Abstract
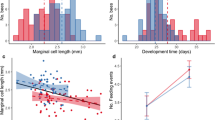
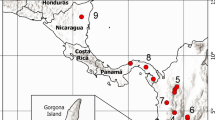
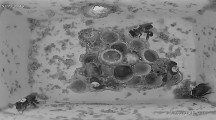
Maternal effects are widespread and influence a variety of traits, for example, life history strategies, mate choice, and capacity to avoid predation. Therefore, maternal effects may also influence phenotypic plasticity of offspring, but few studies have addressed the relationship between maternal effects and phenotypic plasticity of offspring. We examined the relationship between a maternally influenced trait (egg size) and the phenotypic plasticity of the induction rate of the broad-headed morph in the salamander Hynobius retardatus. The relationship between egg size and the induction of the broad-headed morph was tested across experimental crowding conditions (densities of low conspecifics, high conspecifics, and high heterospecific anuran), using eggs and larvae from eight natural populations with different larval densities of conspecifics and heterospecifics. The broad-headed morph has a large mouth that enables it to consume either conspecifics or heterospecifics, and this ability gives survival advantages over the normal morph. We have determined that there is phenotypic plasticity in development, as shown by an increase in the frequency of broad-headed morph in response to an increase in the density of conspecifics and heterospecifics. This reaction norm differed between populations. We also determined that the frequency of the broad-headed morph is affected by egg size in which larger egg size resulted in expression of the broad-headed morph. Furthermore, we determined that selection acting on the propensity to develop the broad-headed morph has produced a change in egg size. Lastly, we found that an increase in egg size alters the reaction norm to favor development of the broad-headed morph. For example, an equal change in experimental density produces a greater change in the frequency of the broad-headed morph in larvae developing from large eggs than it does in larvae developing from small eggs. Population differences in plasticity might be the results of differences in egg size between populations, which is caused by the adaptive integration of the plasticity and egg size. Phenotypic plasticity can not evolve independently of maternal effects.



Similar content being viewed by others
References
Agrawal AA, Laforsch C, Tollrian R (1999) Transgenerational induction of defenses in animals and plants. Nature 401:60–63
Altwegg R, Reyer HU (2002) Patterns of natural selection on size at metamorphosis in water frogs. Evolution 57:872–882
Bernardo J (1996) Maternal effects in animal ecology. Am Zool 36:83–105
Berven KA (1990) Factors affecting fluctuations in larval and adult stages of the wood frog (Rana sylvatica). Ecology 71:1599–1608
Denoel M, Whiteman HH, Wissinger SA (2007) Temporal shift of diet in alternative cannibalistic morphs of the tiger salamander. Biol J Linn Soc 89:373–382
Doughty P (2002) Coevolution of developmental plasticity and large egg size in Crinia georgiana tadpoles. Copeia 2002:928–937
Einum S, Fleming JA (1999) Maternal effects of egg size in brown trout (Salmo trutta): norms of reaction to environmental quality. Proc R Soc Lond B 266:2095–2100
Fleming IA, Gross MR (1990) Latitudinal clines: a trade-off between egg number and size in Pacific salmon. Ecology 71:1–11
Fox CW, Thakar MS, Mousseau TA (1997) Egg size plasticity in a seed beetle: an adaptive maternal effect. Am Nat 149:149–163
Fox CW, Czesak ME, Mousseau TA, Roff DA (1999) The evolutionary genetics of an adaptive maternal effect: egg size plasticity in a seed beetle. Evolution 53:552–560
Gilbert SF (2003) Developmental biology, 7th edn. Sinauer, Sunderland
Kaplan RH (1998) Maternal effects, developmental plasticity, and life history evolution. In: Mousseau TA, Fox CW (eds) Maternal effects as adaptations. Oxford University Press, New York, pp 244–260
Kaplan RH, Phillips PC (2006) Ecological and developmental context of natural selection: maternal effects and thermally induced plasticity in the frog Bombina orientalis. Evolution 60:142–156
Lande R (1979) Quantitative genetics analysis of multivariate evolution, applied to brain: body size allometry. Evolution 33:402–416
Lannoo MJ, Bachmann MD (1984) Aspects of cannibalistic morphs in a population of Ambystoma t. tigrinum larvae. Am Mid Nat 112:103–109
Laurila A, Karttunen S, Merila J (2002) Adaptive phenotypic plasticity and genetics of larval life histories in two Rana temporaria populations. Evolution 56:617–627
Matsushima N, Kawata M (2005) The choice of oviposition site and the effects of density and oviposition timing on survivorship in Rana japonica. Ecol Res 20:81–86
McGinley MA, Temme DH, Geber MA (1987) Parental investment in offspring in variable environments: theoretical and empirical considerations. Am Nat 130:370–398
Michimae H (2006) Differentiated phenotypic plasticity in larvae of the cannibalistic salamander Hynobius retardatus. Behav Ecol Sociobiol 60:205–211
Michimae H (2007) Differentiated egg size of the cannibalistic salamander Hynobius retardatus. J Ethol 25:153–158
Michimae H, Wakahara M (2002) A tadpole-induced polyphenism in the salamander Hynobius retardatus. Evolution 56:2029–2038
Mitchell SE, Read AF (2005) Poor maternal environment enhances offspring disease resistance in an invertebrate. Proc R Soc Lond B 272:2601–2607
Moran NA (1992) The evolutionary maintenance of alternative phenotypes. Am Nat 139:971–989
Mousseau TA, Fox CW (1998) Maternal effects as adaptations. Oxford University Press, New York
Nishihara-Takahashi A (1999) Faster growth of head size of pre-feeding larvae in a cannibalistic population of the salamander Hynobius retardatus. Zool Sci 16:303–307
Nussey DH, Clutton-Brock TH, Elston DA, Albon SD, Kruuk LB (2005) Phenotypic plasticity in a maternal trait in red deer. J Anim Ecol 74:387–396
Pfennig DW, Murphy PJ (2000) Character displacement in polyphenic tadpoles. Evolution 54:1738–1749
Price T (1998) Maternal and paternal effects in birds: effects on offspring fitness. In: Mousseau TA, Fox CW (eds) Maternal effects as adaptations. Oxford University Press, New York, pp 202–226
Royle NJ, Surai PF, Hartley IR (2001) Maternally derived androgens and antioxidants in bird eggs: complementary but opposing effects? Behav Ecol 12:381–385
Scott DE (1990) Effects of larval density in Ambystoma opacum: an experiment in large-scale field enclosures. Ecology 71:296–306
Scott DE (1994) The effect of larval density on adult demographic traits in Ambystoma opacum. Ecology 75:1383–1396
Semlitsch RD, Scott DE, Pechmannn JHK (1988) Time and size at metamorphosis related to adult fitness in Ambystoma talpoideum. Ecology 71:1789–1795
Sinervo B, Doughty P (1996) Interactive effects of offspring size and timing of reproduction on offspring reproduction: experimental, maternal, and quantitative genetic aspects. Evolution 50:1314–1327
Smith DC (1987) Adult recruitment in chorus frogs: effects of size and date at metamorphosis. Ecology 68:344–350
Smith CC, Fretwell SC (1974) The optimal balance between size and number of offspring. Am Nat 108:409–506
Stephen MR, George VL, James PC (1992) Performance consequences of a trophic polymorphism: feeding behavior in typical and cannibal phenotypes of Ambystoma tigrinum. Copeia 1992:672–679
Trussell GC, Nicklin MO (2002) Cue sensitivity, inducible defense, and trade-offs in a marine snail. Ecology 83:1635–1647
Wakahara M (1997) Kin recognition among intact and blinded, mixed-sibling larvae of a cannibalistic salamander Hynobius retardatus. Zool Sci 14:893–899
Whiteman HH, Sheen JP, Johnson EB, VanDeusen A, Gargille R, Sacco TW (2003) Heterospecific prey and trophic polyphenism in larval tiger salamanders. Copeia 2003:56–67
Acknowledgments
We are grateful for the comments and suggestions of Ross Alford, Paul Doughty and anonymous reviewers on earlier versions of this manuscript. This work was supported by a Grant-in-Aid for Scientific Research (No 15009850) from the Japan Society for the Promotion of Science.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Ross Alford.
Rights and permissions
About this article
Cite this article
Michimae, H., Nishimura, K., Tamori, Y. et al. Maternal effects on phenotypic plasticity in larvae of the salamander Hynobius retardatus . Oecologia 160, 601–608 (2009). https://doi.org/10.1007/s00442-009-1319-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-009-1319-8