Abstract
Introduction
Understanding the complex nature of low back pain (LBP) is crucial for effective management. The PainDETECT questionnaire is a tool that distinguishes between neuropathic (NeP), nociceptive (NoP), and ambiguous pain. This study aimed to investigate the relationship between pain classification and lumbar intervertebral degenerative parameters obtained from imaging.
Methods
A cohort study was conducted involving 279 patients, aged 18 years and above, who completed PainDETECT questionnaires and underwent lumbar MRI and/or X-ray scans.
Results
The study included 102 patients with NoP, 78 with ambiguous pain, and 99 with NeP. The NeP group had lower mean age (58.21 vs. 53.63, p < 0.05) and higher mean numerical rating scale score (7.9 vs. 5.9, p < 0.001) compared to the NoP group. A negative correlation was found between PainDETECT scores and pelvic incidence (τ = − 0.177, p = 0.043). The NeP group exhibited significantly higher severity of foraminal stenosis (U = 18.962, p = 0.002), spinal stenosis (U = 14.481, p = 0.005), and Pfirrmann grade (U = 14.221, p = 0.028) compared to the NoP group. A higher proportion of NeP patients had intervertebral disk bulge (96% vs. 78% vs. 78%, p = 0.002) and high-intensity zones (51% vs. 41% vs. 19%, p < 0.001) compared to those with NoP and ambiguous pain.
Conclusion
NeP, as determined by the PainDETECT questionnaire, is associated with more severe neural compression, increased presence of discogenic disease and inflammatory disk severity, and decreased pelvic incidence. This pioneering study establishes a connection between pathological findings and pain categorization, providing clinicians with valuable guidance for formulating tailored management plans and reducing the need for unnecessary pharmacotherapy, imaging, and non-targeted surgical interventions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Low back pain (LBP) is the leading cause of disability world-wide with more than 500 million people at one time significantly limited in their ability to undertake activities of daily living [1]. Additionally, this condition is also extremely debilitating, with an associated increase in depression, anxiety, and sleep disorders [2]. Australia spends roughly $4.8 billion dollars per year on management of LBP and is the most common condition keeping Australians aged 45–64 out of the workforce [3].
The nature of LBP is varied at each individual level and complex at systems level. At an individual level, it is hypothesized that pain is either generated via mechanical compression or chemical irritation via inflammatory biomarkers [4]. Nociceptive pain (NoP) results from activation of nociceptors that innervate ligaments, small joints, muscle tendons and other structures and may be a result of inflammatory response, mechanical compression, or autoimmune response [5,6,7]. Neuropathic pain (NeP) in the context of the lumbar spine is defined by the International Association for the Study of Pain as 'pain caused by a lesion or disease of the somatosensory nervous system' [8]. This can be a result of mechanical compression of radicular nerve tissues in the spinal canal or foramina or the action of inflammatory mediators that originate from the degenerated disk [5,6,7]. Different components of pain may individually or collectively contribute to the overall pain perception. Therefore, it is imperative to associate these pain classifications with underlying pathomechanisms able to be visibly observed on radiological imaging as the current understanding of the nature of LBP and leg pain is unclear, with high rates of degenerative changes visible on imaging in asymptomatic patients.
The PainDETECT questionnaire (supplementary 1) is a screening tool to discriminate between NeP and NoP on a 38 point scale, with 0–12 being negative of NeP, 12–18 being an ambiguous result and 19–38 being positive of NeP [9]. Validation was examined independently by pain specialists and revealed sensitivity, specificity, and positive predictive value all above 80% [10]. Although some authors have independently concluded the reliability of PainDETECT in distinguishing NeP [11], nonetheless, its use in the clinical setting is heavily debated with some studies showing PainDETECT to be a poor indicator of NeP [12]. To date, no studies have been conducted that analyze the association between the PainDETECT questionnaire and visible degenerative and stenosis findings observable on X-ray and MRI imaging in relation to the lumbar spine.
Current literature provides an uncertain conclusion in the relationship between radiological findings and pain related to the lumbar spine. Previous published studies exploring the use of PainDETECT in spinal clinical settings have yielded opposing conclusions. Therefore, it is interesting to ponder if there exists an association between pathological findings on radiology and NeP and NoP. This study aims to conduct an observational cohort study to explore the associations between radiological findings on MRI and X-ray and pain classifications assessed by the PainDETECT questionnaire to allow for an improved understanding into the pathophysiology and development of NoP and NeP.
Materials and methods
Study design and patient population
The study was IRB approved and conducted as a cohort study of adult patients (over 18 years of age) who presented to the Spine Service clinic at St George Private Hospital with completed PainDETECT questionnaire, MRI and/or X-ray scans. Written approval was granted by the original developer of the PainDETECT tool in an electronic format for the purpose of this study [9]. The most recent MRI and X-ray scans were used if multiple scans of the same patient were available in the database. All MRI scans, X-ray scans, radiology reports, demographic data and PainDETECT score were consecutively extracted. Patients were included if and only if they had chronic LBP (> 3 months) with or without leg pain. Patients were excluded if they had a history of lumbar spinal surgery prior to imaging and completing the PainDETECT form. Written informed consent was obtained from all patients to be included in the study.
Data collection
The standing lateral X-ray images, axial and sagittal T1W and T2W MRI scans of the lumbar spine were assessed, and data points were collected before reading the radiology report. SS was trained by an experienced spine surgeon and back pain researcher with extensive experience in interpreting radiological images (ADD). X-ray parameters for sagittal alignment are measured including cobb angle, sacral slope, pelvic tilt, and pelvic incidence. MRI degenerative and stenotic parameters include intervertebral disk (IVD) bulge, Pfirmann grade, endplate changes, high intensity zones (HIZ), spinal stenosis and foraminal stenosis. The specific measurement protocols of the degenerative/stenotic parameters are outlined in Table 1. In patients with multi-level degeneration, the parameter for the most severe level was extracted. The radiology reports were prepared by board certified radiologists.
Data points for ten percent of the patients were also measured by a second rater (ZG) to evaluate inter-rater reliability, and for a second time three weeks after initial extraction by the first author (SS) to evaluate intra-rater reliability. To enhance the quality and applicability of this study, each rater was blinded to their own measurements and findings of the other.
PainDETECT score and demographic data were extracted from the Spine Service PainDETECT RedCap database. PainDETECT was trichotomized into the NoP, ambiguous and NeP groups as defined by the PainDETECT questionnaire.
Statistical analysis
Kruskal–Wallis H test was used to analyze the difference in PainDETECT scores for more than two groups. A post-hoc Mann–Whitney U test using a Bonferroni correction was used to analyze the difference in PainDETECT scores between paired groups. Pearson Chi square was used to analyse the independence of association. One way analysis of variance (ANOVA) was used to analyze the difference in continuous variables for more than two groups. A subgroup analysis of patients with no foraminal and spinal stenosis was conducted to determine the relationship between Pfirmann grade and PainDETECT. Inter-rater reliability was assessed using the intraclass coefficient estimates (ICC) based on single-rating, consistency, 2-way random effects model, and intra-rater reliability was assessed using ICC based on single-rating, absolute agreement, 2-way fixed effects model. ICC values of < 0.05, 0.5–0.75, 0.75–0.90, and > 0.90 indicated poor, moderate, good, and excellent reliability, respectively. Statistical analyses were conducted using the commercially available software SPSS (version 27, IBM Corporation, New York, USA). The level of statistical significance was set at 5% (p = 0.05).
Results
Demographics
A flowchart depicting patient inclusion, exclusion, and separation into pain classification groups is shown in Fig. 1. Of the 279 patients included in the study, 102 had NoP, 78 had ambiguous and 99 had NeP based on the PainDETECT questionnaire scores. There was a statistically significant difference in mean age amongst the NoP, ambiguous, and NeP groups (58.21 \(\pm\) 17.11 vs. 55.33 \(\pm\) 16.45 vs. 53.63 \(\pm\) 15.62, p = 0.043). The NeP group had highest mean numerical rating scale (NRS), then the ambiguous group and finally NoP group (7.9 \(\pm\) 1.6 vs. 6.9 \(\pm\) 1.6 vs. 5.\(8\pm 2.2\), p < 0.001) (Table 2).
Flowchart Depicting the Inclusion of Participants in the Study
Flowchart representing the process of patient and exclusion of the study with the specific data on the number of patients included/excluded at each step. It also shows how the patients in the study were divided into nociceptive, ambiguous, and NeP categories as described by the PainDETECT questionnaire
Relationship between radiological changes and pain classification
Kruskal–Wallis test showed that the distribution of foraminal stenosis (H(2) = 12.742, p = 0.002), spinal stenosis (H(2) = 9.948, p = 0.007) and Pfirmann grade (H(2) = 6.823, p = 0.033) was significantly different across the trichotomized pain classifications of NoP, ambiguous and NeP. Post hoc Mann–Whitney U test revealed a higher foraminal stenosis severity (U = 18.962, p = 0.002), spinal stenosis severity (U = 14.481, p = 0.005) and Pfirmann grade (U = 14.221, p = 0.028) in the NeP group compared to the NoP group. With regards to all three MRI parameters there was no difference between the NeP and ambiguous group, and the ambiguous and NoP group (p > 0.05) (Table 3).
ANOVA showed no significant difference in pelvic tilt, sacral slope, pelvic incidence, and lumbar lordosis across NoP, ambiguous and NeP classifications. There was significantly higher number of NeP patients with intervertebral disk bulge compared to patients with NoP and ambiguous pain (96% vs. 78% vs. 78%, p = 0.002). There was significantly higher number of NeP patients with high intensity zones compared to patients with NoP and subsequently ambiguous pain (51% vs. 41% vs. 19%, p < 0.001). There was no association between pain classification and endplate changes (p = 0.776).
Subgroup analysis of patients without stenosis
Kruskal–Wallis test showed that the distribution of Pfirmann grade was significantly different across the trichotomized pain classifications of NoP, ambiguous and NeP in patients without foraminal or spinal stenosis (H(2) = 7.765, p = 0.021). Post hoc Mann–Whitney U test revealed a higher Pfirmann grade (U = 11.321, p = 0.020) in the NeP group compared to the NoP group. There was no difference between the NeP and ambiguous group, and the ambiguous and NoP group (p > 0.05).
Intra-rater and inter-rater reliability
The intra-rater reliability for all measurements methods of the lumbar degenerative parameters included in this study was good-to-excellent from 0.831 (0.670, 0.918) to 0.983 (0.189, 0.997) apart from IVD bulge which only had a moderate ICC of 0.738 (0.508, 0.870). The inter-rater reliability of the measurement methods was good-to-excellent from 0.752 (0.532, 0.877) to 0.979 (0.947, 0.991) (Table 4).
Discussion
This study used a cohort analysis to examine the association between PainDETECT score and the different classifications of NeP (PainDETECT > 18), ambiguous (12 < PainDETECT < 19) and NoP (PainDETECT < 13) pain as described by the PainDETECT Questionnaire [9]. Additionally, degenerative, and stenotic parameters were evaluated from MRI and gravity-loaded standing x-rays. Of the 279 patients included, 102 had predominantly NoP, 78 had ambiguous and 99 had predominantly NeP. The results showed that patients in the NoP group (58.21 \(\pm\) 17.11) had the highest mean age followed by ambiguous (55.33 \(\pm\) 16.45) and NeP group (53.63 \(\pm\) 15.62). This finding supports the postulation that NoP is associated with global degeneration of the lumbar spine, which is more prevalent in the older population, and that the putative pathological causes of NeP are less age dependent [18]. In this study patients in the NeP group had the highest NRS followed by the ambiguous and NoP group, which corresponds with previous literature that NeP LBP experience higher levels of pain, disability, and reduced quality of life [19].
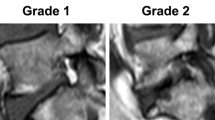
The mechanism of nervous system response to disk degeneration is heavily debated by researchers. Traditionally, studies have described the progression of low back pain as disk degeneration simulating the nociceptors of the anulus fibrosus (AF) resulting in NoP discogenic pain which progresses to NeP when degeneration leads to herniation causing pressure on the adjacent nervous tissue [20]. Interestingly, our study found that patients with NeP had a higher Pfirmann grade compared to patients with NoP pian, positing disk degeneration alone can cause NeP. Nerve endings of the sinuvertebral nerve in degenerated disks have been found to penetrate into the deeper layers of the AF and can even extend into the NP [21]. This is pathoanatomically correlated to discogenic LBP and under repetitive trauma and mechanical stress can result in chronic increase in levels of inflammatory mediators in the IVD. Miyagi et al. demonstrated increase in calcitonin gene-related peptide expression contributing to increase severity of pain and neuronal remodelling, ultimately aiding in the pathogenesis of NeP [22]. The finding of HIZs in patients with NeP also demonstrates the hyperinflammatory state of a degenerated disk. Our study found that there was a higher prevalence of HIZs in patients with NeP compared to NoP and ambiguous pain. HIZs have been proposed to be fluid filled zones in the AF as a result of an inflammatory process and is related to extradural inflammation [5]. Studies have linked upregulation of macrophages in HIZs which results in proliferation of tumour necrosis factor alpha (TNF-a), Interleukin 1 and prostaglandin E2, consequently, causing further neuronal damage and nerve regeneration [23]. Biomechanical and mechanical factors arising from a deteriorated disk influences the pain process of nerve roots and the dorsal root ganglia, ultimately aiding in NeP pathogenesis. Future studies should incorporate other measures of degeneration and inflammation including facet joint arthrosis and paraspinal muscle fat infiltration level.
Mechanically, a herniated disk represented as a disk bulge on MRI can cause compression resulting in spinal and foraminal stenosis. Spinal and foraminal canal narrowing results from degenerative changes of the spine, leading to compression or ischemia of the lumbosacral nerve roots. Consequently, morphological changes can occur to the patient’s nerve roots leading them to present clinically with NeP [24]. This pathway is supported by our study as patients in the NeP group had a higher severity of foraminal stenosis, spinal stenosis and prevalence of disk bulges compared to the NoP group. A positive relationship between PainDETECT score and foraminal and spinal stenosis severity was also observed in our study. However, acute mechanical nerve compression alone does not cause NeP but commonly leads to NoP lumbar radiculopathy, which is often resolved after a discectomy when the nerve is decompressed [25]. A large number of inflammatory and signalling pathways that play a role in NeP are hypothesized to be stimulated as a result of nerve damage. For example, TNF-alpha, a proinflammatory cytokine found in HIZs, is also upregulated in endoneurial macrophages and Schwann cells following neuronal injury which results in NeP pain [26]. Existing literature has identified variations in the manifestation of NeP. Injuries proximal to the dorsal root ganglia (DRG) have been associated with a higher incidence of chronic NeP compared to injuries distal to the DRG [27]. To enhance our understanding of NeP and its diverse presentations, future studies should consider incorporating comprehensive analyses of the extent, location, and duration of compression. By examining radiological findings in conjunction with these factors, researchers can gain valuable insights into the distinct manifestations of NeP among patients.
This study was the first to introduce sagittal alignment measurements on X-ray. None were significantly associated with NeP apart from pelvic incidence, which was shown to be negatively correlated with PainDETECT score. Studies have shown pelvic incidence to be lower in patients with spinal stenosis, foraminal stenosis and chronic LBP [28]. This demonstrates that neuropathia is possibly a local disk based segmental issue with no associations to sagittal imbalance and NoP is mechanical in origin.
Low back pain is complex and multi-faceted. We posit that whilst compression itself is associated with neuropathia it is not exclusive to NeP. Disk degeneration and the resulting proinflammatory state of the degenerated disk are also drivers of neuropathia. Ultimately, it is imperative for future longitudinal prospective studies to investigate the possibility of the existence of a causal pathological pathway between disk degeneration, stenosis, inflammation, and NeP pain, or if all are independent causes of NeP pain and exist simultaneously. This will allow surgeons to identify problematic patients and develop potential predictors for beneficial treatment outcomes, enhancing the customization of spinal treatments.
The results of this study were impacted by certain limitations. Firstly, the study did not have any follow ups, meaning the progression of patient’s clinical and radiological parameters were not analysed over time and compared between the pain groups. Socioeconomic measures and certain patient demographic information such as body mass index, smoking status etc. was not recorded. The population only included patients who presented to a tertiary spinal clinic, therefore, the prevalence of patients who have lower limb NeP pain without a spinal pathology is difficult to assess. To further explore the concept of NeP a prospective registry needs to be established to allow for longitudinal tracking of individual patients to determine whether radiological parameter can reliably predict NeP, based on the concordance between surgeon’s assessments and objective Pain DETECT scores alongside actual radiological findings. This will provide valuable insights into optimizing pain diagnosis and treatment strategies.
Conclusion
NeP group had lowest mean age and exhibited more severe pain levels compared to the NoP group. Positive correlations were observed between PainDETECT scores and foraminal stenosis, spinal stenosis, and Pfirmann grade. Conversely, a negative correlation was found between PainDETECT scores and pelvic incidence. Furthermore, the NeP group had significantly higher severity of foraminal stenosis, spinal stenosis, and Pfirrmann grade compared to the NoP group. Moreover, a higher proportion of NeP patients displayed intervertebral disk bulge and high-intensity zones compared to those with NoP and ambiguous pain. These results emphasize the multifactorial nature of NeP and highlight the importance of considering various pathological factors beyond neuronal compression in the evaluation and management of individuals with back and leg pain. Future research should continue to explore the complex interactions between these factors to gain a more comprehensive understanding of NeP and inform more targeted treatment approaches for individuals experiencing chronic LBP.
References
Knezevic NN, Candido KD, Vlaeyen J, Zundert J, Cohen S (2021) Low back pain: epidemiology, mechanisms, and treatment. Paper presented at the lancet-seminar series. https://doi.org/10.1016/s0140-6736(21)00733-9
Gore M, Sadosky A, Stacey BR, Tai K-S, Leslie D (2012) The burden of chronic low back pain: clinical comorbidities, treatment patterns, and health care costs in usual care settings. Spine 37(11):E668–E677. https://doi.org/10.1097/BRS.0b013e318241e5de
Arthritis and Osteoporosis Victoria (2013)A problem worth solving: the rising cost of musculoskeletal conditions in Australia: a report. Arthritis and Osteoporosis Victoria
Kawaguchi S, Yamashita T, Yokogushi K, Murakami T, Ohwada O, Sato N (2001) Immunophenotypic analysis of the inflammatory infiltrates in herniated intervertebral discs. Spine 26(11):1209–1214. https://doi.org/10.1097/00007632-200106010-00008
Saifuddin A, Mitchell R, Taylor B (1999) Extradural inflammation associated with annular tears: demonstration with gadolinium-enhanced lumbar spine MRI. Eur Spine J 8:34–39. https://doi.org/10.1007/s005860050124
Kang JD, Stefanovic-Racic M, McIntyre LA, Georgescu HI, Evans CH (1997) Toward a biochemical understanding of human intervertebral disc degeneration and herniation: contributions of nitric oxide, interleukins, prostaglandin E: 2: and matrix metalloproteinases. Spine 22(10):1065–1073. https://doi.org/10.1097/00007632-199705150-00003
Brisby H, Balagué F, Schafer D, Sheikhzadeh A, Lekman A, Nordin M, Rydevik B, Fredman P (2002) Glycosphingolipid antibodies in serum in patients with sciatica. Spine 27(4):380–386. https://doi.org/10.1097/00007632-200202150-00011
International Association for the Study of Pain (2017). IASP taxonomy. Pain terms. Neuropathic pain
Freynhagen R, Baron R, Gockel U, Tölle TR (2006) painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Curr Med Res Opin 22(10):1911–1920. https://doi.org/10.1185/030079906X132488
Freynhagen R, Tölle TR, Gockel U, Baron R (2016) The painDETECT project—far more than a screening tool on neuropathic pain. Curr Med Res Opin 32(6):1033–1057. https://doi.org/10.1185/03007995.2016.1157460
Cappelleri JC, Bienen EJ, Koduru V, Sadosky A (2014) Measurement properties of painDETECT by average pain severity. ClinicoEconomics Outcomes Res CEOR 6:497–504. https://doi.org/10.2147/CEOR.S68997
Hasvik E, Haugen AJ, Gjerstad J, Grøvle L (2018) Assessing neuropathic pain in patients with low back-related leg pain: comparing the painDETECT Questionnaire with the 2016 NeuPSIG grading system. Eur J Pain 22(6):1160–1169. https://doi.org/10.1002/ejp.1204
Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine 26(17):1873–1878. https://doi.org/10.1097/00007632-200109010-00011
Liu C, Cai HX, Zhang JF, Ma JJ, Lu YJ, Fan SW (2014) Quantitative estimation of the high-intensity zone in the lumbar spine: comparison between the symptomatic and asymptomatic population. Spine J Off J North Am Spine Soc 14(3):391–396. https://doi.org/10.1016/j.spinee.2013.06.078
Carragee EJ, Paragioudakis SJ, Khurana S (2000) Volvo award winner in clinical studies: lumbar high-intensity zone and discography in subjects without low back problems. Spine 25(23):2987–2992. https://doi.org/10.1097/00007632-200012010-00005
Lee GY, Lee JW, Choi HS, Oh KJ, Kang HS (2011) A new grading system of lumbar central canal stenosis on MRI: an easy and reliable method. Skeletal Radiol 40(8):1033–1039. https://doi.org/10.1007/s00256-011-1102-x
Lee S, Lee JW, Yeom JS, Kim KJ, Kim HJ, Chung SK, Kang HS (2010) A practical MRI grading system for lumbar foraminal stenosis. AJR Am J Roentgenol 194(4):1095–1098. https://doi.org/10.2214/AJR.09.2772
Kitab S, Habboub G, Abdulkareem SB, Alimidhatti MB, Benzel E (2019) Redefining lumbar spinal stenosis as a developmental syndrome: does age matter? J Neurosurg Spine 31(3):357–365. https://doi.org/10.3171/2019.2.SPINE181383
Beith ID, Kemp A, Kenyon J, Prout M, Chestnut TJ (2011) Identifying neuropathic back and leg pain: a cross-sectional study. Pain 152(7):1511–1516. https://doi.org/10.1016/j.pain.2011.02.033
Brisby H (2006) Pathology and possible mechanisms of nervous system response to disc degeneration. J Bone Joint Surg 88(Suppl 2):68–71. https://doi.org/10.2106/JBJS.E.01282
Freemont AJ, Peacock TE, Goupille P, Hoyland JA, O’Brien J, Jayson MI (1997) Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet 350(9072):178–181. https://doi.org/10.1016/s0140-6736(97)02135-1
Miyagi M, Ishikawa T, Kamoda H, Suzuki M, Murakami K, Shibayama M, Orita S, Eguchi Y, Arai G, Sakuma Y, Kubota G, Oikawa Y, Ozawa T, Aoki Y, Toyone T, Takahashi K, Inoue G, Kawakami M, Ohtori S (2012) ISSLS prize winner: disc dynamic compression in rats produces long-lasting increases in inflammatory mediators in discs and induces long-lasting nerve injury and regeneration of the afferent fibers innervating discs: a pathomechanism for chronic discogenic low back pain. Spine 37(21):1810–1818. https://doi.org/10.1097/BRS.0b013e31824ffac6
Takahashi H, Suguro T, Okazima Y, Motegi M, Okada Y, Kakiuchi T (1996) Inflammatory cytokines in the herniated disc of the lumbar spine. Spine 21(2):218–224. https://doi.org/10.1097/00007632-199601150-00011
Singh V, Montgomery SR, Aghdasi B, Inoue H, Wang JC, Daubs MD (2013) Factors affecting dynamic foraminal stenosis in the lumbar spine. Spine J Off J North Am Spine Soc 13(9):1080–1087. https://doi.org/10.1016/j.spinee.2013.03.041
Myers RR, Shubayev V, Campana WM (2001) Neuropathology of painful neuropathies. Pain Peripheral Nerve Disease 13:8–30. https://doi.org/10.1159/000061872
Wagner R, Myers RR (1996) Endoneurial injection of TNF-alpha produces neuropathic pain behaviors. NeuroReport 7(18):2897–2901. https://doi.org/10.1097/00001756-199611250-00018
Sekiguchi M, Sekiguchi Y, Konno S, Kobayashi H, Homma Y, Kikuchi S (2009) Comparison of neuropathic pain and neuronal apoptosis following nerve root or spinal nerve compression. Eur Spine J 18(12):1978–1985. https://doi.org/10.1007/s00586-009-1064-z
Chaléat-Valayer E, Mac-Thiong JM, Paquet J, Berthonnaud E, Siani F, Roussouly P (2011) Sagittal spino-pelvic alignment in chronic low back pain. Eur Spine J 20(Suppl 5):634–640. https://doi.org/10.1007/s00586-011-1931-2
Acknowledgements
Zac Gan for extracting the patient imaging parameters needed for inter-rater analysis.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was supported by a University Postgraduate Award from The University of New South Wales to SS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval
IRB approval was obtained from the Human Research Ethics Committee of the University of Wollongong (HREC No. 2020/329) and the University of New South Wales (HC210096) for retrospective collection of anonymized patient’s lumbar MRI scans, X-ray scans, radiology reports, and PainDETECT data from digital archives.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sima, S., Lapkin, S. & Diwan, A.D. In subjects with chronic low back pain, does neuropathia exclusively correlated to neuronal compression? A correlation study of PainDETECT questionnaire and corresponding MRI and X-ray findings. Eur Spine J 33, 1465–1473 (2024). https://doi.org/10.1007/s00586-024-08156-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-024-08156-2