced graphene oxide nanocomposite for tyrosine enantiomer recognition. Analyst 147:880–888. https://doi.org/10.1039/D1AN02262H
Shang X, Park C, Jung G, Kwak S, Oh J (2018) Highly enantioselective graphene-based chemical sensors prepared by chiral noncovalent functionalization. ACS Appl Mater Interfaces 10:36194–36201. https://doi.org/10.1021/acsami.8b13517
Okur S, Qin P, Chandresh A, Li C, Zhang Z, Lemmer U, Heinke L (2021) An enantioselective e-nose: an array of nanoporous homochiral MOF films for stereospecific sensing of chiral odors. Angew Chem Int Ed 60:3566–3571. https://doi.org/10.1002/anie.202013227
Liu J, Zhou H, Yang W, Ariga K (2020) Soft nanoarchitectonics for enantioselective biosensing. Acc Chem Res 53:644–653. https://doi.org/10.1021/acs.accounts.9b00612
Jang S, Kim H (2020) Direct chiral 19F NMR analysis of fluorine-containing analytes and its application to simultaneous chiral analysis. Org Lett 22:7804–7808. https://doi.org/10.1021/acs.orglett.0c02620
Saz J, Marina M (2016) Recent advances on the use of cyclodextrins in the chiral analysis of drugs by capillary electrophoresis. J Chromatogr A 1467:79–94. https://doi.org/10.1016/j.chroma.2016.08.029
Villar-Guerra R, Trent J, Chaires J (2018) G-quadruplex secondary structure from circular dichroism spectroscopy. Angew Chem Int Ed 57:7171–7175. https://doi.org/10.1002/ange.201709184
Zhang X, Yin J, Yoon J (2014) Recent advances in development of chiral fluorescent and colorimetric sensors. Chem Rev 114:4918–4959. https://doi.org/10.1021/cr400568b
Song G, Xu C, Li B (2015) Visual chiral recognition of mandelic acid enantiomers with L-tartaric acid−capped gold nanoparticles as colorimetric probes. Sens Actuators B: Chem 215:504–509. https://doi.org/10.1016/j.snb.2015.03.109
Nalbant Esenturk E, Hight Walker A (2009) Surface-enhanced Raman scattering spectroscopy via gold nanostars. J Raman Spectrosc 40:86–91. https://doi.org/10.1002/jrs.2084
Niu X, Yang X, Mo Z, Liu N, Guo R, Pan Z, Liu Z (2019) Electrochemical chiral sensing of tryptophan enantiomers by using 3D nitrogen-doped reduced graphene oxide and self-assembled polysaccharides. Mikrochim Acta 186:557. https://doi.org/10.1007/s00604-019-3682-4
Sun Y, Jiang X, Jin H, Gui R (2019) Ketjen black/ferrocene dual-doped MOFs and aptamer-coupling gold nanoparticles used as a novel ratiometric electrochemical aptasensor for vanillin detection. Anal Chim Acta 1083:101–109. https://doi.org/10.1016/j.aca.2019.07.027
Yu S, Wang Y, Chatterjee S, Liang F, Zhu F, Li H (2021) Pillar[5]arene-functionalized nanochannel platform for detecting chiral drugs. Chinese Chem Lett 32:179–183. https://doi.org/10.1016/j.cclet.2020.11.055
Wang L, Gao W, Na S, Pumera M (2021) Chiral protein−covalent organic framework 3D-printed structures as chiral biosensors. Anal Chem 93:5277–5283. https://doi.org/10.1021/acs.analchem.1c00322
Wu S, Ye Q, Wu D, Tao Y, Kong Y (2020) Enantioselective recognition of chiral tryptophan with achiral glycine through the strategy of chirality transfer. Anal Chem 92:11927–11934. https://doi.org/10.1021/acs.analchem.0c02335
Yang Y, Li M, Zhu Z (2021) A disposable dual-signal enantioselective electrochemical sensor based on stereogenic porous chiral carbon nanotubes hydrogel. Talanta 232:122445. https://doi.org/10.1016/j.talanta.2021.122445
Yang Z, Zhu C, Li Z, Liu Y, Liu G, Cui Y (2014) Engineering chiral Fe(salen)-based metal–organic frameworks for asymmetric sulfide oxidation. Chem Commun 50:8775–8778. https://doi.org/10.1039/C4CC03308F
Ma L, Abney C, Lin W (2009) Enantioselective catalysis with homochiral metal–organic frameworks. Chem Soc Rev 38:1248–1256. https://doi.org/10.1039/B807083K
Zhang J, Xie S, Zi M, Yuan L (2020) Recent advances of application of porous molecular cages for enantioselective recognition and separation. J Sep Sci 43:134–149. https://doi.org/10.1002/jssc.201900762
Wei X, Chen Y, He S, Lian H, Cao X, Liu B (2021) L-histidine-regulated zeolitic imidazolate framework modified electrochemical interface for enantioselective determination of L-glutamate. Electrochimica Acta 400:139464. https://doi.org/10.1016/j.electacta.2021.139464
Niu X, Yan S, Chen J, Li H, Wang K (2022) Enantioselective recognition of L/D-amino acids in the chiral nanochannels of a metal-organic framework. Electrochimica Acta 405:139809. https://doi.org/10.1016/j.electacta.2021.139809
Xu M, Chen Q, Xie L, Li J (2020) Exchange reactions in metal-organic frameworks: New advances. Coordin Chem Rev 421. https://doi.org/10.1016/j.ccr.2020.213421
He T, Kong X, Zhou J, Zhao C, Wang K, Wu X, Lv X, Si G, Li J, Nie Z (2021) A practice of reticular chemistry: construction of a robust mesoporous palladium metal-organic framework via metal metathesis. J Am Chem Soc 143:9901–9911. https://doi.org/10.1021/jacs.1c04077
Liang W, Rong Y, Fan L, Zhang C, Dong W, Li J, Niu J, Yang C, Shuang S, Dong C, Wong W (2019) Simultaneous electrochemical sensing of serotonin, dopamine and ascorbic acid by using a nanocomposite prepared from reduced graphene oxide, Fe3O4 and hydroxypropyl-β-cyclodextrin. Microchimica Acta 186. https://doi.org/10.1007/s00604-019-3861-3
Wang H, Chen X, Li W, Zhou W, Guo X, Kang W, Kou D, Zhou Z, Meng Y, Tian Q, Wu S (2018) ZnO nanotubes supported molecularly imprinted polymers arrays as sensing materials for electrochemical detection of dopamine. Talanta 176:573–581. https://doi.org/10.1016/j.talanta.2017.08.083
Xie G, Tian W, Wen L, Xiao K, Zhang Z, Liu Q, Hou G, Li P, Tian Y, Jiang L (2015) Chiral recognition of L-tryptophan with beta-cyclodextrin-modified biomimetic single nanochannel. Chem Commun 51:3135–3138. https://doi.org/10.1039/C4CC09577D
Feng W, Liu C, Lu S, Zhang C, Zhu X, Liang Y, Nan J (2014) Electrochemical chiral recognition of tryptophan using a glassy carbon electrode modified with β-cyclodextrin and graphene. Microchimica Acta 181:501–509. https://doi.org/10.1007/s00604-014-1174-0
Yu Y, Xu N, Zhang J, Wang B, Xie S, Yuan L (2020) Chiral metal-organic framework D-his-ZIF-8@SiO2 core–shell microspheres used for HPLC enantioseparations. ACS Appl. Mater Interfaces 12:16903–16911. https://doi.org/10.1021/acsami.0c01023
Wang Z, Jin X, Zhu C, Liu Y, Tan H, Ku R, Zhang Y, Zhou L, Liu Z, Hwang S, Fan H (2021) Atomically dispersed Co2–N6 and Fe–N4 costructures boost oxygen reduction reaction in both alkaline and acidic media, Fan. Adv Mater 33:e2104718. https://doi.org/10.1002/adma.202104718
Liang Y, Wei J, Hu Y, Chen X, Zhang J, Zhang X, Jiang S, Tao S, Wang H (2017) Metal-polydopamine frameworks and their transformation to hollow metal/N-doped carbon particles. Nanoscale 9:5323–5328. https://doi.org/10.1039/C7NR00978J
Pei H, Chen F, Niu X, Jia Q, Guo R, Liu N, Mo Z (2021) Self-assembled chitosan-sodium alginate composite material for electrochemical recognition of tyrosine isomers. J Electroanal Chem 895. https://doi.org/10.1016/j.jelechem.2021.115525
Wang Y, Wang L, Huang W, Zhang T, Hu X, Perman J, Ma S (2017) A metal–organic framework and conducting polymer based electrochemical sensor for high performance cadmium ion detection. J Mater Chem A 5:8385–8393. https://doi.org/10.1039/C7TA01066D
Feng S, Li Y, Zhang R, Li Y (2019) A novel electrochemical sensor based on molecularly imprinted polymer modified hollow N, S-Mo2C/C spheres for highly sensitive and selective carbendazim determination. Biosens. Bioelectron 142:111491. https://doi.org/10.1016/j.bios.2019.111491
Zou J, Yu J (2020) Nafion-stabilized black phosphorus nanosheets-maltosyl-β-cyclodextrin as a chiral sensor for tryptophan enantiomers. Mater Sci Eng C 112:110910. https://doi.org/10.1016/j.msec.2020.110910
Xu J, Wang Q, Xuan C, Xia Q, Lin X, Fu Y (2016) Chiral recognition of tryptophan enantiomers based on β-cyclodextrin-platinum nanoparticles/graphene nanohybrids modified electrode. Electroanalysis 28:868–873. https://doi.org/10.1002/elan.201500548
Bao L, Tao Y, Gu X, Yang B, Deng L, Kong Y (2016) Potato starch as a highly enantioselective system for temperature-dependent electrochemical recognition of tryptophan isomers. Electrochem Commun 64:21–25. https://doi.org/10.1016/j.elecom.2016.01.004
Jing P, Yin Z, Cai W, Li J, Wu D, Kong Y (2022) The hybrids of perylene tetracarboxylic acid functionalized multi-walled carbon nanotubes and chitosan for electrochemical chiral sensing of tryptophan enantiomers. Bioelectrochemistry 146:108110. https://doi.org/10.1016/j.bioelechem.2022.108110
Chen Q, Zhou J, Han Q, Wang Y, Fu Y (2012) Electrochemical enantioselective recognition of tryptophane enantiomers based on chiral ligand exchange. Colloids Surf B Biointerfaces 92:130–135. https://doi.org/10.1016/j.colsurfb.2011.11.031
Zou J, Lan X, Zhao G, Huang Z, Liu Y, Yu J (2020) Immobilization of 6-O-alpha-maltosyl-beta-cyclodextrin on the surface of black phosphorus nanosheets for selective chiral recognition of tyrosine enantiomers. Mikrochim Acta 187:636. https://doi.org/10.1007/s00604-020-04606-y
Li Z, Mo Z, Meng S, Gao H, Niu X, Guo R, Wei T (2017) The construction of electrochemical chiral interfaces using hydroxypropyl chitosan. RSC Adv 7:8542–8549. https://doi.org/10.1039/C6RA27709H
Funding
This work was supported by the National Nature Science Foundations of China (grant no. 22065021), the Key Research Program of Gansu Province (21YF5GA076), the Province Nature Science Foundations of Gansu (grant no. 21JR7RA213), the Lanzhou Talent Innovation and Entrepreneurship Project (grant no. 2022-RC-33), the Hongliu Outstanding Youth Teacher Cultivate Project of Lanzhou University of Technology, and the Hongliu Excellent Youth Teacher Cultivate Project of Lanzhou University of Technology.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Niu, X., Zhao, R., Yan, S. et al. Chiral MOFs encapsulated by polymers with poly-metallic coordination as chiral biosensors. Microchim Acta 190, 230 (2023). https://doi.org/10.1007/s00604-023-05807-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-023-05807-x