Abstract
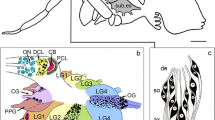
Octopamine is an important neuromodulator in the insect nervous system, influencing memory formation, sensory perception and motor control. In this study, we compare the distribution of octopamine-like immunoreactive neurons in two parasitic wasp species of the Nasonia genus, N. vitripennis and N. giraulti. These two species were previously described as differing in their learning and memory formation, which raised the question as to whether morphological differences in octopaminergic neurons underpinned these variations. Immunohistochemistry in combination with confocal laser scanning microscopy was used to reveal and compare the somata and major projections of the octopaminergic neurons in these wasps. The brains of both species showed similar staining patterns, with six different neuron clusters being identified in the brain and five different clusters in the subesophageal ganglion. Of those clusters found in the subesophageal ganglion, three contained unpaired neurons, whereas the other three consisted in paired neurons. The overall pattern of octopaminergic neurons in both species was similar, with no differences in the numbers or projections of the ventral unpaired median (VUM) neurons, which are known to be involved in memory formation in insects. In one other cluster in the brain, located in-between the optic lobe and the antennal lobe, we detected more neurons in N. vitripennis compared with N. giraulti. Combining our results with findings made previously in other Hymenopteran species, we discuss possible functions and some of the ultimate factors influencing the evolution of the octopaminergic system in the insect brain.





Similar content being viewed by others
References
Agarwal M, Giannoni Guzmán M, Morales-Matos C, Del Valle Díaz RA, Abramson CI, Giray T (2011) Dopamine and octopamine influence avoidance learning of honey bees in a place preference assay. PLoS ONE 6:e25371
Berg M van den, Duivenvoorde L, Wang GH, Tribuhl S, Bukovinszky T, Vet LEM, Dicke M, Smid HM (2011) Natural variation in learning and memory dynamics studied by artificial selection on learning rate in parasitic wasps. Anim Behav 81:325–333
Bleeker MAK, Van der Zee B, Smid HM (2006) Octopamine-like immunoreactivity in the brain and suboesophageal ganglion of two parasitic wasps, Cotesia glomerata and Cotesia rubecula. Anim Biol 56:247–257
Brandt R, Rohlfing T, Rybak J, Krofczik S, Maye A, Westerhoff M, Hege HC, Menzel R (2005) Three-dimensional average-shape atlas of the honeybee brain and its applications. J Comp Neurol 492:1–19
Bräunig P (1991) Suboesophageal DUM neurons innervate the principal neuropiles of the locust brain. Philos Trans R Soc Lond B Biol Sci 332:221–240
Burke CJ, Huetteroth W, Owald D, Perisse E, Krashes MJ, Das G, Gohl D, Silies M, Certel S, Waddell S (2012) Layered reward signalling through octopamine and dopamine in Drosophila. Nature 492:433–437
Busch S, Selcho M, Ito K, Tanimoto H (2009) A map of octopaminergic neurons in the Drosophila brain. J Comp Neurol 513:643–667
Cardona A, Saalfeld S, Schindelin J, Arganda-Carreras I, Preibisch S, Longair M, Tomancak P, Hartenstein V, Douglas RJ (2012) TrakEM2 software for neural circuit reconstruction. PLoS ONE 7:e38011
Dacks AM, Christensen TA, Agricola HJ, Wollweber L, Hildebrand JG (2005) Octopamine-immunoreactive neurons in the brain and subesophageal ganglion of the hawkmoth Manduca sexta. J Comp Neurol 488:255–268
Darling DC, Werren JH (1990) Biosystematics of Nasonia (Hymenoptera: pteromalidae): two new species reared from birds nests in North America. Ann Ent Soc Am 83:352–370
Desjardins CA, Perfectti F, Bartos JD, Enders LS, Werren JH (2010) The genetic basis of interspecies host preference differences in the model parasitoid Nasonia. Heredity 104:270–277
Eckert M, Rapus J, Nurnberger A, Penzlin H (1992) The octopaminergic system within the ventral nerve cord of the American cockroach. Acta Biol Hung 43:201–211
Evans PD (1980) Biogenic amines in the insect nervous system. Adv Insect Physiol 15:317–473
Farooqui T (2007) Octopamine-mediated neuromodulation of insect senses. Neurochem Res 32:1511–1529
Farooqui T (2012) Review of octopamine in insect nervous systems. Open Access Insect Physiol 4:1–17
Farooqui T, Robinson K, Vaessin H, Smith BH (2003) Modulation of early olfactory processing by an octopaminergic reinforcement pathway in the honeybee. J Neurosci 23:5370–5380
Farris SM, Schulmeister S (2011) Parasitoidism, not sociality, is associated with the evolution of elaborate mushroom bodies in the brains of hymenopteran insects. Proc Biol Sci 278:940–951
Giurfa M (2006) Associative learning: the instructive function of biogenic amines. Curr Biol 16:R892–R895
Hammer M (1993) An identified neuron mediates the unconditioned stimulus in associative olfactory learning in honeybees. Nature 366:59–63
Hammer M (1997) The neural basis of associative reward learning in honeybees. Trends Neurosci 20:245–252
Hammer M, Menzel R (1998) Multiple sites of associative odor learning as revealed by local brain microinjections of octopamine in honeybees. Learn Mem 5:146–156
Hoedjes KM, Smid HM (2014) Natural variation in long-term memory formation among Nasonia parasitic wasp species. Behav Process 105:40–45
Hoedjes KM, Kruidhof HM, Huigens ME, Dicke M, Vet LE, Smid HM (2011) Natural variation in learning rate and memory dynamics in parasitoid wasps: opportunities for converging ecology and neuroscience. Proc Biol Sci 278:889–897
Hoedjes KM, Steidle JL, Werren JH, Vet LE, Smid HM (2012) High-throughput olfactory conditioning and memory retention test show variation in Nasonia parasitic wasps. Genes Brain Behav 11:879–887
Hoedjes KH, Smid HM, Vet LEM, Werren JH (2014) Introgression study reveals two quantitative trait loci involved in interspecific variation in memory retention among Nasonia wasp species. Heredity (in press)
Homberg U, Seyfarth J, Binkle U, Monastirioti M, Alkema MJ (2013) Identification of distinct tyraminergic and octopaminergic neurons innervating the central complex of the desert locust, Schistocerca gregaria. J Comp Neurol 521:2025–2041
Jefferis GS, Potter CJ, Chan AM, Marin EC, Rohlfing T, Maurer CR, Luo L (2007) Comprehensive maps of Drosophila higher olfactory centers: spatially segregated fruit and pheromone representation. Cell 128:1187–1203
Klappenbach M, Kaczer L, Locatelli F (2013) Dopamine interferes with appetitive long-term memory formation in honey bees. Neurobiol Learn Mem 106:230–237
Konings PN, Vullings HG, Geffard M, Buijs RM, Diederen JH, Jansen WF (1988) Immunocytochemical demonstration of octopamine-immunoreactive cells in the nervous system of Locusta migratoria and Schistocerca gregaria. Cell Tissue Res 251:371–379
Koon AC, Ashley J, Barria R, DasGupta S, Brain R, Waddell S, Alkema MJ, Budnik V (2011) Autoregulatory and paracrine control of synaptic and behavioral plasticity by octopaminergic signaling. Nat Neurosci 14:190–199
Krashes MJ, DasGupta S, Vreede A, White B, Armstrong JD, Waddell S (2009) A neural circuit mechanism integrating motivational state with memory expression in Drosophila. Cell 139:416–427
Kreissl S, Eichmuller S, Bicker G, Rapus J, Eckert M (1994) Octopamine-like immunoreactivity in the brain and subesophageal ganglion of the honeybee. Comp Neurol 348:583–595
Kruidhof HM, Rijk M de, Hoffmann D, Harvey JA, Vet LEM, Soler R (2013) Effect of belowground herbivory on parasitoid associative learning of plant odours. Oikos 122:1094–1100
Liu C, Plaçais PY, Yamagata N, Pfeiffer BD, Aso Y, Friedrich AB, Siwanowicz I, Rubin GM, Preat T, Tanimoto H (2012) A subset of dopamine neurons signals reward for odour memory in Drosophila. Nature 488:512–516
Lynch JA, Desplan C (2006) A method for parental RNA interference in the wasp Nasonia vitripennis. Nat Protocol 1:486–494
Menzel R (2012) The honeybee as a model for understanding the basis of cognition. Nat Rev Neurosci 13:758–768
Monastirioti M, Gorczyca M, Rapus J, Eckert M, White K, Budnik V (1995) Octopamine immunoreactivity in the fruit fly Drosophila melanogaster. J Comp Neurol 356:275–287
Monastirioti M, Linn CEJ, White K (1996) Characterization of Drosophila tyramine beta-hydroxylase gene and isolation of mutant flies lacking octopamine. J Neurosci 16:3900–3911
Mons N, Geffard M (1987) Specific antisera against the catecholamines—L-3,4-dihydroxyphenylalanine, dopamine, noradrenaline, and octopamine tested by an enzyme-linked-immunosorbent-assay. J Neurochem 48:1826–1833
Niehuis O, Gibson JD, Rosenberg MS, Pannebakker BA, Koevoets T, Judson AK, Desjardins CA, Kennedy K, Duggan D, Beukeboom LW, Zande L van de, Shuker DM, Werren JH, Gadau J (2010) Recombination and its impact on the genome of the haplodiploid parasitoid wasp Nasonia. PLoS ONE 5:e8597
Pannebakker BA, Niehuis O, Hedley A, Gadau J, Shuker DM (2010) The distribution of microsatellites in the Nasonia parasitoid wasp genome. Ins Mol Biol 19:91–98
Papaj DR, Vet LEM (1990) Odor learning and foraging success in the parasitoid, Leptopilina heterotoma. J Chem Ecol 16:3137–3150
Perry CJ, Barron AB (2013) Neural mechanisms of reward in insects. Annu Rev Entomol 58:543–562
Peters RS (2010) Host range and offspring quantities in natural populations of Nasonia vitripennis (Walker, 1836) (Hymenoptera: Chalcidoidea: Pteromalidae). J Hymenopteran Res 19:128–138
Pflüger H-J, Stevenson PA (2005) Evolutionary aspects of octopaminergic systems with emphasis on arthropods. Arthropod Struct Dev 34:379–396
Rehder V (1989) Sensory pathways and motoneurons of the proboscis reflex in the subesophageal ganglion of the honey bee. J Comp Neurol 279:499–513
Roeder T (1999) Octopamine in invertebrates. Prog Neurobiol 59:533–561
Roeder T (2005) Tyramine and octopamine: ruling behavior and metabolism. Annu Rev Entomol 50:447–477
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9:676–682
Schröter U, Malun D, Menzel R (2007) Innervation pattern of suboesophageal ventral unpaired median neurones in the honeybee brain. Cell Tissue Res 327:647–667
Schurmann D, Sommer C, Schinko APB, Greschista M, Smid H, Steidle JLM (2012) Demonstration of long-term memory in the parasitic wasp Nasonia vitripennis. Entomol Exp Appl 143:199–206
Schwaerzel M, Monastirioti M, Scholz H, Friggi Grelin F, Birman S, Heisenberg M (2003) Dopamine and octopamine differentiate between aversive and appetitive olfactory memories in Drosophila. J Neurosci 23:10495–10502
Sinakevitch I, Strausfeld NJ (2006) Comparison of octopamine-like immunoreactivity in the brains of the fruit fly and blow fly. J Comp Neurol 494:460–475
Sinakevitch I, Niwa M, Strausfeld NJ (2005) Octopamine-like immunoreactivity in the honey bee and cockroach: comparable organization in the brain and subesophageal ganglion. J Comp Neurol 488:233–254
Smid HM (1998) Transfer of a male accessory gland peptide to the female during mating in Leptinotarsa decemlineata. Invertebr Reprod Dev 34:47–53
Smid HM, Bleeker MAK, Loon JJA van, Vet LEM (2003) Three-dimensional organization of the glomeruli in the antennal lobe of the parasitoid wasps Cotesia glomerata and C. rubecula. Cell Tissue Res 312:237–248
Smid HM, Wang G, Bukovinszky T, Steidle JL, Bleeker MA, Loon JJ van, Vet LE (2007) Species-specific acquisition and consolidation of long-term memory in parasitic wasps. Proc R Soc Lond B Biol Sci 274:1539–1546
Spivak M, Masterman R, Ross R, Mesce KA (2003) Hygienic behavior in the honey bee (Apis mellifera L.) and the modulatory role of octopamine. J Neurobiol 55:341–354
Spörhase-Eichmann U, Vullings HG, Buijs RM, Hörner M, Schürmann FW (1992) Octopamine-immunoreactive neurons in the central nervous system of the cricket, Gryllus bimaculatus. Cell Tissue Res 268:287–304
Stern M, Thompson KSJ, Zhou P, Watson DG, Midgley JM, Gewecke M, Bacon JP (1995) Octopaminergic neurons in the locust brain—morphological, biochemical and electrophysiological characterization of potential modulators of the visual-system. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 177:611–625
Stevenson PA, Spörhase-Eichmann U (1995) Localization of octopaminergic neurones in insects. Comp Biochem Physiol A Physiol 110:203–215
Suver MP, Mamiya A, Dickinson MH (2012) Octopamine neurons mediate flight-induced modulation of visual processing in Drosophila. Curr Biol 22:2294–2302
Tamo C, Ricard I, Held M, Davison AC, Turlings TCJ (2006) A comparison of naive and conditioned responses of three generalist endoparasitoids of lepidopteran larvae to host-induced plant odours. Anim Biol 56:205–220
Turlings TCJ, Wäckers FL, Vet LEM, Lewis WJ, Tumlinson JH (1993) Learning of host-finding cues by hymenopterous parasitoids. In: Papaj DR, Lewis AC (eds) Insect learning: ecological and evolutionary perspectives. Chapman & Hall, New York, pp 51–78
Unoki S, Matsumoto Y, Mizunami M (2005) Participation of octopaminergic reward system and dopaminergic punishment system in insect olfactory learning revealed by pharmacological study. Eur J Neurosci 22:1409–1416
Vergoz V, Roussel E, Sandoz JC, Giurfa M (2007) Aversive learning in honeybees revealed by the olfactory conditioning of the sting extension reflex. PLoS ONE 2:e288
Verlinden H, Vleugels R, Marchal E, Badisco L, Pflüger H-J, Blenau W, Broeck JV (2010) The role of octopamine in locusts and other arthropods. J Insect Physiol 56:854–867
Vet LEM, Lewis WJ, Cardé RT (1995) Parasitoid foraging and learning. In: Cardé RT, Bell WJ (eds) Chemical ecology of insects. Chapman & Hall, New York, pp 65-101
Waddell S (2013) Reinforcement signalling in Drosophila; dopamine does it all after all. Curr Opin Neurobiol 23:324–329
Werren JH, Loehlin DW (2009) The parasitoid wasp Nasonia: an emerging model system with haploid male genetics. Cold Spring Harb Protoc 2009:pdb.emo134
Werren JH, Richards S, Desjardins CA, Niehuis O, Gadau J, Colbourne JK, Beukeboom LW, Desplan C, Elsik CG et al (2010) Functional and evolutionary insights from the genomes of three parasitoid Nasonia species. Science 327:343–348
Wong AM, Wang JW, Axel R (2002) Spatial representation of the glomerular map in the Drosophila protocerebrum. Cell 109:229–241
Woude E van der, Smid HM, Chittka L, Huigens ME (2013) Breaking Haller’s rule: brain-body size isometry in a minute parasitic wasp. Brain Behav Evol 81:86–92
Wright GA, Mustard JA, Simcock NK, Ross-Taylor AA, McNicholas LD, Popescu A, Marion-Poll F (2010) Parallel reinforcement pathways for conditioned food aversions in the honeybee. Curr Biol 20:2234–2240
Wu CL, Shih MF, Lai JS, Yang HT, Turner GC, Chen L, Chiang AS (2011) Heterotypic gap junctions between two neurons in the Drosophila brain are critical for memory. Curr Biol 21:848–854
Wu CL, Shih MF, Lee PT, Chiang AS (2013) An octopamine-mushroom body circuit modulates the formation of anesthesia-resistant memory in Drosophila. Curr Biol 23:2346–2354
Acknowledgments
We are grateful to Jack Werren (University of Rochester, N.Y., USA) and Bart Pannebakker (Wageningen University, Genetics) for sending Nasonia lines, to Emma van der Woude and Mark Boumans (Wageningen University, Entomology) for preparing some of the confocal stacks and to Henk Schipper (Wageningen University, Animal Sciences) for use of the confocal laser scanning microscope.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haverkamp, A., Smid, H.M. Octopamine-like immunoreactive neurons in the brain and subesophageal ganglion of the parasitic wasps Nasonia vitripennis and N. giraulti . Cell Tissue Res 358, 313–329 (2014). https://doi.org/10.1007/s00441-014-1960-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-1960-3