Abstract
Simultaneous cathodic and anodic electrochemiluminescence (ECL) emissions of needle-like nanostructures of Ru(bpy)32+ (RuNDs) as the only luminophore are reported based on different co-reactants. Cathodic ECL was attained from RuNDs/K2S2O8 system, while anodic ECL was achieved from RuNDs/black phosphorus quantum dots (BPQDs) system. Ferrocene attached to the hairpin DNA could quench the cathodic and anodic ECL simultaneously. Subsequently, the ECL signals recovered in the presence of tumor marker mucin 1 (MUC1), which made it possible to quantitatively detect MUC1. The variation of ECL signal was related linearly to the concentrations of MUC1 in the range 20 pg mL−1 to 10 ng mL−1, and the detection limits were calculated to 2.5 pg mL−1 (anodic system, 3σ) and 6.2 pg mL−1 (cathodic system, 3σ), respectively. The recoveries were 97.0%, 105%, and 95.2% obtained from three human serum samples, and the relative standard deviation (RSD) is 5.3%. As a proof of concept, this work realized simultaneous ECL emission of a single luminophore, which initiates a new thought in biomarker ECL detection beyond the traditional ones.
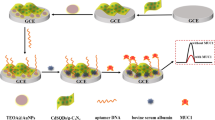
Graphical abstract

Simultaneous cathodic and anodic ECL emissions of RuNDs were reported based on different co-reactants. Ferrocene could quench the ECL emission in the cathode and the anode simultaneously. Thus, an aptasensor was constructed based on the variation of ECL intensity. As a proof of concept, this work realized simultaneous ECL emission of a single luminophore, which initiates a new thought in biomarker ECL detection beyond the traditional ones by avoiding the false positive signals.






Similar content being viewed by others
References
Lv WX, Ye HC, Yuan ZQ, Liu XJ, Chen X, Yang WS (2020) Recent advances in electrochemiluminescence-based simultaneous detection of multiple targets. Trac-Trends Anal Chem 123:115767. https://doi.org/10.1016/j.trac.2019.115767
Zhao JW, Du JW, Luo JH, Chen SH, Yuan R (2020) A novel potential-resolved electrochemiluminescence immunosensor for the simultaneous determination of brain natriuretic peptide and cardiac troponin I. Sensors Actuators B Chem 311:127934. https://doi.org/10.1016/j.snb.2020.127934
Fu XL, Hou F, Liu FR, Ren SW, Cao JT, Liu YM (2019) Electrochemiluminescence energy resonance transfer in 2D/2D heterostructured g-C3N4/MnO2 for glutathione detection. Biosens Bioelectron 129:72–78
Zhou Y, Chai YQ, Yuan R (2019) Highly efficient dual-polar electrochemiluminescence from Au25 nanoclusters: the next generation of multibiomarker detection in a single step. Anal Chem 91:14618–14623
Fang DD, Li JN, Huang DF, Dai H, Lin YY (2020) Dual-modality probe based on black phosphorous and NiFe2O4 NTs for electrochemiluminescence and photothermal detection of ovarian cancer marker. Talanta 211:120660. https://doi.org/10.1016/j.talanta.2019.120660
Fang DD, Zhao DD, Zhang SP, Huang YT, Dai H, Lin YY (2020) Black phosphorus quantum dots functionalized MXenes as the enhanced dual-mode probe for exosomes sensing. Sensors Actuators B 305:127544. https://doi.org/10.1016/j.snb.2019.127544
Wei J, Chang WD, Qileng A, LIiu WP, Zhang Y, Rong SY, Lei HT, Liu YJ (2018) Dual-modal split-type immunosensor for sensitive detection of microcystin-LR: enzyme-induced photoelectrochemistry and colorimetry. Anal Chem 90:9606–9613
Wu MS, Shi HW, He LJ, Xu JJ, Chen YY (2012) Microchip device with 64-site electrode array for multiplexed immunoassay of cell surface antigens based on electrochemiluminescence resonance energy transfer. Anal Chem 84:4207–4213
Babamiri B, Hallaj R, Salimi A (2018) Ultrasensitive electrochemiluminescence immunoassay for simultaneous determination of CA125 and CA15-3 tumor markers based on PAMAM-sulfanilic acid-Ru(bpy)32+ and PAMAM-CdTe@CdS nanocomposite. Biosens Bioelectron 99:353–360
Guo ZY, Wu L, Hu YF, Wang S, Li X (2017) Potential-resolved “in-electrode” type electrochemiluminescence immunoassay based on functionalized g-C3N4 nanosheet and Ru-NH2 for simultaneous determination of dual targets. Biosens Bioelectron 95:27–33
Doeven EH, Zammit EM, Barbante GJ, Francis PS, Barnett NW, Hogan CF (2013) A potential-controlled switch on/off mechanism for selective excitation in mixed electrochemiluminescent systems. Chem Sci 4:977–982
Haghighatbin MA, Lo SC, Burn PL, Hogan CF (2016) Electrochemically tuneable multi-colour electrochemiluminescence using a single emitter. Chem Sci 7:6974–6980
Wang HJ, Song YH, Chai YQ, Yuan R (2019) Highly sensitive biosensor based on target induced dual signal amplification to electrochemiluminescent nanoneedles of Ru(II)complex. Biosens Bioelectron 140:111344. https://doi.org/10.1016/j.bios.2019.111344
Xu YJ, Shi Z, Shi XY, Zhang K, Zhang H (2019) Recent progress in black phosphorus and black-phosphorus-analogue materials: properties, synthesis and applications. Nanoscale 11:14491–14527
Gui RJ, Jin H, Sun YJ, Jiang XW, Sun ZJ (2019) Two-dimensional group-VA nanomaterials beyond black phosphorus: synthetic methods, properties, functional nanostructures and applications. J Mater Chem A 7:25712–25771
Sun ZB, Xie HH, Tang SY, Yu XF, Guo ZN, Shao JD, Zhang H, Huang H, Wang HY, Chu PK (2015) Ultrasmall black phosphorus quantum dots: synthesis and use as photothermal agents. Angew Chem Int Ed 54:11526–11530
Tao W, Zhu XB, Yu XH, Zeng XW, Xiao QL, Zhang XD, Ji XY, Wang XS, Shi JJ, Zhang H, Mei L (2017) Black phosphorus nanosheets as a robust delivery platform for cancer theranostics. Adv Mater 29:1–9
Jiang QH, Suomi J, Håkansson M, Niskanen AJ, Kotiranta M, Kulmala S (2005) Cathodic electrogenerated chemiluminescence of Ru(bpy)32+ chelate at oxide-coated heavily doped silicon electrodes. Anal Chim Acta 541:157–163
Liu H, Zhang Y, Dong YP, Chu XF (2019) Electrogenerated chemiluminescence aptasensor for lysozyme based on copolymer nanospheres encapsulated black phosphorus quantum dots. Talanta 199:507–512
Peng Y, Wu S, Sun ZW, Zhu S, Yin YM, Li GX (2020) Multiple signal amplification via coupling DNAzyme with strand displacement reaction for sensitive colorimetric analysis of MUC1. Sensors Actuators B 313:128046. https://doi.org/10.1016/j.snb.2020.128046
Li Z, Mao GB, Du MY, Tian SB, Niu LQ, Ji XH, He ZK (2019) A fluorometric turn-on aptasensor for mucin 1 based on signal amplification via a hybridization chain reaction and the interaction between a luminescent ruthenium(II) complex and CdZnTeS quantum dots. Microchim Acta 186:233. https://doi.org/10.1007/s00604-019-3347-3
Hu R, Wen W, Wang QL, Xiong HY, Zhang XH, Gu HH, Wang SF (2014) Novel electrochemical aptamer biosensor based on an enzyme–gold nanoparticle dual label for the ultrasensitive detection of epithelial tumour marker MUC1. Biosens Bioelectron 53:384–389
Jiang XY, Wang HJ, Wang HJ, Yuan R, Chai YQ (2016) Signal-switchable Electrochemiluminescence system coupled with target recycling amplification strategy for sensitive mercury ion and Mucin 1 assay. Anal Chem 88:9243–9250
Zhang L, Tian KJ, Dong YP, Ding HC, Wang CM (2018) Electrogenerated chemiluminescence of Ru(bpy)32+ at a black phosphorus quantum dot modified electrode and its sensing application. Analyst 143:304–310
Liu YC, Zhang CH, Li XZ, Wu DC (2017) A self-monitored fluorescence DNA anti-counterfeiting system based on silica coated SYBR Green I/DNA gelatin nanoparticles. J Mater Chem C 5:5939–5948
Babamiri B, Bahari D, Salimi A (2019) Highly sensitive bioaffinity electrochemiluminescence sensors: recent advances and future directions. Biosens Bioelectron 142:111530. https://doi.org/10.1016/j.bios.2019.111530
White HS, Bard AJ (1982) Electrogenerated chemiluminescence and chemiluminescence of the Ru(bpy)32+-S2O82− system in acetonitrile-water solutions. J Am Chem Soc 104:6891–6895
Zhuo Y, Wang HJ, Lei YM, Zhang P, Liu JL, Chai YQ, Yuan R (2018) Electrochemiluminescence biosensing based on different modes of switching signals. Analyst 143:3230–3248
Chen XM, Su BY, Song XH, Chen QA, Chen X, Wang XR (2011) Recent advances in electrochemiluminescent enzyme biosensors. TrAC - Trends Anal Chem 30:665–676
Abdussalam A, Yuan F, Ma XG, Du FX, Zholudov YT, Zafar MN, Xu GB (2020) Tris(2,2′-bipyridine)ruthenium(II) electrochemiluminescence using rongalite as coreactant and its application in detection of foodstuff adulteration. J Electroanal Chem 857:113752. https://doi.org/10.1016/j.jelechem.2020.114649
Shi L, Li XJ, Zhu WJ, Wang YG, Du B, Cao W, Wei Q, Pang XH (2017) Sandwich-type electrochemiluminescence sensor for detection of NT-proBNP by using high efficiency quench strategy of Fe3O4@PDA toward Ru(bpy)32+ coordinated with silver oxalate. ACS Sensors 2:1774–1778
Wang XL, Wang XJ, Cheng SS, Ye MQ, Zhang CL, Xian YZ (2020) Near-infrared light-switched MoS2 nanoflakes@gelatin bioplatform for capture, detection, and nondestructive release of circulating tumor cells. Anal Chem 92:3111–3117
Cao WD, Ferrance JP, Demas J, Landers JP (2006) Quenching of the electrochemiluminescence of tris(2,2′-bipyridine) ruthenium(II) by ferrocene and its potential application to quantitative DNA detection. J Am Chem Soc 128:7572–7578
Li Z, Mao GB, Du MY, Tian SB, Niu LQ, Ji XH, He ZK (2019) A fluorometric turn-on aptasensor for mucin 1 based on signal amplification via a hybridization chain reaction and the interaction between a luminescent ruthenium(II) complex and CdZnTeS quantum dots. Microchim Acta 186:1–7
Funding
This work is financially supported by National Natural Science Foundation of China (Nos. 21575002, 61671019), and Natural Science Foundation from the Bureau of Education of Anhui Province (No. KJ2019A0073).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing of interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Optimization of detection condition and comparison of different methods for determination of MUC1 are provided as Supporting Information.
ESM 1
(DOCX 421 kb)
Rights and permissions
About this article
Cite this article
Yin, H., Shi, Y., Liu, H. et al. Dual-potential electrochemiluminescence of single luminophore for detection of biomarker based on black phosphorus quantum dots as co-reactant. Microchim Acta 188, 181 (2021). https://doi.org/10.1007/s00604-021-04833-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-021-04833-x