Abstract
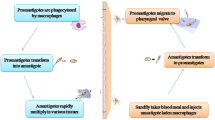
Anaplasma phagocytophilum is a tick-borne Gram-negative intracellular bacterium that could infect a few leukocytes of a large range of mammalian hosts together with humans. The clinical symptoms of the disease (granulocytic anaplasmosis) can vary from subclinical to fatal. The study aimed to detect A. phagocytophilum among camels (Camelus dromedarius) in Iran through two distinctive diagnostic techniques. One hundred twenty-five blood samples of male camels (C. dromedarius) were taken. Thin blood films were stained by Giemsa and DNA was extracted from whole blood for molecular examination. In this study, A. phagocytophilum was detected by microscopy and molecular examination among 125 male camels (C. dromedarius) in Yazd, central Iran. The A. phagocytophilum morula was observed in one sample out of 125 (0.8%) in microscopic examination and was detected in 4 samples out of 125 (3.2%) in the molecular examination. According to the results, A. phagocytophilum infects camels (C. dromedarius) consequently, camels can play a role in the infection of ticks, other animals, or humans. Many aspects of this disease continue to be set up by way of further studies in Iran.

Similar content being viewed by others
References
Ait Lbacha H, Zouagui Z, Alali S, Rhalem A, Petit E, Ducrotoy MJ, Boulouis HJ, Maillard R (2017) Candidatus anaplasma camelii” in one-humped camels (Camelus dromedarius) in Morocco: a novel and emerging Anaplasma species? Infect Dis Poverty 6:1–8. https://doi.org/10.1186/s40249-016-0216-8
Bahrami S, Hamidinejat H, Ganjali Tafreshi A (2018) First molecular detection of Anaplasma phagocytophilum in dromedaries (Camelus dromedarius). J Zoo Wildl Med 49:844–848
Bahrami S, Hamidinejat H, Haji Hajikolaei MR, Kavianifar S (2020) Concurrent occurrence of Anaplasma phagocytophilum and A. marginale in bovine peripheral blood samples from southwest of Iran. J Hellenic Vet Med Soc 71:2301–2308. https://doi.org/10.12681/jhvms.25076
Bastos ADS, Mohammed OB, Bennett NC, Petevinos C, Alagaili AN (2015) Molecular detection of novel Anaplasmataceae closely related to Anaplasma platys and Ehrlichia canis in the dromedary camel (Camelus dromedarius). Vet Microbiol 179:310–314. https://doi.org/10.1016/j.vetmic.2015.06.001
Brodie TA, Holmes PH, Urquhart GM (1986) Some aspects of tick-borne diseases of British sheep. Vet Rec 118:415–418. https://doi.org/10.1136/vr.118.15.415
de la Fuente J, Estrada-Peña A, Cabezas-Cruz A, Kocan KM (2016) Anaplasma phagocytophilum uses common strategies for infection of ticks and vertebrate hosts. Trends Microbiol 24:173–184. https://doi.org/10.1016/j.tim.2015.12.001
Dumler JS, Barbet AF, Bekker CP, Dasch GA, Palmer GH, Ray SC, Rikihisa Y, Rurangirwa FR (2001) Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia, and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int J Syst Evol Microbiol 51:2145–2165. https://doi.org/10.1099/00207713-51-6-2145
Dumler JS, Choi KS, Garcia-Garcia JC, Barat NS, Scorpio DG, Garyu JW, Grab DJ, Bakken JS (2005) Human granulocytic anaplasmosis and Anaplasma phagocytophilum. Emerg Infect Dis 11:1828–1834. https://doi.org/10.3201/eid1112.050898
El-Naga TRA, Barghash SM (2016) Blood parasites in camels (Camelus dromedarius) in Northern West Coast of Egypt. J Bacteriol Parasitol 7:258–265. https://doi.org/10.4172/2155-9597.1000258
Foggie A (1951) Studies on the infectious agent of tick-borne fever in sheep. J Pathol Bacteriol 63:1–15
Garcia-Perez AL, Baradika J, Oporto B, Povedano I, Juste RA (2003) Anaplasma phagocytophila as an abortifacient agent in sheep farms from Northern Spain. Ann NY Acad Sci 990:429–432
Granquist EG (2016) Anaplasma phagocytophilum in sheep. In: Rickettsiales: biology, molecular biology, epidemiology, and vaccine development. Springer, Switzerland
Granquist EG, Stuen S, Crosby L, Lundgren AM, Alleman AR, Barbet AF (2010) Variant-specific and diminishing immune responses towards the highly variable MSP2 (P44) outer membrane protein of Anaplasma phagocytophilum during persistent infection in lambs. Vet Immunol Immunopathol 133:117–124. https://doi.org/10.1016/j.vetimm.2009.07.009
Greene CE (2012) Infectious diseases of the dog and cat, 4th ed. Elsevier Saunders, Saint Louis, Missouri, USA
Hamidinejat H, Bahrami S, Mosalanejad B, Pahlavan S (2019) First molecular survey on Anaplasma phagocytophilum revealed high prevalence in rural dogs from Khuzestan province. Iran Iran J Parasitol 14:297–302
Matei IA, D’Amico G, Yao PK, Ionică AM, Kanyari PWN, Daskalaki AA, Dumitrache MO, Sandor MD, Gherman CM, Qablan M, Modry D, Mihalca AD (2016) Molecular detection of Anaplasma platys infection in free-roaming dogs and ticks from Kenya and Ivory Coast. Parasit Vector 9:157–165. https://doi.org/10.1186/s13071-016-1443-3
Moradi Z, Ebrahimzadeh E, Shayan P, Zarghami F (2021) Morphological and molecular investigation of Anaplasma infection in dromedary camel (Camelus dromedarius) in Bushehr province, Iran. Iran J Vet Med 15:295–300
Noaman V (2018) Molecular detection of novel genetic variants associated to Anaplasma ovis among dromedary camels in Iran. Arch Razi Institute 73(11–18):31
Noaman V, Shayan P (2009) Molecular detection of Anaplasma phagocytophilum in carrier cattle of Iran – first documented report. Iran J Microbiol 1:37–42
Parola P, Roux V, Camicas JL, Baradji I, Brouqui P, Raoult D (2000) Detection of Ehrlichiae in African ticks by polymerase chain reaction. Trans R Soc Trop Med Hyg 94:707–708. https://doi.org/10.1016/S0035-9203(00)90243-8
Rassouli M, Oliya Ardekani A, Jamshidian Mojaver M, Roozbeh M, Beikha M, Rasti Sania SE (2020) Molecular detection of Anaplasma platys among camels (Camelus dromedarius) in Yazd, Iran. Vet Parasitol Reg Stud Rep 22:100462. https://doi.org/10.1016/j.vprsr.2020.100462
Reinbold JB, Coetzee JF, Hollis LC, Nickell JS, Riegel CM, Christopher JA, Ganta RR (2010) Comparison of iatrogenic transmission of Anaplasma marginale in Holstein steers via needle and needle-free injection techniques. Am J Vet Res 71:1178–1188
Sazmand A, Harl J, Eigner B, Hodžić A, Beck R, Hekmatimoghaddam S, Mirzaei M, Fuehrer HP, Joachim A (2019) Vector-borne bacteria in blood of camels in Iran: new data and literature review. Comp Immunol Microbiol Infect Dis 65:48–53. https://doi.org/10.1016/j.cimid.2019.04.004
Selmi R, Ben Said M, Dhibi M, Ben Yahia H, Abdelaali H, Messadi L (2020) Genetic diversity of groEL and msp4 sequences of Anaplasma ovis infecting camels from Tunisia. Parasitol Int 74:101980. https://doi.org/10.1016/j.parint.2019.101980
Sharifiyazdi H, Jafari S, Ghane M, Nazifi S, Sanati A (2017) Molecular investigation of Anaplasma and Ehrlichia natural infections in the dromedary camel (Camelus dromedarius) in Iran. Comp Clin Pathol 26:99–103. https://doi.org/10.1007/s00580-016-2350-x
Stuen S (2003) Anaplasma phagocytophilum (formerly Ehrlichia phagocytophila) infection in sheep and wild ruminants in Norway. A study on clinical manifestation, distribution and persistence. Thesis Dr. Philosophiae, p 132
Stuen S, Bergstrom K, Palmer E (2002) Reduced weight gain due to subclinical Anaplasma phagocytophilum (formerly Ehrlichia phagocytophila) infection. Exp Appl Acarol 28:209–215
Stuen S, Bergström K, Petrovec M, Van de Pol I, Schouls LM (2003) Differences in clinical manifestations and hematological and serological responses after experimental infection with genetic variants of Anaplasma phagocytophilum in sheep. Clin Diagn Lab Immunol 10:692–695
Stuen S, Granquist EG, Silaghi C (2013) Anaplasma phagocytophilum, a widespread multi-host pathogen with highly adaptive strategies. Front Cell Infect Microbiol 3:31. https://doi.org/10.3389/fcimb.2013.00031
Woldehiwet Z (1987) The effects of tick-borne fever on some functions of polymorphonuclear cells of sheep. J Comp Pathol 97:481–485
Woldehiwet Z (2006) Anaplasma phagocytophilum in ruminants in Europe. Ann NY Acad Sci 1078:446–460. https://doi.org/10.1196/annals.1374.084
Woldehiwet Z (2007) Tick-borne diseases. In: Aitken ID (ed) Diseases of sheep, 4th edn. Blackwell publishing, Oxford, pp 347–355
Yousefi A, Chaechi Nosrati MR, Golmohammadi A, Azami S (2019) Molecular detection of Anaplasma phagocytophilum as a zoonotic agent in owned and stray dogs in Tehran, Iran. Arch Razi Inst 74:33–38. https://doi.org/10.22092/ari.2018.114893.1142
Yousefi A, Rahbari S, Shayan P, Sadeghi-dehkordi Z (2017) Molecular evidence of Anaplasma phagocytophilum: an emerging tick-borne pathogen in domesticated small ruminant of Iran; first report. Comp Clin Pathol 26:637–642. https://doi.org/10.1007/s00580-017-2429
Author information
Authors and Affiliations
Contributions
MR: supervision, writing, and editing; AOA: collecting samples; and HM, HR, and ZR doing experiments.
Corresponding author
Ethics declarations
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rassouli, M., Ardekani, A.O., Moazzezi, H. et al. Identification of Anaplasma phagocytophilum infection among camels (Camelus dromedarius) in Yazd, Iran. Comp Clin Pathol 31, 847–851 (2022). https://doi.org/10.1007/s00580-022-03388-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-022-03388-4