Abstract
The structure and functionality of digestive system and gastrointestinal microbiota composition play a crucial role in the development of the gastrointestinal tract and gut morphology. A well-developed gastrointestinal tract will have a proper balanced proportion between the beneficial and harmful microbes; therefore, this balance will promote nutrient utilization and stimulate the development of immune system. The gut microbiota living in an animal’s gastrointestinal tract can be modulated through the feeding of prebiotic or probiotic compounds which have effects on the animal’s humoral immunity, hence improving the nutrient utilization and thus improving growth performance and the overall health condition of the animal. Therefore, this review aimed to provide a comprehensive understanding and recent reports on the role of insect meal in poultry feed aimed at improving health condition and gut status in poultry production. A literature search was performed with an electronic database of Directory of Open Access Journals (DOAJ), Research gate, Web of Science, Science Direct, Google Scholar, and PubMed. In addition, recently published manuscripts were selected, and the citations included in articles from the databases were used to search for other relevant articles. The studies were evaluated, and the level of evidence varied with insect types, inclusion levels, and the supplementing method. Extensive research has shown that insects can be safely used in poultry feeds to modulate the development of digestive and immune system without compromising the production parameters and the quality of the produce. Insects can be directly fed to chickens or can be processed into insect meals and supplemented in the diet. Nonetheless, most literatures done on the effects of insect meal are focused commercial chicken breeds and little attention is given to other poultry species; therefore, more studies are recommended to ascertain these findings.
Similar content being viewed by others
Introduction
Growth performance, reproduction traits, and health condition of chickens are affected by factors which are plane of nutrition, ambient temperatures, and diseases; however, the plane of nutrition and ambient temperature can be manipulated through proper management (Bedford 2000; Goodarzi et al. 2014). Pathogens are microorganisms that cause diseases in animals and the common ones include bacteria, viruses, and fungi which are everywhere in feeds, water, and the environment and hence difficult to mitigate (Bedford 2000). The pathogens interfere with the digestive functioning by causing inflammation in the gut and imbalance between the beneficial and harmful microbes and thus disturbing nutrient digestion and absorption as well as affecting the overall health condition of birds (Cross et al. 2007), and hence reducing growth and productivity of the chickens. This leads to the introduction of synthetic antibodies and the pathogens have become resistant to the synthetic antibodies (Nasir and Grashorn 2006). To counteract the risk of the emergence of drug-resistant microorganisms and drug residuals in chicken meat and meat products (Burgat 1999), attempts are being made to identify novel antimicrobial agents to improve chicken performance and overall health condition. The use of insects in poultry feeds is gaining popularity due to their high nutritional content, and feasibility to grow fast and production (Józefiak and Engberg 2015). Therefore, it is evident that the colony of microbiota living in the intestinal tract of chickens can be modulated through the feeding of prebiotic or probiotic compounds which in turn stimulate the development of humoral immunity (Gasco et al. 2018), hence improving the nutrient utilization and thus improving growth performance and the overall health condition of the animal.
Polysaccharides in chitin from the exoskeleton of insects have a positive effect on the functioning of the immune system (Food and Agriculture Organization 2017). Insects are regarded as a good source of protein with good amino acid composition and contain numerous bioactive compounds, chitin, antimicrobial, and lauric acid (Gasco et al. 2018), with proven antibacterial and immunomodulatory effects, hypolipidemic efficiency, and growth promoters (Hossain and Blair 2007; Bovera et al. 2015). Several studies have shown that among the potential insects, yellow mealworm (Tenebrio molitor), housefly (Musca domestica), black carp (Mylopharyngodon piceus), super mealworm (Zophobas morio), and black soldier fly (Hermetia illucens) have positive effects on the gut microbiota of the animals and could be used in poultry industry without compromising the production (Food and Agriculture Organization 2017; Gasco et al. 2018). Therefore, it is evident that polysaccharides in chitin in the exoskeleton of insects have a positive effect on the immune system functioning when fed to livestock (Food and Agriculture Organization 2017). The addition of black soldier flies into chickens’ diets may alter gastrointestinal tract through the alteration of gut microbiota (Detilleux et al. 2022). Biasato et al. (2018) investigated the effect of yellow mealworm on gut and microbiome modulation and observe no effect on gut morphometrics in free-range chickens. Gastrointestinal microbiota has a tremendous effect on the wellbeing of animals, and it is of great importance to manipulate the microbiota populations and improve the overall health condition in birds.
However, due to genetic improvements of chickens and everchanging climate conditions, insect inclusion levels for optimal performance change. Therefore, this review aimed to provide a comprehensive understanding of nutritional composition and bioactive compounds of potential feeder insects as feed additives and mechanisms in which insects modulate the overall health condition in birds as well as reports on the effects of using insects for gut health and status, immune system modulation, and overall growth performance in poultry as well as the recommended level and effective methods of supplementation.
Nutritional composition of potential insects
The nutritional values of insects are high enough to substitute many conventional protein sources such as soybean meal, fishmeal, or bonemeal (Govorushko 2019). Khusro et al. (2012) reviewed published literature on the nutritional composition of insect meals and observed that insects have high good quality nutrients such as protein, crude fat, crude fiber, minerals, and vitamins. Apart from having high nutritional content, insects are best known for their utilizable protein, lips, carbohydrates, vitamins, and minerals (Payne et al. 2016). The summary report on the global status of insects as food and feed source stated that most insects have a well-balanced nutrient content and good amino acid profile and are rich in micronutrients such as copper, iron, magnesium, and zinc (Govorushko 2019). Similarly, Nogales‐Mérida et al. (2019) referred to insect meal as a useful source of crude protein, crude fat, crude fiber, and fatty acids. Given the reasons mentioned above, insects appear to be one of the best-suited feedstuffs to address the issue of protein sources and certain health problems in poultry industries. The researchers are currently focusing on the larvae stage of yellow mealworm (Tenebrio molitor), black soldier fly (Hermetia illucens), the maggot, and pupae of the housefly (Musca domestica) as they are being regarded as a novel food due to their relatively high protein, vitamins, and mineral values and source of chitin compared other insect growing stages (Józefiak and Engberg 2015; Nowak et al. 2016; Zhao et al. 2016). Edible insects such as yellow mealworm (Tenebrio molitor) and black soldier are gaining popularities in animal food industries, used as a source of protein and chitin for growth promoter and immune-modulatory as well. Proximate analysis of raw larvae of these insects indicates that they contain protein content that varies between 40 and 60% (Józefiak and Engberg 2015), with an amino acid profile that is comparable to traditional protein sources (Finke 2002). Carbohydrates are contained in chitin, ranging from 5 to 20% of the dry weight (Chen et al. 2009). DiGiacomo and Leury (2019) investigated the nutritional composition of the black soldier fly and reported that raw larvae have about 20–44% dry matter content, protein content of 37–63%, and fat content ranging from 7 to 39%. The nutritional composition values depend on the life stage at which the insect is harvested, the rearing composition, and the type and quality of feeds ingested (Ghaly and Alkoaik 2009). For instance, larvae grown on pig manure result in higher protein content than that fed ruminant manure (DiGiacomo and Leury 2019); feeding insects spent grain will result in higher protein content and crude fat content (Oonincx et al. 2015). Furthermore, the nutrient content of most common insects is determined by the processing method, and thermal processing results in better nutrient content (Bordiean et al. 2020). Reports on the nutrition composition of common edible insects are summarized in Table 1.
The amino acid composition determines the protein quality and the nutritional values of the feedstuff (Ghaly and Alkoaik 2009). It is evident that insect meals are a good source of amino acids than most meal types and some of the essential amino acid content is higher than those of plant or animal origin (Nogales‐Mérida et al. 2019). Although insects are considered to contain all the essential amino acids, compared to fishmeal, yellow mealworm has lower methionine concentration which is one of the limiting amino acids in poultry (Józefiak and Engberg 2015). Reports on amino acids of insect meal are summarized in Table 1.
Bioactive compounds of insects
Besides being a good source of protein, insects have been validated to contain bioactive compounds with characteristics that could have potential health benefits and modulate the immune system (Roos and Van Huis 2017). The exoskeleton of insects contains chitin, antimicrobial peptides, and lauric acid compounds having a subsequent effect on the animal’s humoral immune when supplemented with diets (Montalban-Arques et al. 2015). Chitin is a bioactive compound found in the exoskeleton of crustaceans and insects (Finke 2007). Chitin contains hypolipidemic and hypercholesterolemic compounds which stimulate an innate immune response to various diseases in animals (Komi et al. 2018; Bovera et al. 2015). Among the livestock, neither poultry nor mammals synthesize chitin; therefore, insects are a potential target for chitin (Komi et al. 2018; Bovera et al. 2015). Insects have been categorized to have a tremendous diversity of bioactive peptides such as antimicrobial, antioxidant, and antidiabetic properties (de Silva Lucas et al. 2020), and recently, there are more than 150 insect proteins with antimicrobial properties which are classified as follows: α-helical peptides which include cecropin and moricin; cysteine-rich peptides such as insect defensin and drosomycin; proline-rich peptides which include apidaecin, drosocin, and lebocin; and glycine-rich proteins such as attacin and gloverin (Otvos 2000; Tonk and Vilcinskas 2017; Gasco et al. 2018; de Silva Lucas et al. 2020). According to Chernysh et al. (2015), insect antimicrobial peptides are effective in various bacteria with fewer chances for the bacteria to become resistant. These insect antimicrobial peptides have proven to be effective in pigs and broiler chickens in terms of growth traits and gut health through enhanced intestinal microbiota and immune function (Wang et al. 2016).
Insects’ mechanism/modes of action in promoting health and gut status of poultry
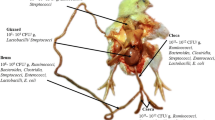
The larvae of insects have known to contain natural antibiotic, antipathogenic, and anti-inflammatory properties capable of modifying the microbial ecology in the gastrointestinal tract of the host animal and balancing the proportion between the beneficial and harmful microbe (Borrelli et al. 2017). Insects contain polysaccharides in the form of chitin, which is fermented by the microbiota in the large intestine balancing microbiota community as well as stimulating the development of immune system (Finke 2007). Maintained equilibrium between the host and gut microbial will benefit the host animal through modification of the development and function of the digestive and immune system (Pan and Yu 2014). Microbial modulates the digestive system through their positive effects on the development of villus height and crypt depth of the small intestine (Forder et al. 2007; Biasato et al. 2018). The villus height has direct positive effect on the rate of nutrient absorption in the small intestine (Montagne et al. 2003); therefore, it is evident that microbiota may stimulate mucin synthesis and modify its compositions (Forder et al. 2007). Mucin is a heavily glycosylated protein gel-like mucus layer produced in the epithelial tissues of the small intestines which serves as a medium in food digestion and absorption and defense against pathogens entering through the intestinal tract (Forder et al. 2007; Biasato et al. 2018). Microbiotas modulate mucin composition by colonizing the outer layer preventing pathogenic microorganism from penetrating into the intestinal epithelium (Pan and Yu 2014). Modes of action in which bioactive compounds promote and modulate health and gut status and immune system of poultry are presented in Fig. 1.
Bioactive compounds’ mode of action. Effects of bioactive compounds on gut health and status in birds (Abd El-Hack et al. 2022)
Reports on the effects of feeding insect-based diets on gut morphology of birds
The structure and functionality of gastrointestinal tract play a fundamental role in housing gut microbial population and balancing the proportion between the beneficial and harmful microorganisms colonizing the intestinal gut (Celi et al. 2017). The intestinal morphologies such as the length, muscle thickness, villus height, crypt depth, and villus height to crypt ratio are key factors contributing to intestinal development, health, and functionality, such inducing nutrient digestibility and absorption (Wang and Peng 2008; Lei et al. 2015). The height of villus is directly proportional to the rate of absorption and the ratio of villus height to crypt depth is an indicator of nutrient absorption in the small intestine (Montagne et al. 2003). Therefore, the ideal gut morphology is characterized by long villus and shallow crypts (Biasato et al. 2018). Several studies demonstrated that feeding insect meal can improve the structure and functionality of gastrointestinal tract in chickens and subsequently improved the health status (Wang et al. 2016; Borrelli et al. 2017; Kim et al. 2021). Polysaccharides in chitin from the exoskeleton of insects have a positive effect on the functioning of the immune system (Food and Agriculture Organization 2017). Kim et al. (2021) observed that the substitution of soybean meal with black soldier fly larvae meal (Hermetia illucens) improved intestinal morphology and enhanced nutrient absorption. Feeding insect meal, yellow mealworm in particular, has shown a positive effect villus height and crypt depth in Japanese quails (Zadeh et al. 2019). However, Sedgh-Gooya et al. (2021) reported no effect of yellow mealworm on gut morphology in broiler chickens throughout the experimental unit. Similarly, Biasato et al. (2018) observed that yellow meal supplementation in diets had no effect on gut morphometrics indices in free-range chickens. The same author suggested that yellow mealworm could be safely used in poultry diets to modulate the gut microbial ecology without compromising the gut morphology and mucin composition. Sedgh-Gooya et al. (2021) investigated the effect of replacing soybean and fishmeal using the levels 7.5, 15, 22.5, or 30 g per kg of diet on gut morphology of broiler chickens and reported no effect on gut morphology. Reports on the effect of supplementing insect meals in diets on gut morphology of poultry are represented in Table 2.
Role of insects on modulating gut microbiota composition of birds
Gut health is determined by the balance between the host, intestinal microbiota, and intestinal barrier to prevent harmful microbial (Biasato et al. 2018). Intestinal microbiota helps to digest low-quality feeds, thus improving nutrient utilization by the host animal and modulating the development and function of the digestive and immune system (Pan and Yu 2014). A proper proportion of beneficial and harmful microbiota modulates the growth performance and lowers the risk of infections (Józefiak et al. 2020). The gut microbiota living in an animal’s gastrointestinal tract can be modulated through the feeding of prebiotic or probiotic compounds which have effects on the animal’s humoral immunity (Gasco et al. 2018), hence improving the nutrient utilization and thus improving growth performance and the overall health condition of the animal. Several studies have shown that the potential use of insects in poultry diets has positive effects on the gut microbiota of the animals and could be used in the poultry industry without compromising production (Food and Agriculture Organization 2017; Gasco et al. 2018). Józefiak et al. (2020) reported that a small amount of insect meal in chickens’ diet could stimulate colonization of beneficial microbes and reduce the population of harmful bacteria in the gastrointestinal tract. Insect meal supplementation had increased the proportion between intestinal beneficial and harmful microbes which resulted in better nutrient utilization in chickens (Biasato et al. 2018). The same author reported an increased in the abundance of bacterium populations which are known to promote growth and intestinal villus structure, with anti-inflammatory effects, and control antipathogenic properties. The dietary treatment containing black soldier fly larvae meal increased rich bacterial community in laying hens; furthermore, these microbes are associated with a healthy status (Biasato et al. 2018). Colombino et al. (2021) insect larvae altered microbiota composition in the intestinal gut of broiler chickens. Little information has been reported on the role of insects in modulating the microbial ecology of the gastrointestinal tract of poultry. Findings on the effects of insects in the diet on microbial modulation of poultry are summarized in Table 3.
Effects of feeding insect-based diets on humoral immune response of birds
Among the livestock, neither poultry nor mammals synthesize chitin; therefore, insects are a potential target for chitin (Komi et al. 2018; Bovera et al. 2015). It has been shown that modulating intestinal microbial through feeding of insects may influence antibody titters in birds (Bovera et al. 2015; Ido et al. 2015). Feeding insect meals to livestock has positive effects on gastrointestinal microorganisms which plays a significant role in the body’s defense mechanisms through modulating the immune system and serving as a barrier to harmful microbes (Józefiak et al. 2020). The same author stated that the establishment of the proper balance between beneficial and harmful microorganisms is a crucial factor for health improvements and insect meal offers viable bioactive compounds which could modulate gastrointestinal microbial activity. Khempaka et al. (2011) reported that feeding broiler chickens shrimp chitin inhibited the growth of foodborne pathogens and Salmonella in the intestine. Supplementing yellow mealworm (Tenebrio molitor) and super mealworm (Zophobas morio) in the diets reduced cecal E. coli and Salmonella spp. in broiler chicks (Islam and Yang 2017). Bovera et al. (2015) reported lower albumin to globin ratio which induced immune response and better disease resistance in broiler chickens fed diets containing yellow mealworm. The same author suggested that these effects could be due to the probiotic effects of chitin. Similar findings were found for laying hens fed diets having black soldier fly meal (Marono et al. 2017). In the study investigating the effects of replacing soybean with super mealworm on intestinal health and blood profile of broiler chickens, Kim et al. (2021) concluded that black soldier fly meal can be safely used to replace soybean meal in broiler diets without any adverse effects on chicken health. Similarly, yellow mealworm could be supplemented to free-range chickens as mode of modulating digestive system and intestinal microbiota without having adverse effects on the gut health of the birds (Biasato et al. 2018). In another study by Borrelli et al. (2017), the dietary treatment consisting of yellow mealworm did not affect health status of laying hens. Feeding fish chitin had reduced pathogens’ growth by promoting the growth of beneficial intestinal microbiota with improved growth performance and health (Karlsen et al. 2017). Ido et al. (2015) reported a positive effect of housefly larval meal on the innate immune system activation against Edwardsiella tarda infection in red sea bream. The authors suggested that the immunomodulatory effects of insect-based diets could be linked to the presence of chitin in the exoskeleton of insects. Insect live larvae have no effects on mucin composition or immune response when fed to broiler chickens (Colombino et al. 2021). However, Mbhele et al. (2019) observed that black soldier fly larvae meal compromises autoimmunity in jumbo quails when included in higher levels. Reports on the effects of insect meal on humoral immune response of domesticated birds are summarized in Table 4.
Role of insects in promoting growth performance of poultry
The use of black soldier fly larvae in chicken feeds showed an improvement in terms of production performance, feed efficiency, and meat quality (DiGiacomo and Leury 2019). Studies by Maurer et al. (2016) and Nogales‐Mérida et al. (2019) indicated that insect meal can partially or completely replace fishmeal and soybean cake in the diet of fish or layers without affecting the production performance as well as the feed efficiency. There is evidence that the addition of yellow mealworm (Tenebrio molitor) in broilers’ diets improved the growth performance of broiler chickens during the starter period (Sedgh-Gooya et al. 2021). Adding a small amount of yellow mealworm or super mealworm into broilers’ diets can improve their growth performance traits (Benzertiha et al. 2020). These improvements could be due to increased daily feed intake which is be associated with the palatability of insect meal supplemented in the diet (Islam and Yang 2016). The addition of black soldier flies into chickens’ diets may influence their growth performance traits through the alteration of gut microbiota (Detilleux et al. 2022). Yellow mealworm supplementation in the diets could improve growth performance in growing Japanese quails (Zadeh et al. 2019). Ballitoc and Sun (2013) observed positive effects of supplementing yellow mealworm on overall feed intake and body weight gain in broiler chickens. However, Selaledi et al. (2020) stated that yellow mealworm supplementation in the growing diet of indigenous chickens could not be considered a potential protein source. Results of the effects of insect meal on the growth performance of chickens are summarized in Table 5.
Conclusion
Poultry gut health has been a focus of many researchers since it is regarded as a major factor that determines the growth performance and health condition of the animal. However, the gut health condition depends on the features such as intestinal microscopic structures, dietary compounds, and balance between beneficial and harmful microorganisms’ ratios and intestinal barrier. Intestinal microbiotas play a fundamental role in regulating metabolic processes of birds. Extensive research has shown that insects can be safely used in poultry feeds to modulate the development of digestive and immune system without compromising the production parameters and the quality of the produce. Edible insects are a good source of protein with good amino acid composition, and they have been regarded as a good source of antioxidants with antipathogenic properties and their results are two-fold. With these properties, insects are being targeted as an important future source of sustainable raw feed ingredients in the poultry industry, protein, and chitin in particular. Insects are promising feed additive for animals because they contain not only valuable nutrients but also bioactive compounds which are known to modulate the gut functionality as well as the gut microbiota, which could optimize the growth performance as well as the overall health condition of the birds. Insects can be directly fed to chickens or can be processed into insect meals and supplemented in the diet. Based on the recent literature, yellow mealworm and black soldier fly larvae are the most used insects in poultry production and insect meal is more effective than larvae direct feeding and could serve as a promising protein and chitin source. Therefore, the addition of insect meal in poultry diets could promote gut microbiota community, development of gut, and immune system, hence improving growth performances of birds. Nonetheless, most of literatures done on the effects of insect meal are focused commercial chicken breeds; hence, more studies on the use of insect meal in poultry are recommended to ascertain these findings especially with other poultry species.
Source of data
The data used in this review article was acquired from recently published manuscripts from different journals. Databases were accessed using electronic data sources such as Directory of Open Access Journals (DOAJ), Research gate, Web of Science, Science Direct, Google Scholar, and PubMed. In addition, the citations included in articles from the databases were used to search for other relevant articles. The keywords “Insect meal,” “Edible insects,” and “poultry” were used in the search engines.
References
Abd El-Hack ME, El-Saadony MT, Salem HM, El-Tahan AM, Soliman MM, Youssef GB, Taha AE, Soliman SM, Ahmed AE, El-Kott AF, Al Syaad KM (2022) Alternatives to antibiotics for organic poultry production: types, modes of action and impacts on bird's health and production. Poult Sci 101696
Altmann BA, Neumann C, Velten S, Liebert F, Mörlein D (2018) Meat quality derived from high inclusion of a micro-alga or insect meal as an alternative protein source in poultry diets: a pilot study. Foods 7(3):34
Ballitoc DA, Sun S (2013) Ground yellow mealworms (Tenebrio molitor L.) feed supplementation improves growth performance and carcass yield characteristics in broilers. Open Sci Repository Agric e23050425
Bedford M (2000) Removal of antibiotic growth promoters from poultry diets: implications and strategies to minimize subsequent problems. World Poult Sci 56(4):347–365
Benzertiha A, Kierończyk B, Kołodziejski P, Pruszyńska-Oszmałek E, Rawski M, Józefiak D, Józefiak A (2020) Tenebrio molitor and Zophobas morio full-fat meals as functional feed additives affect broiler chickens’ growth performance and immune system traits. Poult Sci 99(1):196–206
Biasato I, Ferrocino I, Biasibetti E, Grego E, Dabbou S, Sereno A, Gai F, Gasco L, Schiavone A, Cocolin L, Capucchio MT (2018) Modulation of intestinal microbiota, morphology and mucin composition by dietary insect meal inclusion in free-range chickens. BMC Vet Res 14(1):1–15
Bordiean A, Krzyżaniak M, Stolarski MJ, Czachorowski S, Peni D (2020) Will yellow mealworm become a source of safe proteins for Europe? Agriculture 10(6):233
Borrelli L, Coretti L, Dipineto L, Bovera F, Menna F, Chiariotti L, Nizza A, Lembo F, Fioretti A (2017) Insect-based diet, a promising nutritional source, modulates gut microbiota composition and SCFAs production in laying hens. Sci Rep 7(1):1–11
Bovera F, Piccolo G, Gasco L, Marono S, Loponte R, Vassalotti G, Mastellone V, Lombardi P, Attia YA, Nizza A (2015) Yellow mealworm larvae (Tenebrio molitor, L.) as a possible alternative to soybean meal in broiler diets. Br Poult Sci 56(5):569–575
Burgat V (1999) Residues of drugs of veterinary use in food. Rev Prat 41(11):985–990
Celi P, Cowieson AJ, Fru-Nji F, Steinert RE, Kluenter AM, Verlhac V (2017) Gastrointestinal functionality in animal nutrition and health: new opportunities for sustainable animal production. Anim Feed Sci Technol 234:88–100
Chen X, Feng Y, Chen Z (2009) Common edible insects and their utilization in China. Entomol Res 39(5):299–303
Chernysh S, Gordya N, Suborova T (2015) Insect antimicrobial peptide complexes prevent resistance development in bacteria. PLoS ONE 10:e0130788
Colombino E, Biasato I, Ferrocino I, Bellezza Oddon S, Caimi C, Gariglio M, Dabbou S, Caramori M, Battisti E, Zanet S, Ferroglio E (2021) Effect of insect live larvae as environmental enrichment on poultry gut health: gut mucin composition, microbiota and local immune response evaluation. Animals 11(10):2819
Cross DE, McDevitt RM, Hillman K, Acamovic T (2007) The effect of herbs and their associated essential oils on performance, dietary digestibility and gut microflora in chickens from 7 to 28 days of age. Br Poult Sci 48(4):496–506
da Silva Lucas AJ, de Oliveira LM, da Rocha M, Prentice C (2020) Edible insects: an alternative of nutritional, functional and bioactive compounds. Food Chem 311:126022
Detilleux J, Moula N, Dawans E, Taminiau B, Daube G, Leroy P (2022) A probabilistic structural equation model to evaluate links between gut microbiota and body weights of chicken fed or not fed insect larvae. Biology 11(3):357
DiGiacomo K, Leury BJ (2019) Insect meal: a future source of protein feed for pigs? Animal 13(12):3022–3030
Elahi U, Wang J, Ma YB, Wu SG, Wu J, Qi GH, Zhang HJ (2020) Evaluation of yellow mealworm meal as a protein feedstuff in the diet of broiler chicks. Animals 10(2):224
Finke M (2007) An estimate of chitin in raw whole insects. Zoo Biol 26:105–115
Finke MD (2002) Complete nutrient composition of commercially raised invertebrates used as food for insectivores. Zoo Biology: Published in Affiliation with the American Zoo and Aquarium Association 21(3):269–285
Finke MD (2013) Complete nutrient content of four species of feeder insects. Zoo Biol 32:27–36
Fontes TV, de Oliveira KRB, Gomes Almeida IL, Maria Orlando T, Rodrigues PB, Costa DVD (2019) Digestibility of insect meals for Nile tilapia fingerlings. Animals 9(4):181
Food and Agriculture Organization (FAO) (2017) The state of food and agriculture. leveraging food systems for inclusive rural transition. FAO, Rome, Italy. Available at: http://www.fao.org/3/a-I7658e.pdf
Forder RE, Howarth GS, Tivey DR, Hughes RJ (2007) Bacterial modulation of small intestinal goblet cells and mucin composition during early posthatch development of poultry. Poult Sci 86:2396–2403
Gasco L, Finke M, Van Huis A (2018) Can diets containing insects promote animal health? J Insects Food Feed 4(1):1–4
Ghaly AE, Alkoaik FN (2009) The yellow mealworm as a novel source of protein. Am J Agric Biol Sci 4(4):319–331
Goodarzi M, Nanekarani S, Landy N (2014) Effect of dietary supplementation with onion (Allium cepa L.) on performance, carcass traits and intestinal microflora composition in broiler chickens. Asian Pac J Trop Dis 4(1):297–301
Govorushko S (2019) Global status of insects as food and feed source: a review. Trends Food Sci Technol 91:436–445
Hossain SM, Blair R (2007) Chitin utilisation by broilers and its effect on body composition and blood metabolites. Br Poult Sci 48:33–38
Hussain I, Sarzamin K, Asad S, Naila C, Rafiullah K, Waqas A, Naseer A (2017) Mealworm (Tenebrio molitor) as potential alternative source of protein supplementation in broiler. Int J Biol Sci 10:255–262
Ido A, Iwai T, Ito K, Ohta T, Mizushige T, Kishida T, Miura C, Miura T (2015) Dietary effects of housefly (Musca domestica) (Diptera: Muscidae) pupae on the growth performance and the resistance against bacterial pathogen in red sea bream (Pagrus major) (Perciformes: Sparidae). Appl Entomol Zool 50:213–221
Islam MM, Yang CJ (2016) Efficacy of mealworm and supper mealworm larvae probiotics as an alternative to antibiotics challenged orally with Salmonella and E. coli infection in broiler chicks. Poult Sci 96:27–34
Islam MM, Yang C-J (2017) Efficacy of mealworm and super mealworm larvae probiotics as an alternative to antibiotics challenged orally with Salmonella and E. coli infection in broiler chicks. Poult Sci 96:27–34
Józefiak A, Benzertiha A, Kierończyk B, Łukomska A, Wesołowska I, Rawski M (2020) Improvement of cecal commensal microbiome following the insect additive into chicken diet. Animals 10(4):577
Józefiak D, Engberg RM (2015) Insects as poultry feed. In 20th European Symposium on poultry nutrition 24–27
Karlsen Ø, Amlund H, Berg A, Olsen RE (2017) The effect of dietary chitin on growth and nutrient digestibility in farmed Atlantic cod, Atlantic salmon and Atlantic halibut. Aquac Res 48:123–133
Khan S, Khan RU, Alam W, Sultan A (2017) Evaluating the nutritive profile of three insect meals and their effects to replace soya bean in broiler diet. J Anim Physiol Anim Nutr 102(2):662–668
Khempaka S, Chitsatchapong C, Molee W (2011) Effect of chitin and protein constituents in shrimp head meal on growth performance, nutrient digestibility, intestinal microbial populations, volatile fatty acids, and ammonia production in broilers. J Appl Poultry Res 20:1–11
Khusro M, Andrew NR, Nicholas A (2012) Insects as poultry feed: a scoping study for poultry production systems in Australia. Worlds Poult Sci J 68(3):435–446
Kim B, Bang HT, Jeong JY, Kim M, Kim KH, Chun JL, Ji SY (2021) Effects of dietary supplementation of black soldier fly (Hermetia illucens) larvae oil on broiler health. J Poult Sci 58(4):222–229
Komi EAD, Sharma L, Dela Cruz CS (2018) Chitin and its effects on inflammatory and immune responses. Clin Rev Allergy Immunol 54(2):213–223
Lei X, Piao X, Ru Y, Zhang H, Péron A, Zhang H (2015) Effect of Bacillus amyIoliquefaciens-based direct-fed microbial on performance, nutrient utilization, intestinal morphology, and caecal microflora in broiler chickens. Asian Australian J Anim Sci 28:239
Marono S, Loponte R, Lombardi P, Vassalotti G, Pero ME, Russo F, Gasco L, Parisi G, Piccolo G, Nizza S, Di Meo C, Attia YA, Bovera F (2017) Productive performance and blood profiles of laying hens fed Hermetia illucens larvae meal as total replacement of soybean meal from 24 to 45 weeks of age. Poult Sci 96:1783–1790
Maurer V, Holinger M, Amsler Z, Früh B, Wohlfahrt J, Stamer A, Leiber F (2016) Replacement of soybean cake by Hermetia illucens meal in diets for layers. J Insects Food Feed 2:83–90
Mbhele FG, Mnisi CM, Mlambo V (2019) A nutritional evaluation of insect meal as a Sustainable protein source for Jumbo quails: physiological and meat quality responses. Sustainability 11(23):6592
Montagne L, Pluske J, Hampson D (2003) A review of interactions between dietary fibre and the intestinal mucosa, and their consequences on digestive health in young non-ruminant animals. Anim Feed Sci Technol 108:95–117
Montalban-Arques A, De Schryver P, Bossier P, Gorkiewicz G, Mulero V, Gatlin DM III, Galindo-Villegas J (2015) Selective manipulation of the gut microbiota improves immune status in vertebrates. Front Immunol 6:512
Nasir Z, Grashorn MA (2006) Use of Black cumin (Nigella sativa Linn.) as alternative to antibiotics in poultry diets. 9th Tagung Schweine-und Geflügelernährung, Martin-Luther-Universität Halle-Wittenberg, Halle, Germany, 210–213
Nogales-Mérida S, Gobbi P, Józefiak D, Mazurkiewicz J, Dudek K, Rawski M, Kierończyk B, Józefiak A (2019) Insect meals in fish nutrition. Rev Aquac 11(4):1080–1103
Nowak V, Persijn D, Rittenschober D, Charrondiere UR (2016) Review of food composition data for edible insects. Food Chem 193:39–46
Oonincx DGAB, Van Huis A, Van Loon JJA (2015) Nutrient utilisation by black soldier flies fed with chicken, pig, or cow manure. J Insects Food Feed 1:131–139
Otvos L Jr (2000) Antibacterial peptides isolated from insects. J Pept Sci 6:497–511
Pan D, Yu Z (2014) Intestinal microbiome of poultry and its interaction with host and diet. Gut Microbes 5:108–119
Payne CLR, Scarborough P, Rayner M, Nonaka K (2016) A systematic review of nutrient composition data available for twelve commercially available edible insects, and comparison with reference value. Trends Food Sci Technol 47:69–77
Roos N, Van Huis A (2017) Consuming insects: are there health benefits? J Insects Food Feed 3(4):225–229
Sedgh-Gooya S, Torki M, Darbemamieh M, Khamisabadi H, Abdolmohamadi A (2021) Growth performance and intestinal morphometric features of broiler chickens fed on dietary inclusion of yellow mealworm (Tenebrio Molitor), Larvae Powder
Selaledi L, Mbajiorgu CA, Mabelebele M (2020) The use of yellow mealworm (T. molitor) as alternative source of protein in poultry diets: a review. Tropical animal health and production 52(1):7–16
Star L, Arsiwalla T, Molist F, Leushuis R, Dalim M, Paul A (2020) Gradual provision of live black soldier fly (Hermetia illucens) larvae to older laying hens: effect on production performance, egg quality, feather condition and behaviour. Animals 10(2):216
Tonk M, Vilcinskas A (2017) The medical potential of antimicrobial peptides from insects. Curr Top Med Chem 17:554–575
Wang JX, Peng KM (2008) Developmental morphology of the small intestine of African ostrich chicks. Poult Sci 87(12):2629–2635
Wang S, Zeng X, Yang Q, Qiao S (2016) Antimicrobial peptides as potential alternatives to antibiotics in food animal industry. Int J Mol Sci 17:603
Zadeh ZS, Kheiri F, Faghani M (2019) Use of yellow mealworm (Tenebrio molitor) as a protein source on growth performance, carcass traits, meat quality and intestinal morphology of Japanese quails (Coturnix japonica). Vet Anim Sci 8:100066
Zhao X, Vázquez-Gutiérrez JL, Johansson DP, Landberg R, Langton M (2016) Yellow mealworm protein for food purposes-extraction and functional properties. PLoS ONE 11(2):147791
Funding
Open access funding provided by University of South Africa.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not supported by any funding.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study requires no IRB/IACUC approval because there is no human and animal participants.
Informed consent
For this type of study, informed consent is not required.
Consent for publication
For this type of study, consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Malematja, E., Manyelo, T., Sebola, N. et al. The role of insects in promoting the health and gut status of poultry. Comp Clin Pathol 32, 501–513 (2023). https://doi.org/10.1007/s00580-023-03447-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-023-03447-4