Abstract
Key message
Whereas cold temperatures and artificial smoke pollution (during World War II) cause negative pointer years in northern Fennoscandian downy birches, mass outbreaks of Epirrita autumnata L. and Operophtera brumata Bkh. are the strongest growth-influencing and -synchronizing factor.
Abstract
Variations in radial tree growth of downy birch can be affected by short-term environmental changes. Here we examine the tree-ring width of downy birch trees for signals caused by three different disturbance factors: climatic extremes, insect outbreaks, and artificial smoke employed to hide the German battleship Tirpitz during World War II at the Kåfjord in northern Norway. Besides growth/climate response analysis using gridded climate data and a tree ring width-chronology of the studied birch trees, we analyze missing rings, pointer years, and the ability of recovery after severe disturbances on an individual tree basis using the percent change of the trees’ basal area increment with respect to a reference period. The downy birches reveal (1) a significant positive correlation with May and June temperatures, (2) a high growth sensitivity to moth epidemics (Epirrita autumnata L. and Operophtera brumata Bkh.), and (3) a distinct growth deviation in the year of intense but short-term artificial smoke pollution. We conclude that downy birches are not exclusively sensitive to unusual cold temperatures, but short-term artificial pollution and insect outbreaks cause similar growth declines in the year of occurrence. Whereas for temperature and pollution, the growth response is less coherent among trees, and fades within 2 years, the insect outbreaks synchronize growth among all downy birches. Growth declines during years of mass insect outbreaks can further be amplified by cold May and June temperatures. Our findings thereby improve the understanding of competing disturbance factors on single birch trees but also on the growth of a whole site in Fennoscandia and facilitates the detection of disturbances in birch chronologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Disturbances are short-term, temporal variations of environmental conditions and force impulses in changes of the nature of forest ecosystems (Seidl et al. 2017; Dale et al. 2001). Factors such as classified in natural (e.g. orbital forcing, volcanic eruptions, drought, fire, frost, windstorms, landslides, animal attacks; Esper et al. 2012; Esper et al. 2015; Hartl-Meier et. al. 2014a; b; Attiwill 1994) and anthropogenic (e.g. settlement, harvesting, land use, pollution; Neumann 1991; Popradit et al. 2015; Hartl et al. 2019) can affect plant physiology (Attiwill 1994; Büntgen et al. 2009) which can have negative effects on tree growth and vitality or even lead to tree mortality due to their intensity (Seidl et al. 2014). However, the impacts of disturbance factors on forest dynamics do not only depend on the intensities of the factors but also on the characteristics of the tree’s site and, particularly, on the tree species (Kollas et al. 2013).
In our study, we examined trees of the species Betula pubescens Ehrh. (downy birch) at the latitudinal tree-line at the Kåfjord next to Alta in northern Norway. Downy birches are the predominant tree species in this region (Tømmervik et al. 2009). B. pubescens is a mid-sized, broadleaved tree species that occurs naturally all over Europe. Its ability of growing rapidly on well-drained soils as well as on poorly developed sites predestinate downy birch trees to be a pioneer tree species, even in boreal regions (Seppä et al. 2002; Beck et al. 2016). Thus, downy birches are well adapted to the environmental conditions at the latitudinal tree-line, but, nevertheless, there are also growth-limiting disturbances, especially for rapid changes in climate conditions (e.g. Opala et al. 2015; Levanic and Eggertsson 2008), moth outbreaks (e.g. Karlsen et al. 2013; Jepsen et al. 2011) and, particularly around the Kåfjord, short-term artificial pollution (Hartl et al. 2019).
At extreme sites like altitudinal or latitudinal tree-lines, tree species respond generally sensitively on changes of climate conditions (Körner 2012; Kullman and Öberg 2009). Whereas precipitation shortage can lead to a negative growth response at relatively dry sites (Beck et al. 2016; Lyu et al. 2019), the northern tree-line is typically controlled by temperature (Kullman 2003; Körner 2012). In a strict sense, long-term changes in temperature belong to climate variability and are no disturbances. However, especially exceptional, short-term changes in temperatures can lead to a disturbance of tree’s growth period. In fact, unusual cold temperatures at the beginning of the bud-burst period of B. pubescens in May can interrupt the tree’s unflowering and shift the onset of the growing season to a later time (Karlsson et al. 2003; Delpierre et al. 2018; Eckstein et al. 1991). Low temperatures in late summer can terminate the tree growth earlier as expected (Klaveness and Wielgolaski 1996). Both phenomena can shorten the time of growth severely and disturb the current tree’s growth and even the subsequent years that can lead, in serious cases, to a tree’s dieback (Karlsson et al. 2004; Eckstein et al. 1991). Mean July temperature with additional effects of June and/or August as expressed by JJA mean temperatures is a good indicator for the general growth conditions in northern Fennoscandia (Karlsson et al. 2004; Kirchhefer 2001; Esper et al. 2014).
The most common biological disturbance factors of birch trees in Fennoscandia that have severe negative impact on tree growth are insect outbreaks of autumnal moths (Epirrita autumnata Bkh.) and winter moths (Operophtera brumata L.) (Tenow 1972; Karlsen et al. 2013; Jepsen et al. 2008, 2011). The life cycle of the moths begins with the egg hatch between Mid-May and late June. Their larvae feed on expanding birch leaves (Fig. 1e) and can cause massive defoliation and diebacks of a whole site during vegetation period until the moth population collapses (Babst et al. 2010b; Beck et al. 2016). Moth outbreaks can stay over 2 years at the same site, but they occur most of the time synchronously in a 10-year cycle barring that the winter moth pests delay sometimes by 1–2 years. Thus, there is a negative, periodically occurring effect on growth of B. pubescens that is not only limited on the first year of disturbance event (Bylund 1999; Neuvonen et al. 1999; Hagen et al. 2007).

© Tirpitz Museum), and e a foliation feeding moth (here E. autumnata, © Photo: Moritz Klinghardt, Norwegian Institute for Nature Research)
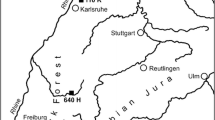
a Map of Fennoscandia showing the location of the study site (orange dot) in northern Norway. b The sample site is a pure birch site at the southeast coast of the Kåfjord. c Climate diagram from Alta Lufthavn considering data from 1964 to 2017. d Pollution through smoke screens hiding the battleship Tirpitz in 1944 (
Growth response of trees on long-term artificial air pollution was a topic for many studies of the last decades (e.g. Elling et al. 2009; Malik et al. 2012; Däßler and Lux 1984; Haselhoff and Lindau 1903). However, the kind of pollution at the Kåfjord is very unusual. The artificial pollutions, caused by the usage of smoke screens of the German battleship Tirpitz in 1944 (Fig. 1d), were locally restricted, short-term disturbances in the area around the Kåfjord (Hartl et al. 2019). The Tirpitz (the sister ship of the Bismarck) anchored at the fjord of Alta as a fleet-in-being to disturb allied convoys through the Arctic Ocean (Frère-Cook 1977). Recognized as a military threat, the allies tried to sink the vessel during six raids between April and September 1944. The German battleship protected itself against the aerial attacks with local ignited smoke consisting of sulfur trioxide, zinc/hexachloroethane, sulfuric and hydrochloric acid (Oberkommando der Kriegsmarine 1942). These artificial fogs covered the whole fjord of Alta and were very noxious for the German soldiers (H. Dv. 211/2 1939; H. Dv. 211/1 1940; H. Dv. 211/3 1941, Hampe 1963), furthermore for trees close to the anchor place of the Tirpitz (Hartl et al. 2019).
To analyze the growth responses of the birch trees to disturbances of extreme climate conditions, moth outbreaks and artificial pollution back in time, an analysis of tree rings is an appropriate method due to providing precise detection of annual resolved growth reactions with the opportunity of forest dynamics reconstructions in decadal to even millennial scales (Trotsiuk et al. 2018; Fritts 1976; Fritts and Swetnam 1989). An analysis of birch tree rings is challenging due to the non-straight stem growths combined with a more frequent appearance of missing rings than in many other tree species (Kirchhefer 1996). This growth sensitivity can be traced back to the way B. pubescens cope with (mechanical) disturbances. Mechanical disturbances influence the anatomical structure negatively, so the xylem-specific hydraulic conductivity decreases; the according tree ring will be narrow. Thus, the inter-annual tree-ring width (TRW) variability is depending on the intensity of the disturbance on single tree level (Tumajer and Treml 2019). To consider this behavior, we will investigate the growth response to varying disturbance factors by analyzing growth/climate response using climate data and TRW but also by analyzing growth variability around the years of the different disturbances, i.e. the basal area increment (BAI) of single trees to detect growth decline and trees’ recovery ability (Zang et al. 2014). Finally, we rank the disturbance factors by their harmfulness for B. pubescens at our site.
We assume that (1) the TRW at the latitudinal tree-line of northern Norway will show a correlation to exceptional negative climate conditions that will be visible as a decline of single tree growth and a low growth variability throughout the site in the rings of the current and following years; (2) the most severe moth outbreaks recorded for Alta affected growth of all trees negatively, so that the growth decline for all the tree will be similar, the growth variability will be low. In addition, we expect that the growth decline after moth outbreaks was stronger and the time for recovery took longer than after climate extremes. For the disturbance due to (3) artificial pollution, we hypothesize that there could be a pronounced single tree response in 1944 due to probably different local exposure to the artificial smoke. The results will show a pattern of growth variability in the year of disturbance and in the aftermaths that is not comparable to the two other disturbances due to the local emission of pollution.
Materials and methods
Study site and tree-ring data
In August 2017, 36 downy birch trees were sampled at the Kåfjord close to Alta in northern Norway (69.939° N 23.063° E, Fig. 1). The birches root in a poorly developed soil on a sandy and gravelly glacier moraine at ~ 10 m asl (Geological Survey of Norway 2019). Climatic conditions are characterized by snowy and cold winters, and cool summers (Kottek et al. 2006) including 412 mm precipitation and annual mean temperatures of 1.7 °C.
Each tree was cored twice at breast height (~ 1.30 m) with a 5 mm increment borer. The preparation for TRW measurements included cutting the 64 cores with a microtome (Gärtner and Nievergelt 2010) and staining them with Astra blue to improve the contrast of tree-ring boundaries (Rothe and Hartl-Meier 2014). The samples were measured at a resolution of 10 μm using a LINTAB measurement device and the software TSAPWin (both Rinntech 2011). Missing rings occurred relatively frequently and were set to a minimum thickness of 10 µm to support later data analysis. Cross-dating accuracy was checked visually and statistically using COFECHA (Holmes 1983).
The TRW data were power transformed to reduce heteroscedasticity within the chronology, detrended and standardized using cubic smoothing splines with a 50% frequency cut-off at 30 years to remove age-related growth trends. By this procedure, dimensionless ring width indices (RWI) were calculated in ARSTAN (Cook and Peters 1981; Cook 1983). A mean RWI chronology was produced by averaging the detrended single series using a robust mean (Mosteller and Tukey 1977). As we were also interested in actual growth, we also transformed the raw ring-width series into BAI, the annual ring area increase, based on the ring circumference approximated by a circle (Biondi and Qeadan 2008).
To estimate the internal coherence of the chronology, inter-series correlations (Rbar) were calculated over 30 years windows, lagged by 15 years (Table 1; Wigley et al. 1984). The RWI series were finally truncated at the year 1901 in line with the first year of gridded climate data and after 2000 due to the very high numbers of missing rings hindering an accurate dating (see changing Rbar values in Table 1). Other descriptive statistics for the chronology include the mean series length (MSL), mean sensitivity (year-to-year variability in tree ring width), average growth rate (AGR), and mean BAI, all provided in Table 1.
Statistical analysis
The estimation of the influence of the climate elements temperature and precipitation on birch growth at the Kåfjord was calculated with bootstrapped correlations using the treeclim package in R (Zang and Biondi 2015) with the RWI chronology and monthly climate data from the KNMI Climate Explorer (KNMI 2020). We considered previous-year June to current-year December. As the local climate station Alta Lufthavn data covers only the period 1964–2018 but the gridded CRU TS 4.01 data correlates with r = 0.99 for temperature and 0.57 for precipitation, we used the grid point 70.25° N/22.75° E and the period 1901–2000 for further analyses (University of East Anglia Climatic Research Unit, Harris, Jones 2017).
To assess the growth responses of the Norwegian birch trees to different disturbance factors, we considered cold extremes, severe moth outbreaks and pollution as event years (see Fig. 3). Based on the growth/climate response analyses, we derived May/June (MJ) temperatures are most influential for birch growth. Therefore, we selected the five coldest MJ temperatures as cold extremes that are below the 1.96 * standard deviation of the reference period’s (1961–1990) temperature anomalies mean for the twentieth century (see Fig. S1), namely 1902, 1909, 1915, 1951 and 1955. Regional moth outbreaks for northern Norway were recorded in several studies (Fig. 3, green shadings) (e.g. Tenow 1972; Tømmervik et al. 2004; Karlsson et al. 2004; Hagen et al. 2007; Jepsen et al. 2008; Babst et al. 2010a; Karlsen et al. 2013). According to the area around Alta, the most severe moth outbreaks occurred most likely in 1955, 1962, 1975, 1982 and 1989 (Kirchhefer 1996) so that we selected these years (Fig. 1, green lines). For the growth response to pollution, we considered the multiple smoke screen events in 1944 hiding the Tirpitz. To analyze the disturbance factors, we calculated percentage growth changes by dividing the BAI of each single series by the mean BAI of the five preceding years (Hartl et al. 2019). Afterwards, we used the superposed epoch analysis (SEA) (Panofsky and Brier 1958) with the individual BAI series to rank the disturbances. In this study, the time span of 5 years before to 2 years after the particular disturbance were examined to visualize the growth change around the years of disturbance. The variance in BAI were stated as percentual growth changes with respect to the mean of the 5 years preceding disturbance events (− 5 to − 1).
Results
The growth/climate response analysis reveals temperature to be important for tree growth, whereas precipitation correlates insignificantly (Fig. 2). Birch growth is sensitive to early season temperatures including current-year May and June. The seasonal MJ mean temperatures correlate at r1901–2000 = 0.33 (p ≤ 0.05) with the birch chronology.
The occurrence of disturbance factors is shown in Fig. 3 together with the RWI chronology and missing ring frequency. Thereby, it is obvious that cold events can appear inconspicuous and do not necessarily cause RWI minima, except for 1902 and 1955. In 1902, there is a growth decline followed by a RWI minimum and a high percentage of absent rings (14%) in the subsequent year 1903. In 1955, the cold temperatures correspond with minimum growth indicated by the RWI chronology.
Characteristics of the birch chronology and temporal distribution of disturbance events. The upper histogram shows the percentage number of missing rings (in grey), the black curve in the middle is the detrended RWI chronology, and the lower grey curve shows the chronology replication, i.e. number of tree-ring series. The blue vertical lines mark the 5 years with the lowest May/June monthly mean temperatures, and the orange line indicates the pollution event with the smoke screen actions of the German Kriegsmarine for hiding the battleship Tirpitz. Green shadings highlight possible moth outbreaks, and the green lines indicate the five most severe outbreaks in the study region
In 1944, the year of the battleship Tirpitz smoke screen incidents, growth rates were relatively low, though not conspicuously compared to other years. However, 8% of the trees already showed an absent ring (Fig. 3, orange line), and these values increased in 1945 to the highest number of missing rings recorded throughout the twentieth century (14 of all 101 absent rings, ~ 22% of the trees). Most of the other growth minima are connected to insect outbreaks, particularly during the more recent decades, including the five most severe outbreaks in 1955, 1962, 1975, 1982 and 1989 (green lines and shadings in Fig. 3) which were all accompanied by missing rings. High numbers of absent rings are also detected in 1975 (~ 16%) and in 1955 (~ 14%).
The single tree growth responses to varying disturbance factors differ substantially (Figs. 4; S2). In 1951, a year of unusual cold MJ temperatures, more than 75% of the trees showed a negative growth change and 50% of the trees showed a growth decline > 25%. The other 3 years of extreme cold MJ temperatures (1909, 1915, 1902) are characterized by a wide spread of growth responses among trees. In 1909, 75% of the trees showed a growth increase compared to the five preceding years. In 1902 as well as in 1915, birch growth is ordinary without any extreme deviations. However, after the year with the coldest MJ temperatures, i.e. in 1903, 86% of the trees show a growth decline followed by a negative growth response of only 38% in the subsequent year.
Percentage growth change of single trees to different disturbance events. The extreme events for temperature (blue) and moth outbreaks (green) are sorted with increasing intensity from upper to lower panels. The growth change for the event year in 1955, a combination of low May/June temperatures and a moth outbreak (green/blue) and the 1944 smoke screen event are shown in the bottom panel. The grey background represents the respective five-year reference period preceding the single events
In 1944, the year of the Tirpitz concealing tasks, the trees reacted differently. Whereas the growth response in the event year is similar to the cold year 1951, with three-quarter of the trees showing a growth decline and 50% of the trees having a growth reduction of more than 25%, in the subsequent year, three-quarter of the trees react again with a growth decline and the median growth reduction is even stronger (~ 45%). In 1946, > 50% of the trees started their recovery while the growth variability was still high (interquartile range of > 89%) and the other trees showed a negative growth response.
The growth response to severe moth outbreaks is less variable, compared to the responses to cold extremes and artificial fog events, as almost every tree reacted with a decline. In 1962, more than 50% of the trees reduced their growth by > 17% in comparison to the 5 years preceding the event. In the years with heavier outbreaks the growth response is even worse, e.g. median growth declines > 54% in 1975 and 1982, and > 57% in 1989. Importantly, the recovery of almost every tree started immediately after the event year while the growth variability between the individual trees increased.
The persistence of growth reductions increases with the intensity of the recorded moth outbreaks, as seen in Fig. 4. After 1962 and 1975, the trees needed only 1 year for (almost) recovery, after 1982 > 50% of the trees and 1989 > 75% of the trees needed 2 years to reach the pre-disturbance growth level. The year 1955 is outstanding, however, as very cold MJ temperatures co-occurred with a grave moth epidemic, resulting in severe growth reductions of all trees by 33% or more, and half of the birches showing decreases > 76% with respect to the five preceding years. In 1956, still 75% of the trees showed a negative growth response and > 50% of the trees did not recover until 1957 2 years after the event.
The SEA juxtaposes the median growth change behavior of the birch trees to different types of disturbance factors (Fig. 5). The years of low MJ temperatures did not cause any noticeable growth change, but growth even increased in the two following years. In contrast, the pollution events in 1944 caused a growth decrease of 28%, but in contrast to the other disturbance factors, the growth decline was even greater in the following year. Finally, there is a distinct growth response in years with severe moth outbreaks including a median growth reduction of 44%. This negative response was not long lasting as the trees showed a 12% growth increase in the year after the mass outbreak. The combination of moth outbreaks and very low MJ temperatures, as seen in 1955, was most harmful, caused a growth decline > 60%, and trees needed 2 years and more to fully recovery.
Percentage growth change to varying disturbance factors combined in a superposed epoch analysis. The percentage growth changes refer to the 5 years preceding the event years represented by the dashed line. Blue indicates the response to cold May/June-temperatures, green to insect outbreaks, blue/green to a combination of low May/June temperatures and insect epidemics (1955) and orange to pollution through smoke screen actions in 1944
Discussion
A detailed analysis of birch growth responses from the Kåfjord in northern Norway revealed varying sensitivities to disturbance factors including unusual cold mean MJ temperatures, foliage feeding moth outbreaks, and artificial pollution. Growth/climate response analysis showed the sensitivity of downy birch to early season temperatures as we found significant positive correlation with mean MJ temperatures. This association suggests a connection of the onset of radial growth with the beginning of budburst, triggered by MJ temperatures as reported by Klaveness and Wiegolaski (1996), Siljamo et al. (2008) and Karlsen et al. (2009). Accordingly, low mean MJ temperatures can cause a late onset of cambial activity or at least slow down the speed of cambial cell productions and xylem expansions triggering reduced growth rates during such “event” years (Ren et al. 2019). A good example is 1951, during which low growth rates of 75% of the trees likely represents a short growing season. However, we also found highly variable growth responses during most cold event years. This reflects the fact that the trees’ foliation and the resulting ability to do photosynthesis are not necessarily affected by low MJ temperatures, the influence of beneficial environmental conditions during their later growth period can balance the disturbance. Thus, the growth responses were heterogenous among trees, regardless of the intensity of the cold years.
The chemical impact of the Tirpitz’ smoke screens in 1944 was more severe. The artificial fogs caused a growth decline for more than 50% of the trees within the growth period whereas the variability of growth responses in the subsequent year 1945 was very high combined with the highest number of missing rings within the studied time span. The high quantity of absent rings are evidences that the downy birch canopies could (partly) be defoliated by the artificial fogs which took place within three months of the growth period (Hartl et al. 2019). Defoliation of birches reduces the photosynthesis rates massively and confines radial growth accordingly (Treshow and Anderson 1991; Hoogesteger and Karlsson 1992). A study by Haselhoff and Lindau (1903) supports this suggestion: They detail deciduous tree’s ability of enfolding new leaves to ease the impact of acid during the same vegetation period, compared to evergreens (see also Hartl et al. 2019). The high growth variability in the subsequent year 1945 suggests that the impact of smoke screens might have varied among the individual trees, i.e. some of the trees were still harmed in 1945 due to high (multiple) defoliation rates whereas others already recovered. In general, these artificial pollutions as a disturbance had the strongest effects on growth of single trees with respect to aftermaths.
Our results also reveal that moth outbreaks have serious effects on the tree growth. In the 5 years of most severe epidemics, more than 75% of the trees showed distinct growth declines. Similar to pollution effects, the growth decline can be explained by defoliation and reduced photosynthesis. The defoliation of birches in consequence of massive moth outbreaks is common in northern Scandinavia (Eckstein et al. 1991; Karlsson et al. 2004; Jepsen et al. 2008; Babst et al. 2010a). Even the high numbers of missing rings, such as in 1955, can be related to a well-known outbreak of E. autumnata in this area (Tenow 1972; Eckstein et al. 1991; Kirchhefer 1996; Karlsson et al. 2004).
Generally, the moth epidemics are the dominating disturbance factor at our study site: The more severe the intensity of moth outbreaks, the greater the growth decline but also the higher the growth synchronization within the studied area. However, the most severe impact on birch growth was not caused by a single disturbance factor, but by the combination of two: In 1955, mean MJ temperatures were low and likely shifted the onset of the growing season towards June, synchronous to the beginning of the egg hatch of birch moths. Tenow (1972) and Beck et al. (2016) concluded that a temporal synchronization of egg hatch and birch foliation supports larvae feeding and sets the foundation for a severe defoliation event at the site-to-regional scale. In severe cases, this can lead to the entire die-off of a birch site. The co-occurring disturbances in 1955 resulted in the strongest growth decline in our studied birch population More than 50% of the trees still suffered in 1956 and it took the trees 2 years to recover. This conclusion is supported by Kirchhefer (1996), who recorded a moth epidemic in 1955 as the most severe outbreak in Alta region. Tenow (1972) even stated that the intensity of this moth outbreak was exceptional for the whole of Fennoscandia within the last century.
Conclusions
In this study, we focused on single tree growth responses of B. pubescens in years of different disturbance factors in the Kåfjord in northern Norway. Birch tree ring growth from 1901 to 2000 is affected by three main factors: low MJ temperature, (artificial) chemical impacts and moth outbreaks. The growth responses do, however, not depend on the severity of low MJ temperatures. We find a close association between the chemical impact of the Tirpitz concealing tasks in 1944 and a growth decline for three quarter of the birch trees with a strong effect on the cambial activity of the following year. This combination of high growth variability and the highest number of missing rings leads to the fact that some trees were recovering fast while other trees were still suffering. In contrast, severe moth outbreaks of E. autumnata and O. brumata have similar effects on the growth of the entire site. The results demonstrate a positive association between the intensity of documented epidemics and strong growth declines. However, strongest growth declines are recorded if both cold MJ temperatures and moth outbreaks co-occur such as in 1955. A synergy of disturbance factors leads to a synchronization of the birch foliation in the early growing season with the moth’s egg hatch. Considering all disturbances, we are able to rank the factors by their growth impact: Cold MJ temperatures have the least influence on birch growth, followed by the artificial smoke in 1944, and moth outbreaks. The most severe disturbance factor is a combination of low monthly MJ temperatures and moth outbreaks.
Change history
07 September 2021
A Correction to this paper has been published: https://doi.org/10.1007/s00468-021-02175-y
References
Attiwill PM (1994) Ecological disturbance and the conservative management of eucalypt forests in Australia. For Ecol Manage 63:301–346
Babst F, Esper J, Parlow E (2010a) Landsat TM/ETM+ and tree-ring based assessment of spatiotemporal patterns of the autumnal moth (Epirrita autumnata) in northernmost Fennoscandia. Remote Sens Environ 114:637–646. https://doi.org/10.1016/j.rse.2009.11.005
Babst F, Frank DC, Parlow E, Esper J (2010b) Age and susceptibility of Fennoscandian mountain birch (Betula pubescens) towards insect outbreaks. In: Levanic T et al. (Eds.) Tree rings in archaeology, climatology and ecology. TRACE 8:26–32
Beck P, Caudullo G, De Rigo D, Tinner W (2016) Betula pendula, Betula pubescens and other birches in Europe: distribution, habitat, usage and threats. In: San-Miguel-Ayanz J, De Rigo D, Caudullo G, Houston Durrant T, Mauri (ed) European Atlas of Forest Tree Species. Publication Office of the European Union, Luxembourg, pp 70–73
Biondi F, Qeadan F (2008) A theory-driven approach to tree-ring standardization: defining the biological trend from expected basal area increment. Tree-Ring Bull 64:81–96. https://doi.org/10.3959/2008-6.1
Büntgen U, Frank DC, Liebhold A, Johnson D, Carrer M, Urbinati C, Grabner M, Nicolussi K, Levanic T, Esper J (2009) Three centuries of Alpine-wide insect outbreaks inferred from tree rings. New Phytol 182:929–941. https://doi.org/10.1111/j.1469-8137.2009.02825.x
Bylund H (1999) Climate and the population dynamics of two insect outbreak species in the North. Ecol Bull 47:54–62
Cook ER (1983) A time series analysis approach to tree ring standardization. Dissertation, University of Arizonahttps://ltrr.arizona.edu/sites/ltrr.arizona.edu/files/bibliodocs/CookER-Dissertation.pdf. Accessed 12 March 2020
Cook ER, Peters K (1981) The smoothing spline: a new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree-Ring Bull 41:45–55
Dale VH, Joyce LA, McNulty S, Neilson RP, Ayres MP, Flannigan MD, Hanson PJ, Irland LC, Lugo AE, Peterson CJ, Simberloff D, Swanson FJ, Stocks BJ, Wotton BM (2001) Climate change and forest disturbances: climate change can affect forests by altering the frequency, intensity, duration, and timing of fire, drought, introduced species, insect and pathogen outbreaks, hurricanes, windstorms, ice storms, or landslides. Bioscience 51:723–734. https://doi.org/10.1641/0006-3568(2001)051[0723:CCAFD]2.0.CO;2
Däßler HG, Lux H (1984) Zur Problematik der “Sauren Niederschläge” und ihres Einflusses auf den Nadelwald. Hercynia N F 21:73–83
Delpierre N, Lireux S, Hartig F et al (2018) Chilling and forcing temperatures interact to predict the onset of wood formation in Northern Hemisphere conifers. Glob Change Biol 25:1089–1105. https://doi.org/10.1111/gcb.14539
Eckstein D, Hoogesteger J, Holmes R (1991) Insect-related differences in growth of Birch and Pine at Northern treeline in Swedish Lapland. Holarctic Ecol 14:18–23
Elling W, Dittmar C, Pfaffelmoser K, Rötzer T (2009) Dendroecological assessment of the complex causes of decline and recovery of the growth of silver fir (Abies alba Mill.) in Southern Germany. For Ecol Manage 257:1175–1187. https://doi.org/10.1016/j.foreco.2008.10.014
Esper J, Frank DC, Timonen M, Zorita E, Wilson RJS, Luterbacher J, Holzkämper S, Fischer N, Wagner S, Nievergelt D, Verstege A, Büntgen U (2012) Orbital forcing of tree-ring data. Nature Clim Change 2:862–866. https://doi.org/10.1038/NCLIMATE1589
Esper J, Düthorn E, Krusic P, Timonen M, Büntgen U (2014) Northern European summer temperature variations over the Common Era from integrated tree-ring density records. J Quat Sci 29:487–494. https://doi.org/10.1002/jqs.2726
Esper J, Schneider L, Smerdon J, Schöne B, Büntgen U (2015) Signals and memory in tree-ring width and density data. Dendrochronologia 35:62–70. https://doi.org/10.1016/j.dendro.2015.07.001
Frère-Cook G (1977) Die “Tirpitz” muss unter Wasser. Die Angriffe auf das größte deutsche Schlachtschiff 1940–1944. Motorbuch, Stuttgart
Fritts HC (1976) Tree rings and climate. Academic Press, New York
Fritts HC, Swetnam TW (1989) Dendroecology: a tool for evaluating variations in past and present forest environments. Adv Ecol Res 19:111–189
Gärtner H, Nievergelt D (2010) The core-microtome: a new tool for surface preparation on cores and time series analysis of varying cell parameters. Dendrochronologia 28:85–92. https://doi.org/10.1016/j.dendro.2009.09.002
Geological Survey Norway (2019) Bedrock—National bedrock database. https://geo.ngu.no/kart/berggrunn_mobil/?lang=eng. Accessed 12 March 2020
H. Dv. (Heeresdienstvorschrift) 211/1 (1940) Verwendung künstlichen Nebels durch die Luftwaffe. Thormann & Goetsch, Berlin
H. Dv. (Heeresdienstvorschrift) 211/2 (1939) Die Nebelmittel und ihre Handhabung Die Nebelkerze. Die Nebelhandgranate. Reichsdruckerei, Berlin
H. Dv. (Heeresdienstvorschrift) 211/3 (1941) Die Nebelmittel und ihre Handhabung. Die Nebelzerstäuber. Reichsdruckerei, Berlin
Hagen SB, Jepsen JU, Ims RA, Yoccoz G (2007) Shifting altitudinal distribution of outbreak zones of winter moth Operophtera brumata in sub-arctic birch forest: a response to recent climate warming? Ecography 30:299–307
Hampe E (1963) Der Zivile Luftschutz im Zweiten Weltkrieg. Dokumentation und Erfahrungsberichte über Aufbau und Einsatz. Bernard & Graefe Verlag für Wehrwesen, Frankfurt
Hartl C, St. George S, Konter O, Harr L, Scholz D, Kirchhefer A, Esper J (2019) Warfare dendrochronology: trees witness the deployment of the German battleship Tirpitz in Norway. Anthropocene 27:1–8
Hartl-Meier C, Zang C, Dittmar C, Esper J, Göttlein A, Rothe A (2014a) Vulnerability of Norway spruce to climate change in mountain forests of the European Alps. Clim Res 60:119–132. https://doi.org/10.3354/cr01226
Hartl-Meier C, Dittmar C, Zang C et al (2014b) Mountain forest growth response to climate change in the Northern Limestone Alps. Trees 28:819–829. https://doi.org/10.1007/s00468-014-0994-1
Haselhoff E, Lindau G (1903) Die Beschädigung der Vegetation durch Rauch. Gebr. Borntraeger, Leipzig
Holmes R (1983) Analysis of tree rings and fire scars to establish fire history. Tree-Ring Bull 43:51–67
Hoogesteger J, Karlsson P (1992) Effects of defoliation on radial stem growth and photosynthesis in the mountain birch (Betula pubescens ssp. tortuosa). Funct Ecol 6:317–323. https://doi.org/10.1111/10.2307/2389523
Jepsen JU, Hagen SB, Ims RA, Yoccoz NG (2008) Climate change and outbreaks of the geometrids Operophtera brumata and Epirrita autumnata in subarctic birch forest: evidence of a recent outbreak range expansion. J Anim Ecol 77:257–264. https://doi.org/10.1111/j.1365-2656.2007.01339.x
Jepsen JU, Kapari L, Hagen SB, Schott T, Vindstad OPL, Nilssen A, Ims RA (2011) Rapid northwards expansion of a forest insect pest attributed to spring phenology matching with sub-Arctic birch. Glob Change Biol 17:2071–2083. https://doi.org/10.1111/j.1365-2486.2010.02370.x
Karlsen SR, Ramfjord H, Høgda HA, Johansen B, Danks FS, Brobakk TE (2009) A satellite-based map on onset of birch (Betula) flowering in Norway. Aerobiologia 25:15–25
Karlsen SR, Jepsen JU, Odland A, Ims RA, Elvebakk A (2013) Outbreaks by canopy-feeding geometrid moth cause state-dependent shifts in understorey plant communities. Oecologia 173:859–870
Karlsson PS, Bylund H, Neuvonen S, Heino S, Tjus M (2003) Climatic response of budburst in the mountain birch at two areas in northern Fennoscandia and possible responses to global change. Ecography 26:617–625. https://doi.org/10.1034/j.1600-0587.2003.03607.x
Karlsson PS, Tenow O, Bylund H, Hoogesteeger J, Weih M (2004) Determinants of mountain birch growth in situ: effects of temperature and herbivory. Ecography 27:659–667
Kirchhefer A (1996) A dendrochronological study on the effect of climate, site, and insect outbreaks on the growth of Betula pubescens coll. in northern Norway. Paleoclim Res 20:93–106
Kirchhefer A (2001) Reconstruction of summer temperatures from tree-rings of Scots pine (Pinus sylvestris L.) in coastal northern Norway. Holocene 11:41–52. https://doi.org/10.1191/095968301670181592
Klaveness D, Wielgolaski FE (1996) Plant phenology in Norway—a summary of past and present first flowering dates (FFDs) with emphasis on conditions within three areas. Phenol Season 1(1):47–61
KNMI Climate Explorer (2020) Metadata. Alta Lufthavn. Internet: https://climexp.knmi.nl/showmetadata.cgi?TYPE=peca&WMO=5001&station=ALTA_LUFTHAVN&id=someone@somewhere. Accessed 12 Mar 2020
Kollas C, Körner C, Randin CF (2013) Spring frost and growing season length co-control the cold range limits of broad-leaved trees. J Biogeogr 41:773–783. https://doi.org/10.1111/jbi.12238
Körner C (2012) Alpine treelines. Functional ecology of the global high elevation tree limits. Springer, Basel, Heidelberg, Dordrecht, London
Kottek M, Grieser J, Beck C, Rudolf B, Rubel F (2006) World Map of the Köppen-Geiger climate classification updated. Meteorl Zeitschrift 15:259–263
Kullman L (2003) Recent reversal of Neoglacial climate cooling trend in the Swedish Scandes as evidenced by mountain birch tree-limit rise. Global Planet Change 36:77–88. https://doi.org/10.1016/S0921-8181(02)00165-0
Kullman L, Öberg L (2009) Post-Little Ice Age tree line rise and climate warming in the Swedish Scandes: a landscape ecological perspective. J Ecol 97:415–429. https://doi.org/10.1111/j.1365-2745.2009.01488.x
Levanic T, Eggertsson O (2008) Climatic effects on birch (Betula pubescens Ehrh.) growth in Fnjoskadalur valley, northern Iceland. Dendrochronologia 25:135–143. https://doi.org/10.1016/j.dendro.2006.12.001
Lyu L, Zhang QB, Pellatt MG, Büntgen U, Li MH, Cherubini P (2019) Drought limitation on tree growth at the Northern Hemisphere’s highest tree line. Dendrochronologia 53:40–47. https://doi.org/10.1016/j.dendro.2018.11.006
Malik I, Danek M, Marchwińska-Wyrwał E, Danek T, Wistuba M, Krąpiec M (2012) Scots pine (Pinus sylvestris L.) growth suppression and adverse effects on human health due to air pollution in the Upper Silesian Industrial District (USID), Southern Poland. Water Air Soil Poll 223:3345–3364. https://doi.org/10.1007/s11270-012-1114-8
Mosteller F, Tukey J (1977) Data analysis and regression: a second course in statistics. Addison-Wesley, Boston
Neumann FG (1991) Responses of litter arthropods to major natural or artificial ecological disturbances in mountain ash forest. Aust J Ecol 16:19–32. https://doi.org/10.1111/j.1442-9993.1991.tb01478.x
Neuvonen S, Niemelä P, Virtanen T (1999) Climatic change and insect outbreaks in boreal forest: the role of winter temperatures. Ecol Bull 47:63–67
Oberkommando der Kriegsmarine (1942) Handbuch über Nebelmittel. Heft 3. Nebelboje 1938. NB 38. Nebelboje 1938 ¼. NB 38 ¼. Reichsdruckerei, Berlin
Opala M, Migala K, Owczarek P (2015) Tree rings of downy birch (Betula pubescens) from island of Tromsøya (Norway) as proxies for past temperature changes in the Low Arctic. New perspectives in polar research. University of Wrocław
Panofsky HA, Brier GW (1958) Some applications of statistics to meteorology. Pennsylvania State University, College of Earth and Mineral Sciences, University Park, Pennsylvania
Popradit A, Srisatit T, Kiratiprayoon S et al (2015) Anthropogenic effects on a tropical forest according to the distance from human settlements. Sci Rep 5:14689. https://doi.org/10.1038/srep14689
Ren P, Ziaco E, Rossi S, Biondi F, Prislan P, Liang E (2019) Growth rate rather than growing season length determines wood biomass in dry environments. Agric For Meteorol 271:46–53. https://doi.org/10.1016/j.agrformet.2019.02.031
Rinn F (2011) RINNTECH. Technology for tree and wood analysis. LINTAB. Packages for your individual needs. https://www.rinntech.de/content/view/16/47/lang,english/index.html. Accessed 12 Mar 2020
Rothe A, Hartl-Meier C (2014) Jahrringuntersuchungen an Bergahorn in Wäldern der Nördlichen Kalkalpen. Der Bergahorn präsentiert sich als eine interessante Mischbaumart im Klimawandel. LWF Aktuell 100:55–57
Seidl R, Schelhaas MJ, Rammer W, Verkerk PJ (2014) Increasing forest disturbances in Europe and their impact on carbon storage. Nature Clim Change 4:806–810. https://doi.org/10.1038/nclimate2318
Seidl R, Thom D, Kautz M, Martin-Benito D, Peltoniemi M, Vacchiano G, Wild J, Ascoli D, Petr M, Honkaniemi J, Lexer MJ, Trotsiuk V, Mairota P, Svoboda M, Fabrika M, Nagel TA, Reyer CPO (2017) Forest disturbances under climate change. Nature Clim Change 7:395–402. https://doi.org/10.1038/NCLIMATE3303
Seppä H, Nyman M, Korhola A, Weckström J (2002) Changes of treelines and alpine vegetation in relation to post-glacial climate dynamics in northern Fennoscandia based on pollen and chironomid records. J Quart Sci 17:287–301. https://doi.org/10.1002/jqs.678
Siljamo P, Sofiev M, Ranta H, Linkosalo T, Kubin E, Ahas R, Genikhovich E, Jatczak K, Jato V, Nekovar J, Minin A, Severova E, Shalaboda V (2008) Representativeness of point-wise phenological Betula data collected in different parts of Europe. Glob Ecol Biogeogr 2008(17):489–502
Tenow O (1972) The outbreaks of Oporinia autumnata Bkh. and Operopthera spp. (Lep., Geometridae) in the Scandinavian mountain chain and Northern Finland 1862–1968. Zool Bidr Upps 2(Suppl):1–107
Tømmervik H, Johansen B, Tombre I, Thannheiser D, Høgda KA, Gaare E, Wiegolaski FE (2004) Vegetation changes in the Nordic mountain Birch Forest: the influence of grazing and climate change. Arctic, Antarctic and Alpine Research. Interdiscipl J 36:323–332. https://doi.org/10.1657/1523-0430(2004)036[0323:VCITNM]2.0.CO;2
Tømmervik H, Johansen B, Riseth JA, Karlsen SR, Solberg B, Høgda KA (2009) Above ground biomass changes in the mountain birch forests and mountain heaths of Finnmarksvidda, northern Norway, in the period 1957–2006. For Ecol Manage 257(2009):244–257
Treshow M, Anderson FK (1991) Plant stress from air pollution. Guildford and Kings Lynn, Chichester
Trotsiuk V, Pederson N, Druckenbrod DL, Orwig DA, Bisho DA, Barker-Plotkin A, Fraver S, Martin-Benito D (2018) Testing the efficacy of tree-805 ring methods for detecting past disturbances. Forest Ecol Manag 425:59–67. https://doi.org/10.1016/j.foreco.2018.05.045
Tumajer J, Treml V (2019) Disentangling the effects of disturbance, climate and tree age on xylem hydraulic conductivity of Betula pendula. Ann Botany 5:783–792. https://doi.org/10.1093/aob/mcy209
Wigley T, Briffa K, Jones P (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorology. J Clim Appl Meteorol 23:201–213
Zang C, Biondi F (2015) Treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38:001–006. https://doi.org/10.1111/ecog.01335
Zang C, Hartl-Meier C, Dittmar C, Rothe A, Menzel A (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and variability. Glob Change Biol 20:3767–3779
Acknowledgements
We thank Markus Kochbeck for laboratory support and several students from Johannes Gutenberg University Mainz supporting fieldwork during excursions to North Scandinavia in summer 2017. Supported by the German Science Foundation projects “Inst 247/665-1 FUGG”, “ES 161/9-1”, and “HA 8084/1-1”.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
The work presented here was carried out in collaboration between all authors. CH and LH designed the study. CH carried out the field work, LH and CH the laboratory work. Data analyses and interpretation were joint work among all authors. The paper was written by LH and revised by AK, JE, ZW and CH.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Piovesan.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original article has been revised to update the Open access.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Harr, L., Esper, J., Kirchhefer, J.A. et al. Growth response of Betula pubescens Ehrh. to varying disturbance factors in northern Norway. Trees 35, 421–431 (2021). https://doi.org/10.1007/s00468-020-02043-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-020-02043-1