Abstract
Background
Nephropathic cystinosis is an autosomal recessive disease caused by a mutation in the CTNS gene which encodes cystinosin, a lysosomal cystine transporter. The spectrum of mutations in the CTNS gene is not well defined in the North African population. Here, we investigated twelve patients with nephropathic cystinosis belonging to eight Tunisian families in order to analyze the clinical and genetic characteristics of Tunisian children with infantile nephropathic cystinosis.
Methods
Clinical data were collected retrospectively. Molecular analysis of the CTNS gene was performed by Sanger sequencing.
Results
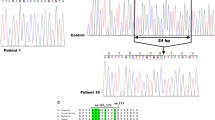
We describe a new splicing mutation c.971-1G > C in the homozygous state in 6/12 patients which seems to be a founder mutation. The reported deletion of 23nt c.771_793 Del (p.Gly258Serfs*30) was detected in a homozygous state in one patient and in a heterozygous compound state with the c.971-1G > C mutation in 3/12 patients. Two of 12 patients have a deletion of exons 4 and 5 of the CTNS gene. None of our patients had the most common 57-kb deletion.
Conclusions
The mutational spectrum in the Tunisian population is different from previously described populations. Thus, a molecular diagnostic strategy must be implemented in Tunisia, by targeting as a priority the common mutations described in this country. Such a strategy will allow a cost-effective diagnosis confirmation as well as early administration of treatment with oral cysteamine.
Graphical abstract
A higher resolution version of the Graphical abstract is available as Supplementary information






Similar content being viewed by others
References
Elmonem MA, Veys KR, Soliman NA, van Dyck M, van den Heuvel LP, Levtchenko E (2016) Cystinosis: a review. Orphanet J Rare Dis 11:47. https://doi.org/10.1186/s13023-016-0426-y
Gahl WA, Thoene JG, Schneider JA (2002) Cystinosis. N Engl J Med 347:111–121. https://doi.org/10.1056/NEJMra020552
Kalatzis V, Cherqui S, Antignac C, Gasnier B (2001) Cystinosin, the protein defective in cystinosis, is a H+-driven lysosomal cystine transporter. EMBO J 20:5940–5949. https://doi.org/10.1093/emboj/20.21.5940
Bellomo F, Taranta A, Petrini S, Venditti R, Rocchetti MT, Rega LR, Corallini S, Gesualdo L, De Matteis MA, Emma F (2016) Carboxyl-terminal SSLKG motif of the human cystinosin-LKG plays an important role in plasma membrane sorting. PLoS One 11:e0154805
Bertholet-Thomas A, Berthiller J, Tasic V, Kassai B, Otukesh H, Greco M, Ehrich J, de Paula BR, Deschênes G, Hulton S-A, Fischbach M, Soulami K, Saeed B, Valavi E, Cobenas CJ, Hacihamdioglu B, Weiler G, Cochat P, Bacchetta J (2017) Worldwide view of nephropathic cystinosis: results from a survey from 30 countries. BMC Nephrol 18:210. https://doi.org/10.1186/s12882-017-0633-3
Chkioua L, Khedhiri S, Grissa O, Aloui C, Turkia HB, Ferchichi S, Miled A, Froissart R, Acquaviva C, Laradi S (2015) Genetic basis of cystinosis in Tunisian patients: identification of novel mutation in CTNS gene. Meta Gene 5:144–149. https://doi.org/10.1016/j.mgene.2015.07.003
Owen EP, Nandhlal J, Leisegang F, Van der Watt G, Nourse P, Gajjar P (2015) Common mutation causes cystinosis in the majority of black South African patients. Pediatr Nephrol 30:595–601. https://doi.org/10.1007/s00467-014-2980-7
WHO Multicentre Growth Reference Study Group (2006) WHO Child Growth Standards based on length/height, weight and age. Acta Paediatr Suppl 450:76–85. https://doi.org/10.1111/j.1651-2227.2006.tb02378.x
Harrison CM, Johnson K, McKechnie E (2008) Osteopenia of prematurity: a national survey and review of practice. Acta Paediatr 97:407–413. https://doi.org/10.1111/j.1651-2227.2007.00721.x
Schwartz GJ, Muñoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL (2009) New equations to estimate GFR in children with CKD. J Am Soc Nephrol 20:629–637. https://doi.org/10.1681/ASN.2008030287
Alcántara-Ortigoza MÁ, Belmont-Martínez L, Vela-Amieva M, González-Del Angel A (2008) Analysis of the CTNS gene in nephropathic cystinosis Mexican patients: report of four novel mutations and identification of a false positive 57-kb deletion genotype with LDM-2/Exon 4 Multiplex PCR Assay. Genet Test 12:409–414. https://doi.org/10.1089/gte.2008.0014
Ouhenach M, Zrhidri A, Jaouad IC, Smaili W, Sefiani A (2020) Application of next generation sequencing in genetic counseling a case of a couple at risk of cystinosis. BMC Med Genet 21:240. https://doi.org/10.1186/s12881-020-01167-y
Mason S, Pepe G, Dall’Amico R, Tartaglia S, Casciani S, Greco M, Bencivenga P, Murer L, Rizzoni G, Tenconi R, Clementi M (2003) Mutational spectrum of the CTNS gene in Italy. Eur J Hum Genet 11:503–508. https://doi.org/10.1038/sj.ejhg.5200993
Najafi M, Tamandani DMK, Azarfar A, Bakey Z, Behjati F, Antony D, Schüle I, Sadeghi-Bojd S, Karimiani EG, Schmidts M (2019) A 57 kB genomic deletion causing CTNS loss of function contributes to the CTNS mutational spectrum in the Middle East. Front Pediatr 7:89. https://doi.org/10.3389/fped.2019.00089
Soliman NA, Elmonem MA, van den Heuvel L, Abdel Hamid RH, Gamal M, Bongaers I, Marie S, Levtchenko E (2014) Mutational spectrum of the CTNS gene in Egyptian patients with nephropathic cystinosis. JIMD Rep 14:87–97. https://doi.org/10.1007/8904_2013_288
Aldahmesh MA, Humeidan A, Almojalli HA, Khan AO, Rajab M, Al-Abbad AA, Meyer BF, Alkuraya FS (2009) Characterization of CTNS mutations in Arab patients with cystinosis. Ophthalmic Genet 30:185–189. https://doi.org/10.3109/13816810903200953
Topaloglu R, Vilboux T, Coskun T, Ozaltin F, Tinloy B, Gunay-Aygun M, Bakkaloglu A, Besbas N, van den Heuvel L, Kleta R, Gahl WA (2011) Genetic basis of cystinosis in Turkish patients: a single-center experience. Pediatr Nephrol 27:115–121. https://doi.org/10.1007/s00467-011-1942-6
Shahkarami S, Galehdari H, Ahmadzadeh A, Babaahmadi M, Pedram M (2013) The first molecular genetics analysis of individuals suffering from nephropathic cystinosis in the Southwestern Iran. Nefrologia 33:308–315. https://doi.org/10.3265/Nefrologia.pre2012.Sep.11558
Topaloglu R (2021) Nephropathic cystinosis: an update on genetic conditioning. Pediatr Nephrol 36:1347–1352. https://doi.org/10.1007/s00467-020-04638-9
Ben Halim N, Ben Alaya Bouafif N, Romdhane L, Kefi Ben Atig R, Chouchane I, Bouyacoub Y, Arfa I, Cherif W, Nouira S, Talmoudi F, Lasram K, Hsouna S, Ghazouani W, Azaiez H, El Matri L, Abid A, Tebib N, Ben Dridi M-F, Kachboura S, Amouri A, Mokni M, Ben Arab S, Dellagi K, Abdelhak S (2012) Consanguinity, endogamy, and genetic disorders in Tunisia. J Community Genet 4:273–284. https://doi.org/10.1007/s12687-012-0128-7
Bois E, Feingold J, Frenay P, Briard ML (1976) Infantile cystinosis in France: genetics, incidence, geographic distribution. J Med Genet 13:434–438. https://doi.org/10.1136/jmg.13.6.434
Hutchesson AC, Bundey S, Preece MA, Hall SK, Green A (1998) A comparison of disease and gene frequencies of inborn errors of metabolism among different ethnic groups in the West Midlands, UK. J Med Genet 35:366–370. https://doi.org/10.1136/jmg.35.5.366
Manz F, Gretz N (1985) Cystinosis in the Federal Republic of Germany. Coordination and analysis of the data. J Inherit Metab Dis 8:2–4. https://doi.org/10.1007/BF01805472
Meikle PJ (1999) Prevalence of lysosomal storage disorders. JAMA 281:249. https://doi.org/10.1001/jama.281.3.249
Ebbesen F, Mygind KI, Holck F (1976) Infantile nephropatic cystinosis in Denmark. Dan Med Bull 23:216–222
Higashi S, Matsunoshita N, Otani M, Tokuhiro E, Nozu K, Ito S (2017) Diagnostic challenge in a patient with nephropathic juvenile cystinosis: a case report. BMC Nephrol 18:300. https://doi.org/10.1186/s12882-017-0721-4
Kasimer RN, Langman CB (2021) Adult complications of nephropathic cystinosis: a systematic review. Pediatr Nephrol 36:223–236. https://doi.org/10.1007/s00467-020-04487-6
Emma F, Hoff WV, Hohenfellner K, Topaloglu R, Greco M, Ariceta G, Bettini C, Bockenhauer D, Veys K, Pape L, Hulton S, Collin S, Ozaltin F, Servais A, Deschênes G, Novo R, Bertholet-Thomas A, Oh J, Cornelissen E, Janssen M, Haffner D, Ravà L, Antignac C, Devuyst O, Niaudet P, Levtchenko E (2021) An international cohort study spanning five decades assessed outcomes of nephropathic cystinosis. Kidney Int 100:1112–1123. https://doi.org/10.1016/j.kint.2021.06.019
Baralle D, Baralle M (2005) Splicing in action: assessing disease causing sequence changes. J Med Genet 42:737–748. https://doi.org/10.1136/jmg.2004.029538
Kurosaki T, Maquat LE (2016) Nonsense-mediated mRNA decay in humans at a glance. J Cell Sci 129:461–467. https://doi.org/10.1242/jcs.181008
Kalatzis V, Antignac C (2009) Hereditary cystinosis. In: Lifton RP, Somlo S, Giebisch GH, Seldin DW (eds) Genetic diseases of the kidney, 1st edn. Elsevier, Burlington, pp 661–679
Stearns PN, Langer WL (eds) (2001) The encyclopedia of world history: ancient, Medieval, and Modern, chronologically arranged. Houghton Mifflin, Boston
Sadeghipour F, Basiratnia M, Derakhshan A, Fardaei M (2017) Mutation analysis of the CTNS gene in Iranian patients with infantile nephropathic cystinosis: identification of two novel mutations. Hum Genome Var 4:17038. https://doi.org/10.1038/hgv.2017.38
Jaradat S, Al-Rababah B, Hazza I, Akl K, Saca E, Al-Younis D (2015) Molecular analysis of the CTNS gene in Jordanian families with nephropathic cystinosis. Nefrologia 35:547–553. https://doi.org/10.1016/j.nefro.2015.09.009
Rupar CA (2001) A G339R mutation in the CTNS gene is a common cause of nephropathic cystinosis in the south western Ontario Amish Mennonite population. J Med Genet 38:615–616. https://doi.org/10.1136/jmg.38.9.615
David D, PrincieroBerlingerio S, Elmonem Mohamed A, Oliveira Arcolino F, Soliman N, van den Heuvel B, Gijsbers R, Levtchenko E (2019) Molecular basis of cystinosis: geographic distribution, functional consequences of mutations in the CTNS gene, and potential for repair. Nephron 141:133–146. https://doi.org/10.1159/000495270
Topaloglu R, Gulhan B, İnözü M, Canpolat N, Yilmaz A, Noyan A, Dursun İ, Gökçe İ, Gürgöze MK, Akinci N, Baskin E, Serdaroğlu E, DemircioğluKiliç B, Yüksel S, ÖvünçHacihamdioğlu D, Korkmaz E, Hayran M, Ozaltin F, contributors of The Turkish Cystinosis Study Group (2017) The clinical and mutational spectrum of Turkish patients with cystinosis. Clin J Am Soc Nephrol 12:1634–1641. https://doi.org/10.2215/CJN.00180117
Brasell EJ, Chu L, El Kares R, Seo JH, Loesch R, Iglesias DM, Goodyer P (2018) The aminoglycoside geneticin permits translational read through of the CTNS W138X nonsense mutation in fibroblasts from patients with nephropathic cystinosis. Pediatr Nephrol 34:873–881. https://doi.org/10.1007/s00467-018-4094-0
Kalatzis V, Cherqui S, Jean G, Cordier B, Cochat P, Broyer M, Antignac C (2001) Characterization of a putative founder mutation that accounts for the high incidence of cystinosis in Brittany. J Am Soc Nephrol 12:2170–2174. https://doi.org/10.1681/ASN.V12102170
Papizh S, Serzhanova V, Filatova A, Skoblov M, Tabakov V, van den Heuvel L, Levtchenko E, Prikhodina L (2019) CTNS mRNA molecular analysis revealed a novel mutation in a child with infantile nephropathic cystinosis: a case report. BMC Nephrol 20:400. https://doi.org/10.1186/s12882-019-1589-2
Attard M, Jean G, Forestier L, Cherqui S, van’t Hoff W, Broyer M, Antignac C, Town M (1999) Severity of phenotype in cystinosis varies with mutations in the CTNS gene: predicted effect on the model of cystinosin. Hum Mol Genet 8:2507–2514. https://doi.org/10.1093/hmg/8.13.2507
Prencipe G, Caiello I, Cherqui S, Whisenant T, Petrini S, Emma F, De Benedetti F (2014) Inflammasome activation by cystine crystals: implications for the pathogenesis of cystinosis. J Am Soc Nephrol 25:1163–1169. https://doi.org/10.1681/ASN.2013060653
Beck CR, Garcia-Perez JL, BadgeMoran RM (2011) LINE-1 elements in structural variation and disease. Annu Rev Genomics Hum Genet 12:187–215. https://doi.org/10.1146/annurev-genom-082509-141802
Sen SK, Han K, Wang J, Lee J, Wang H, Callinan PA, Dyer M, Cordaux R, Liang P, Batzer MA (2006) Human genomic deletions mediated by recombination between Alu elements. Am J Hum Genet 79:41–53. https://doi.org/10.1086/504600
Forestier L, Jean G, Attard M, Cherqui S, Lewis C, van’t Hoff W, Broyer M, Town M, Antignac C (1999) Molecular characterization of CTNS deletions in nephropathic cystinosis: development of a PCR-based detection assay. Am J Hum Genet 65:353–359. https://doi.org/10.1086/302509
Nesta AV, Tafur D, Beck CR (2021) Hotspots of human mutation. Trends Genet 37:717–729. https://doi.org/10.1016/j.tig.2020.10.003
Vargas-Poussou R, Dahan K, Kahila D, Venisse A, Riveira-Munoz E, Debaix H, Grisart B, Bridoux F, Unwin R, Moulin B, Haymann JP, Vantyghem MC, Rigothier C, Dussol B, Godin M, Nivet H, Dubourg L, Tack I, Gimenez-Roqueplo AP, Houillier P, Blanchard A, Devuyst O, Jeunemaitre X (2011) Spectrum of mutations in Gitelman syndrome. J Am Soc Nephrol 22:693–703. https://doi.org/10.1681/ASN.2010090907
Romdhane L, Kefi R, Azaiez H, Ben Halim N, Dellagi K, Abdelhak S (2012) Founder mutations in Tunisia: implications for diagnosis in North Africa and Middle East. Orphanet J Rare Dis 7:52. https://doi.org/10.1186/1750-1172-7-52
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The study was performed in accordance with the ethical standards of the institutional research and Charles Nicolle Hospital local ethics committee.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
El Younsi, M., Trabelsi, M., Ben Youssef, S. et al. Clinical and genetic characteristics of Tunisian children with infantile nephropathic cystinosis. Pediatr Nephrol 38, 119–129 (2023). https://doi.org/10.1007/s00467-022-05525-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-022-05525-1