Abstract
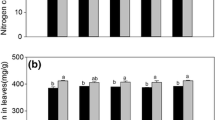
Herbivore-induced changes in host quality mediate indirect interactions between herbivores. The nature of these indirect interactions can vary depending on the identity of herbivores involved, species-specific induction of defense-signaling pathways, and sequence of attack. However, our understanding of the role of these signaling pathways in the success of multiple exotic herbivores is less known. Eastern hemlock (Tsuga canadensis) is attacked by two invasive herbivores [elongate hemlock scale (EHS; Fiorinia externa) and hemlock woolly adelgid (HWA; Adelges tsugae)] throughout much of its range, but prior attack by EHS is known to deter HWA. The potential role of phytohormones in this interaction is poorly understood. We measured endogenous levels of phytohormones in eastern hemlock in response to attack by these invasive herbivores. We also used exogenous application of methyl jasmonate (MJ) and acibenzolar-S-methyl (ASM), a salicylic acid (SA) pathway elicitor, to test the hypothesis that defense-signaling phytohormones typically induced by herbivores could deter HWA. Resistance to adelgid attack was assessed using a behavioral assay. Adelgid feeding significantly elevated both abscisic acid (ABA) and SA in local tissues, while EHS feeding had no detectable effect on either phytohormone. HWA progrediens and sistens crawlers preferred to settle on ASM-treated foliage. In contrast, HWA crawlers actively avoided settlement on MJ-treated foliage. We suggest that induction of ABA- and SA-signaling pathways, in concert with defense-signaling interference, may aid HWA invasion success, and that defense-signaling interference, induced by exotic competitors, may mediate resistance of native hosts.


Similar content being viewed by others
References
Ahmed S, Orians CM, Griffin TS et al (2014) Effects of water availability and pest pressures on tea (Camellia sinensis) growth and functional quality. AoB Plants. https://doi.org/10.1093/aobpla/plt054
Alborn HT, Turlings TCJ, Jones TH et al (1997) An elicitor of plant volatiles from beet armyworm oral secretion. Science 276:945–949. https://doi.org/10.1126/science.276.5314.945
Bates D, Maechler M, Bolker B et al (2015) Fitting linear mixed-effects models using lme4. J Stat Soft 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bhiry N, Filion L (1996) Mid-holocene hemlock decline in eastern North America linked with phytophagous insect activity. Quat Res 45:312–320. https://doi.org/10.1006/qres.1996.0032
Bos JIB, Prince D, Pitino M et al (2010) A functional genomics approach identifies candidate effectors from the aphid species Myzus persicae (green peach aphid). PLoS Genet 6:e1001216. https://doi.org/10.1371/journal.pgen.1001216
Butin E, Preisser E, Elkinton J (2007) Factors affecting settlement rate of the hemlock woolly adelgid, Adelges tsugae, on eastern hemlock, Tsuga canadensis. Agric For Entomol 9:215–219. https://doi.org/10.1111/j.1461-9563.2007.00334.x
Christensen SA, Nemchenko A, Borrego E et al (2013) The maize lipoxygenase, ZmLOX10, mediates green leaf volatile, jasmonate and herbivore-induced plant volatile production for defense against insect attack. Plant J 74:59–73
Chung SH, Rosa C, Scully ED et al (2013) Herbivore exploits orally secreted bacteria to suppress plant defenses. Proc Natl Acad Sci 110:15728–15733. https://doi.org/10.1073/pnas.1308867110
Conconi A, Miquel M, Browse JA, Ryan CA (1996) Intracellular levels of free linolenic and linoleic acids increase in tomato leaves in response to wounding. Plant Physiol 111:797–803. https://doi.org/10.1104/pp.111.3.797
Creelman RA, Tierney ML, Mullet JE (1992) Jasmonic acid/methyl jasmonate accumulate in wounded soybean hypocotyls and modulate wound gene expression. Proc Natl Acad Sci 89:4938–4941
De Vos M, Van Oosten VR, Van Poecke RMP et al (2005) Signal signature and transcriptome changes of Arabidopsis during pathogen and insect attack. Mol Plant Microbe Interact 18:923–937. https://doi.org/10.1094/MPMI-18-0923
Delauney AJ, Verma DPS (1993) Proline biosynthesis and osmoregulation in plants. Plant J 4:215–223. https://doi.org/10.1046/j.1365-313X.1993.04020215.x
Domec J-C, Rivera LN, King JS et al (2013) Hemlock woolly adelgid (Adelges tsugae) infestation affects water and carbon relations of eastern hemlock (Tsuga canadensis) and Carolina hemlock (Tsuga caroliniana). New Phytol 199:452–463. https://doi.org/10.1111/nph.12263
Ellison AM, Bank MS, Clinton BD, et al (2005) Loss of foundation species: consequences for the structure and dynamics of forested ecosystems. Front Ecol Environ 3:479–486. https://doi.org/10.1890/1540-9295(2005)003[0479:lofscf]2.0.co;2
Erb M, Robert CAM, Hibbard BE, Turlings TCJ (2011) Sequence of arrival determines plant-mediated interactions between herbivores. J Ecol 99:7–15. https://doi.org/10.1111/j.1365-2745.2010.01757.x
Erbilgin N, Krokene P, Christiansen E et al (2006) Exogenous application of methyl jasmonate elicits defenses in Norway spruce (Picea abies) and reduces host colonization by the bark beetle Ips typographus. Oecologia 148:426–436. https://doi.org/10.1007/s00442-006-0394-3
Gandhi KJK, Herms DA (2010) Direct and indirect effects of alien insect herbivores on ecological processes and interactions in forests of eastern North America. Biol Invasions 12:389–405. https://doi.org/10.1007/s10530-009-9627-9
Gómez S, Orians CM, Preisser EL (2012) Exotic herbivores on a shared native host: tissue quality after individual, simultaneous, and sequential attack. Oecologia 169:1015–1024. https://doi.org/10.1007/s00442-012-2267-2
Gómez S, Gonda-King L, Orians CM, Preisser EL (2014) Competitor avoidance drives within-host feeding site selection in a passively dispersed herbivore. Ecol Entomol 39:10–16. https://doi.org/10.1111/een.12059
Gonda-King L, Radville L, Preisser EL (2012) False ring formation in eastern hemlock branches: impacts of hemlock woolly adelgid and elongate hemlock scale. Environ Entomol 41:523–531. https://doi.org/10.1603/EN11227
Gonda-King L, Gómez S, Martin JL et al (2014) Tree responses to an invasive sap-feeding insect. Plant Ecol 215:297–304. https://doi.org/10.1007/s11258-014-0298-y
Hadley JL, Schedlbauer JL (2002) Carbon exchange of an old-growth eastern hemlock (Tsuga canadensis) forest in central New England. Tree Physiol 22:1079–1092. https://doi.org/10.193/treephys/22.15-16.1079
Halitschke R, Schittko U, Pohnert G et al (2001) Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. III. Fatty acid-amino acid conjugates in herbivore oral secretions are necessary and sufficient for herbivore-specific plant responses. Plant Physiol 125:711–717. https://doi.org/10.1104/pp.125.2.711
Halitschke R, Gase K, Hui D et al (2003) Molecular interactions between the specialist herbi vore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata. VI. Microarray analysis reveals that most herbivore-specific transcriptional changes are mediated by fatty acid-amino acid conjugates. Plant Physiol 131:1894–1902. https://doi.org/10.1104/pp.102.018184
Heijari J, Nerg A-M, Kainulainen P et al (2005) Application of methyl jasmonate reduces growth but increases chemical defence and resistance against Hylobius abietis in scots pine seedlings. Entomol Exp Appl 115:117–124. https://doi.org/10.1111/j.1570-7458.2005.00263.x
Hillwig MS, Chiozza M, Casteel CL et al (2016) Abscisic acid deficiency increases defence responses against Myzus persicae in Arabidopsis. Mol Plant Pathol 17:225–235. https://doi.org/10.1111/mpp.12274
Hogenhout SA, Bos JIB (2011) Effector proteins that modulate plant-insect interactions. Curr Op Plant Bio 14:422–428. https://doi.org/10.1016/j.pbi.2011.05.003
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Ann Rev Plant Bio 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Jenkins JC, Aber JD, Canham CD (1999) Hemlock woolly adelgid impacts on community structure and N cycling rates in eastern hemlock forests. Can J For Res 29:630–645. https://doi.org/10.1139/x99-034
Kaloshian I, Walling LL (2005) Hemipterans as plant pathogens. Ann Rev Phytopath 43:491–521. https://doi.org/10.1146/annurev.phyto.43.040204.135944
Karban R, Baldwin IT (2007) Induced responses to herbivory. University of Chicago Press, Chicago
Kawazu K, Mochizuki A, Sato Y et al (2012) Different expression profiles of jasmonic acid and salicylic acid inducible genes in the tomato plant against herbivores with various feeding modes. Arthropod-Plant Interact 6:221–230. https://doi.org/10.1007/s11829-011-9174-z
Krokene P, Nagy NE, Solheim H (2008) Methyl jasmonate and oxalic acid treatment of Norway spruce: anatomically based defense responses and increased resistance against fungal infection. Tree Physiol 28:29–35
Martin DM, Gershenzon J, Bohlmann J (2003) Induction of volatile terpene biosynthesis and diurnal emission by methyl jasmonate in foliage of Norway spruce. Plant Physiol 132:1586–1599. https://doi.org/10.1104/pp.103.021196
McClure MS (1980) Foliar nitrogen: a basis for host suitability for elongate hemlock scale, Fiorinia externa (Homoptera: Diaspididae). Ecology 61:72–79. https://doi.org/10.2307/1937157
McClure MS (1991) Density-dependent feedback and population cycles in Adelges tsugae (Homoptera: Adelgidae) on Tsuga canadensis. Environ Entomol 20:258–264. https://doi.org/10.1093/ee/20.1.258
McClure M (2002) The elongate hemlock scale, Fiorinia externa Ferris (Homoptera: Diaspididae): a new look at an old nemesis. Proceedings, Hemlock Woolly Adelgid in the Eastern United States. USDA Forest Serv., East Brunswick, NJ. pp 248–253
Miller B, Madilao LL, Ralph S, Bohlmann J (2005) Insect-induced conifer defense. White pine weevil and methyl jasmonate induce traumatic resinosis, de novo formed volatile emissions, and accumulation of terpenoid synthase and putative octadecanoid pathway transcripts in sitka spruce. Plant Physiol 137:369–382. https://doi.org/10.1104/pp.104.050187
Miller-Pierce MR, Preisser EL (2012) Asymmetric priority effects influence the success of invasive forest insects. Ecol Entomol 37:350–358. https://doi.org/10.1111/j.1365-2311.2012.01371.x
Moran PJ, Thompson GA (2001) Molecular responses to aphid feeding in Arabidopsis in relation to plant defense pathways. Plant Physiol 125:1074–1085. https://doi.org/10.1104/pp.125.2.1074
Moreira X, Sampedro L, Zas R (2009) Defensive responses of Pinus pinaster seedlings to exogenous application of methyl jasmonate: concentration effect and systemic response. Environ Exp Bot 67:94–100. https://doi.org/10.1016/j.envexpbot.2009.05.015
Moreira X, Zas R, Sampedro L (2012) Methyl jasmonate as chemical elicitor of induced responses and anti-herbivory resistance in young conifer trees. In: Mérillon JM, Ramawat KG (eds) Plant defence: biological control. Springer, Netherlands, pp 345–362
Moreira X, Mooney KA, Rasmann S et al (2014) Trade-offs between constitutive and induced defences drive geographical and climatic clines in pine chemical defences. Ecol Lett 17:537–546. https://doi.org/10.1111/ele.12253
Naeem ul Hassan M, Zainal Z, Ismail I (2015) Green leaf volatiles: biosynthesis, biological functions and their applications in biotechnology. Plant Biotech J 13:727–739. https://doi.org/10.1111/pbi.12368
Nelson LA, Dillaway DN, Rieske LK (2014) Effect of an exotic herbivore, Adelges tsugae, on photosynthesis of a highly susceptible Tsuga host, with notes on conspecifics. Arthropod-Plant Interact 8:9–15. https://doi.org/10.1007/s11829-013-9285-9
Paradis AF (2011) Population dynamics of the hemlock woolly adelgid (Hemiptera: Adelgidae). Ph.D., University of Massachusetts Amherst
Park S-W, Kaimoyo E, Kumar D et al (2007) Methyl salicylate is a critical mobile signal for plant systemic acquired resistance. Science 318:113–116. https://doi.org/10.1126/science.1147113
Pezet J, Elkinton J, Gomez S et al (2013) Hemlock woolly adelgid and elongate hemlock scale induce changes in foliar and twig volatiles of eastern hemlock. J Chem Ecol 39:1090–1100. https://doi.org/10.1007/s10886-013-0300-5
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2014) nlme: linear and nonlinear mixed effects models. R package version 3.1-118. http://CRAN.R-project.org/package=nlme
Preisser EL, Elkinton JS (2008) Exploitative competition between invasive herbivores benefits a native host plant. Ecology 89:2671–2677. https://doi.org/10.1890/08-0299.1
Preisser EL, Elkinton JS, Abell K (2008) Evolution of increased cold tolerance during range expansion of the elongate hemlock scale Fiorinia externa Ferris (Hemiptera: Diaspididae). Ecol Entomol 33:709–715. https://doi.org/10.1111/j.1365-2311.2008.01021.x
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Radville L, Chaves A, Preisser EL (2011) Variation in plant defense against invasive herbivores: evidence for a hypersensitive response in eastern hemlocks (Tsuga canadensis). J Chem Ecol 37:592–597. https://doi.org/10.1007/s10886-011-9962-z
Rasmann S, Vos MD, Casteel CL et al (2012) Herbivory in the previous generation primes plants for enhanced insect resistance. Plant Physiol 158:854–863. https://doi.org/10.1104/pp.111.187831
Raymer PCL, Orwig DA, Finzi AC (2013) Hemlock loss due to the hemlock woolly adelgid does not affect ecosystem C storage but alters its distribution. Ecosphere 4:1–16. https://doi.org/10.1890/ES12-00362.1
Sackett TE, Record S, Bewick S et al (2011) Response of macroarthropod assemblages to the loss of hemlock (Tsuga canadensis), a foundation species. Ecosphere 2:1–16. https://doi.org/10.1890/ES11-00155.1
Sampedro L, Moreira X, Zas R (2011) Resistance and response of Pinus pinaster seedlings to Hylobius abietis after induction with methyl jasmonate. Plant Ecol 212:397–401. https://doi.org/10.1007/s11258-010-9830-x
Sasscer ER (1912) The genus Fiorinia in the United States. No. 16. US Government Printing Office
Shulaev V, Silverman P, Raskin I (1997) Airborne signalling by methyl salicylate in plant pathogen resistance. Nature 385:718–721. https://doi.org/10.1038/385718a0
Smith CM, Boyko EV (2007) The molecular bases of plant resistance and defense responses to aphid feeding: current status. Entomol Exp Appl 122:1–16. https://doi.org/10.1111/j.1570-7458.2006.00503.x
Soltis NE, Gómez S, Gonda-King L et al (2015) Contrasting effects of two exotic invasive hemipterans on whole-plant resource allocation in a declining conifer. Entomol Exp Appl 157:86–97. https://doi.org/10.1111/eea.12343
Souto D, Luther T, Chianese B (1996) Past and current status of HWA in eastern and Carolina hemlock stands. Proceedings of the first hemlock woolly adelgid review. Charlottesville, Virginia, USA. 12 October 1995. pp 9–15
Studham ME, MacIntosh GC (2012) Multiple phytohormone signals control the transcriptional response to soybean aphid infestation in susceptible and resistant soybean plants. Mol Plant Microbe Interact 26:116–129. https://doi.org/10.1094/MPMI-05-12-0124-FI
Thaler JS, Humphrey PT, Whiteman NK (2012) Evolution of jasmonate and salicylate signal crosstalk. Trends Plant Sci 17:260–270. https://doi.org/10.1016/j.tplants.2012.02.010
Underwood NC (1998) The timing of induced resistance and induced susceptibility in the soybean-Mexican bean beetle system. Oecologia 114:376–381. https://doi.org/10.1007/s004420050460
von Dahl CC, Baldwin IT (2007) Deciphering the role of ethylene in plant-herbivore interactions. J Plant Growth Regul 26:201–209
von Dohlen CD, Spaulding U, Shields K et al (2013) Diversity of proteobacterial endosymbionts in hemlock woolly adelgid (Adelges tsugae) (Hemiptera: Adelgidae) from its native and introduced range. Environ Microbiol 15:2043–2062. https://doi.org/10.1111/1462-2920.12102
Walling LL (2000) The myriad plant responses to herbivores. J Plant Growth Regul 19:195–216. https://doi.org/10.1007/s003440000026
Ward JS, Montgomery ME, Cheah CAS-J, et al (2004) Eastern Hemlock Forests: guidelines to minimize the impacts of hemlock woolly adelgid
Wilson CM, Vendettuoli JF, Orwig DA, Preisser EL (2016) Impact of an invasive insect and plant defense on a native forest defoliator. Insects 7:45. https://doi.org/10.3390/insects7030045
Wu J, Bladwin IT (2010) New insights into plant responses to the attack from insect herbivores. Ann Rev Genet 44:1–24. https://doi.org/10.1146/annurev-genet-102209-163500
Young RF, Shields KS, Berlyn GP (1995) Hemlock woolly adelgid (Homoptera: Adelgidae): stylet bundle insertion and feeding sites. Ann Entomol Soc Am 88:827–835. https://doi.org/10.1093/aesa/88.6.827
Zeneli G, Krokene P, Christiansen E et al (2006) Methyl jasmonate treatment of mature Norway spruce (Picea abies) trees increases the accumulation of terpenoid resin components and protects against infection by Ceratocystis polonica, a bark beetle-associated fungus. Tree Physiol 26:977–988. https://doi.org/10.1093/treephys/26.8.977
Zvereva EL, Lanta V, Kozlov MV (2010) Effects of sap-feeding insect herbivores on growth and reproduction of woody plants: a meta-analysis of experimental studies. Oecologia 163:949–960. https://doi.org/10.1007/s00442-010-1633-1
Acknowledgements
We are especially grateful to K. Boroczky, A. Agrawal, and the Chemical Ecology Core Facility at Cornell University for assistance with analyses of phytohormones. We also thank P. Candelas and K. Pieper for laboratory and field assistance, and C. Blubaugh, C. Rigsby, R. Schweiger, and two anonymous reviewers for helpful comments on an earlier draft of this manuscript. This project was funded by the National Science Foundation, grants NSF-DEB 1256826 to CMO and NSF-DEB 1256769 to ELP and CST. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the National Science Foundation.
Author information
Authors and Affiliations
Contributions
RNS, CST, ELP, and CMO conceived and designed the experiments, RNS and ZW performed the experiments, and RNS analyzed data and wrote the first draft of the manuscript. All authors contributed to revisions.
Corresponding author
Additional information
Communicated by Evan H. DeLucia.
Rights and permissions
About this article
Cite this article
Schaeffer, R.N., Wang, Z., Thornber, C.S. et al. Two invasive herbivores on a shared host: patterns and consequences of phytohormone induction. Oecologia 186, 973–982 (2018). https://doi.org/10.1007/s00442-018-4063-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4063-0