Abstract
Key message
Outplanted Polylepis australis seedling growth, survival and mycorrhizal response were not influenced by inoculation with soil from different vegetation types. Seedling inoculation would not be essential for reforestation practices.
Abstract
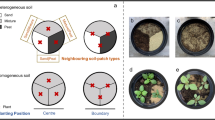
Polylepis forests are one of the most endangered high mountain ecosystems of South America and reforestation with native Polylepis species has been recommended. To determine whether native soil inoculation could help in reforestation success, a field trial was set up to evaluate the response of outplanted P. australis seedlings to the inoculation with soils from three vegetation types (a grassland, a mature forest and a degraded forest) and a sterile soil, used as control. We evaluated seedlings performance: growth and survival for 18 months, root/shoot ratio, phosphorous content and arbuscular mycorrhizal fungal (AMF) colonization. To interpret performance patterns we evaluated the colonization potential of the three inoculum soils and the changes of the AMF community composition of the seedlings rhizosphere in relation to inoculation treatment and season. Our main results showed no significant differences in seedlings survival and growth between treatments. The colonization potential of grassland and degraded forest soils was ~25 times greater than mature forest soil and specific spore density of some morphospecies varied with season. However, AMF spore community of seedlings rhizosphere became homogenized after outplanting and was similar between treatments after 12 months. Therefore, we conclude that soil inoculation is not essential for outplanted P. australis survival and increase in height, and thus all the tested soils could be used as inocula, including grassland soils which in practice are the easiest to collect.




Similar content being viewed by others
References
Allen EB, Allen MF, Egerton-Warburton LM, Corkidi L, Gómez-Pompa A (2003) Impacts of early-and late-seral mycorrhizae during restoration in seasonal tropical forest. Ecol Appl 13:1701–1717
Amaranthus M, Steinfeld D (2005) Arbuscular mycorrhizal inoculation following biocide treatment improves Calocedrus decurrens survival and growth in nursery and outplanting sites. In: Dumroese R, Riley LE, Landis T (eds) National Proceedings: forest and conservation nursery associations 2004; 2004 July 12–15; Charleston, NC; and 2004 July 26–29; Medford, OR. Proc. RMRS-P-35. Fort Collins, pp 103–108
Asbjornsen H, Montagnini F (1994) Vesicular-arbuscular mycorrhizal inoculum potential affects the growth of Stryphnodendron microstachyum seedlings in a costa rican human tropical lowland. Mycorrhiza 5:45–51
Becerra AG, Cabello M, Zak MR, Bartoloni N (2009) Arbuscular mycorrhizae of dominant plant species in Yungas forests, Argentina. Mycologia 101:612–621
Bever JD, Schultz PA, Pringle A, Morton JB (2001) Arbuscular mycorrhizal fungi: more diverse than meets the eye, and the ecological tale of why. Bioscience 51:923–932
Cabello MN (1997) Hydrocarbon pollution: its effect on native arbuscular mycorrhizal fungi (AMF). FEMS Microbiol Ecol 22:233–236
Cabido M (1985) Las comunidades vegetales de Pampa de Achala, Sierras de Córdoba. Argen Doc Phytosociol 9:431–443
Cingolani AM, Cabido M, Renison D, Solís Neffa V (2003) Combined effects of environment and grazing on vegetation structure in argentine granite grasslands. J Veg Sci 14:223–232
Cingolani AM, Renison D, Zak MR, Cabido M (2004) Mapping vegetation in a heterogeneous mountain rangeland using landsat data: an alternative method to define and classify land-cover units. Remot Sens Environ 92:84–97
Cingolani AM, Renison D, Tecco PA, Gurvich DE, Cabido M (2008) Predicting cover types in a mountain range with long evolutionary grazing history: a GIS approach. J Biogeogr 35:538–551
Closa I, Goicoechea N (2011) Infectivity of arbuscular mycorrhizal fungi in naturally regenerating, unmanaged and clear-cut beech forests. Pedosphere 21:65–74
Cuenca G, De Andrade Z, Lovera M, Fajardo L, Meneses E (2003) Mycorrhizal response of Clusia pusilla growing in two different soils in the field. Trees 17:200–206
Cuenca G, De Andrade Z, Lovera M, Fajardo L, Meneses E (2004) The effect of two arbuscular mycorrhizal inocula of contrasting richness and the same mycorrhizal potential on the growth and survival of wild plant species from La Gran Sabana, Venezuela. Can J Bot 82:582–589
Díaz G, Honrubia M (1993) Infectivity of mine soils from southeast Spain. II. Mycorrhizal population levels in spoilt sites. Mycorrhiza 4:85–88
Dumbrell AJ, Nelson M, Helgason T, Dytham C, Fitter AH (2010) Relative roles of niche and neutral processes in structuring a soil microbial community. ISME J 4:337–345
Dumbrell AJ, Ashton PD, Aziz N, Feng G, Nelson M, Dytham C, Fitter AH, Helgason T (2011) Distinct seasonal assemblages of arbuscular mycorrhizal fungi revealed by massively parallel pyro-sequencing. New Phytol 190:794–804
Ellenberg H (1979) Man’s influence on tropical mountain ecosystems in South America. J Ecol 67:401–416
Fischer CR, Janos DP, Perry DA, Linderman RG, Sollins P (1994) Mycorrhiza inoculum potentials in tropical secondary succession. Biotropica 26:369
Fjeldså J, Kessler M (1996) Conserving the biological diversity of Polylepis woodlands of the highland of Peru and Bolivia. A contribution to sustainable natural resource management in the Andes, Nordeco
Friese CF, Koske RE (1991) The spatial dispersion of spores of vesicular-arbuscular mycorrhizal fungi in a sand dune: microscale patterns associated with the root architecture of American beachgrass. Mycol Res 95:952–957
Gerdemann JW, Nicolson TH (1963) Spores of mycorrhizal endogone species extracted from soil by wet sieving and decanting. Trans Br Mycol Soc 46:235–244
Giovannetti M, Mosse B (1980) An evaluation of techniques for measuring vesicular-arbuscular mycorrhizal infection in roots. New Phytol 84:489–500
Grace C, Stribley DP (1991) A safer procedure for routine staining of vesicular-arbuscular mycorrhizal fungi. Mycol Res 95:1160–1162
Hempel S, Renker C, Buscot F (2007) Differences in the species composition of arbuscular mycorrhizal fungi in spore, root and soil communities in a grassland ecosystem. Environ Microbiol 9:1930–1938
Heneghan L, Miller SP, Baer S, Callaham MA, Montgomery J, Pavao-Zuckerman M, Rhoades CC, Richardson S (2008) Integrating soil ecological knowledge into restoration management. Rest Ecol 16:608–617
Hoeksema JD, Chaudhary VB, Gehring CA, Collins Johnson N, Karst J, Koide RT, Pringle A, Zabinski C, Bever JD, Moore JC, Wilson GWT, Klironomos JN, Ubanhowar J (2010) A meta-analysis of context-dependency in plant response to inoculation with mycorrhizal fungi. Ecol Lett 13:394–407
Irrazabal G, Velázquez S, Cabello MN (2004) Infectividad y diversidad de HMA de la rizosfera de los talares de Magdalena, provincia de Buenos Aires, Argentina. Bol Micol 19:49–57
Jasper DA, Abbot LK, Robson AD (1991) The effect of soil disturbance on vesicular arbuscular mycorrhizal fungi in soils from different vegetation types. New Phytol 118:471–476
Kauffman JB, Steele MD, Cummings DL, Jaramillo VJ (2003) Biomass dynamics associated with deforestation, fire, and conversion to cattle pasture in a mexican tropical dry forest. For Ecol Man 176:1–12
Kiers ET, Lovelock CE, Krueger EL, Herre EA (2000) Differential effects of tropical arbuscular mycorrhizal fungal inocula on root colonization and tree seedling growth: implications for tropical forest diversity. Ecol Lett 3:106–113
Klironomos JN, Hart MM (2002) Colonization of roots by arbuscular mycorrhizal fungi using different sources of inoculum. Mycorrhiza 12:181–184
Koske RE, Gemma JN (1990) VA mycorrhizae in strand vegetation of Hawaii: evidence for long-distance codispersal of plants and fungi. Am J Bot 77:466–474
Lekberg Y, Koide RT (2005) Is plant performance limited by abundance of arbuscular mycorrhizal fungi? A meta-analysis of studies published between 1988 and 2003. New Phytol 168:189–204
López-García A, Hempel S, Miranda JdeD, Rillig MC, Barea JM, Azcón-Aguilar C (2013) The influence of environmental degradation processes on the arbuscular mycorrhizal fungal community associated with yew (Taxus baccata L.), an endangered tree species from mediterranean ecosystems of southeast Spain. Plant Soil 370:355–366
Lugo M, Cabello MN (2002) Native arbuscular mycorrhizal fungi (AMF) from mountain grassland (Cordoba, Argentina) I. Seasonal variation of fungal spore diversity. Mycologia 94:579–586
Magurran AE, McGill BJ (2011) Biological diversity: frontiers in measurement and assessment. Oxford University Press, Oxford, UK
Maherali H, Klironomos JN (2007) Influence of phylogeny on fungal community assembly and ecosystem functioning. Science 316:1746–1748
Maron JL, Marler M, Klironomos JN, Cleveland CC (2011) Soil fungal pathogens and the relationship between plant diversity and productivity. Ecol Lett 14:36–41
Martino J, Urcelay C, Renison D (2011) Crecimiento y colonización micorrícica de Polylepis australis Bitter (Rosaceae) en suelos con distinta historia de pastoreo. Kurtziana 36:69–77
McGonigle TP, Miller M, Evans DG, Fairchild GL, Swan JA (1990) A method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501
Menoyo E, Becerra AG, Renison D (2007) Mycorrhizal associations in Polylepis woodlands of central Argentina. Can J Bot 85:526–531
Merryweather J, Fitter AH (1998) The arbuscular mycorrhizal fungi of hyacinthoides non-scripta II. Seasonal and spatial patterns of fungal populations. New Phytol 138:131–142
Oehl F, Sieverding E, Ineichen K, Mäder P, Wiemken A, Boller T (2009) Distinct sporulation dynamics of arbuscular mycorrhizal fungal communities from different agroecosystems in long-term microcosms. Agric Ecosys Environ 134:257–268
Oehl F, Sieverding E, Palenzuela J, Ineichen K, Alves da Silva G (2011) Advances in Glomeromycota taxonomy and classification. IMA Fungus 2:191–199
Omar MB, Bolland L, Heather WA (1979) P.V.A. (polyvinyl alcohol). A permanent mounting medium for fungi. Bull Br Mycol Soc 13:31–32
Phillips JM, Hayman DS (1970) Improved procedures for clearing roots and staining parasitic and vesicular arbuscular mycorrhizal fungi for rapid assessment of infection. Tran Br Mycol Soc 55:158–161
Picone C (2000) Diversity and abundance of arbuscular-mycorrhizal fungus spores in tropical forest and pasture. Biotropica 32:734–750
Plenchette C, Perrin R, Duvert P (1989) The concept of soil infectivity and a method for its determination as applied to endomycorrhizas. Can J Bot 67:112–115
Pringle A, Bever JD (2002) Divergent phenologies may facilitate the coexistence of arbuscular mycorrhizal fungi in a North Carolina grassland. Am J Bot 89:1439–1446
R Development Core Team (2011) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org
Renison D, Cingolani AM (2002) Evaluación de la supervivencia y crecimiento de plantines de Polylepis australis (Rosaceae) para la elección de plantas semilleras. Agriscientia 19:63–66
Renison D, Cingolani AM, Schinner D (2002) Optimizing restoration of Polylepis australis woodlands: when, where and how to transplant seedlings to the mountains? Ecotropica 8:219–224
Renison D, Cingolani AM, Suarez R, Menoyo E, Coutsiers C, Sobral A, Hensen I (2005) The restoration of degraded mountain woodlands: effects of seed provenance and microsite characteristics on Polylepis australis seedling survival and growth in central Argentina. Res Ecol 13:129–137
Renison D, Hensen I, Suarez R, Cingolani AM, Marcora P, Giorgis MA (2010) Soil conservation in Polylepis mountain forests of central Argentina: Is livestock reducing our natural capital? Austral Ecol 35:435–443
Renison D, Hensen I, Suarez R (2011) Landscape structural complexity of high-mountain Polylepis australis forests: a new aspect of restoration goals. Res Ecol 19:390–398
Renison D, Cuyckens GAE, Pacheco S, Guzmán GF, Grau HR, Marcora P, Robledo GL, Cingolani AM, Dominguez J, Landi M, Bellis L, Hensen I (2013) Distribución y estado de conservación de las poblaciones de árboles y arbustos del género Polylepis (Rosaceae) en las montañas de Argentina. Ecol Austral 23:27–36
Rillig MC (2004) Arbuscular mycorrhizae and terrestrial ecosystem processes. Ecol Lett 7:740–754
Salgado Salomón ME, Barroetaveña C, Rajchenberg M (2010) Do pine plantations provide mycorrhizal inocula for seedlings establishment in grasslands from Patagonia, Argentina? New For 41:191–205
Sánchez-Castro I, Ferrol N, Cornejo P, Barea JM (2012) Temporal dynamics of arbuscular mycorrhizal fungi colonizing roots of representative shrub species in a semi-arid mediterranean ecosystem. Mycorrhiza 22:449–460
Schenk NC, Perez Y (1990) Manual of identification of vesicular-arbuscular mycorrhizal fungi. Gainesville, USA
Sene G, Thiao M, Manga A, Kane A, Samba-Mbaye R, Samba-Mbaye M, Khasa D, Sylla SN (2012) Arbuscular mycorrhizal soil infectivity and spores distribution across plantations of tropical, subtropical and exotic tree species: a case study from the forest reserve of Bandia. Senegal Afr J Ecol 50:218–232
Smith SE, Read D (2008) Mycorrhizal symbiosis, 3rd edn. Academic Press, Great Britain
Smith SE, Jakobsen I, Grønlund M, Smith FA (2011) Roles of arbuscular mycorrhizas in plant phosphorus nutrition: interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiol 156:1050–1057
Soteras F, Becerra A, Cofré N, Bartoloni J, Cabello M (2012) Arbuscular mycorrhizal fungal species in saline environments of Central Argentina: seasonal variation and distribution of spores at different soil depths. Sydowia 64:301–311
Soteras F, Renison D, Becerra AG (2013) Growth response, phosphorus content and root colonization of Polylepis australis Bitt. seedlings inoculated with different soil types. New For 44:577–589
Stürmer SL, Filho OK, De Queiroz H, De Mendonça MM (2006) Occurrence of arbuscular mycorrhizal fungi in soils of early stages of a secondary succession of Atlantic forest in south Brazil. Acta Bot Bras 20:513–521
Tommerup IC (1983) Temperature relations of spore germination and hyphal growth of vesicular-arbuscular mycorrhizal fungi in soil. Trans Brit Mycol Soc 81:381–387
Torrecillas E, Alguacil del Mar M, Roldán A (2012) Differences in the AMF diversity in soil and roots between two annual and perennial gramineous plants co-occurring in a mediterranean, semiarid degraded area. Plant Soil 354:97–106
Torres R, Renison D, Hensen I, Suarez R, Enrico L (2008) Polylepis australis’ regeneration niche in relation to seed dispersal, site characteristics and livestock density. For Ecol Man 254:255–260
Velázquez S, Cabello M (2011) Occurrence and diversity of arbuscular mycorrhizal fungi in trap cultures from El Palmar national park soils. Eur J Soil Biol 47:230–235
Walker C, Mize W, McNabb HS (1982) Populations of endogonaceous fungi at two populations in central Iowa. Can J Bot 60:2518–2529
Zak MR, Cabido M (2002) Spatial patterns of the Chaco vegetation of central argentina: integration of remote sensing and phytosociology. Appl Veg Sci 5:213–226
Acknowledgments
This work was financially supported by Agencia de Promoción Científica y Tecnológica––PICT 438-2008, Consejo Nacional de Investigaciones Científicas y Técnicas––PIP 0269 and Secretaría de Ciencia y Técnica de la Universidad Nacional de Córdoba (SECyT-UNC). F.S. is grateful to CONICET for providing her fellowship. A.B. and D.R. are researchers of CONICET. We thank to the Quebrada del Condorito National Park authorities that generously allowed the use of the lands for the reforestation assay. This research would not have been possible without numerous volunteers (especially Barberá I., Berizzo A., Bernaschini M.L., Domínguez J., Farias G., Flores S., Galli E., Gatica L., Peralta G., Street E., and researchers and PhD students of the Laboratorio de Micología-IMBIV-) who provided exceptional field assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. E. Courty.
Rights and permissions
About this article
Cite this article
Soteras, F., Renison, D. & Becerra, A.G. Restoration of high altitude forests in an area affected by a wildfire: Polylepis australis Bitt. seedlings performance after soil inoculation. Trees 28, 173–182 (2014). https://doi.org/10.1007/s00468-013-0940-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-013-0940-7