Abstract
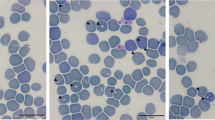
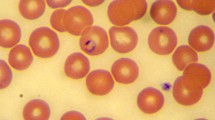
The growing proximity of wildlife to large urban niches arouses greater interest in understanding wild reservoirs in the epidemiology of diseases of importance to animal and human health. The aim of the present study was to investigate the presence of piroplasmids in opossums rescued from the metropolitan region of Rio de Janeiro state, Brazil. Blood and bone marrow samples were collected from 15 Didelphis aurita and subjected to DNA extraction and PCR using primers for the 18S rRNA, cox1, cox3, and hsp70 genes of piroplasmids. Clinical and hematological evaluation of the animals was also performed. Five (33.3%) of the 15 opossums tested positive for piroplasms in the nested PCR based on the 18S rRNA, and in two animals, it was possible to observe intra-erythrocytic structures compatible with merozoites. One of the positive animals showed clinical signs of infection such as jaundice, fever, and apathy. Anemia, low level of plasma protein, leukocytosis, and regenerative erythrocyte signs were observed in positive animals. Phylogenetic analysis based on both 18S rRNA and cox-3 genes demonstrated that the piroplasmids detected in D. aurita formed a unique sub-clade, albeit related to piroplasmids previously detected in Didelphis albiventris and associated ticks from Brazil. This study proposes the novel Piroplasmida Clade, namely “South American Marsupialia Group,” and reinforces the need for new clinical-epidemiological surveys to understand the dynamics of these infections in didelphids in Brazil.






Similar content being viewed by others
Data availability
The datasets supporting the conclusions of this article are included within the article. Raw data are available from the corresponding author.
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Alvarado-Rybak M, Solano-Gallego L, Millán J (2016) A review of piroplasmid infections in wild carnivores worldwide: importance for domestic animal health and wildlife conservation. Parasit Vectors 9:538. https://doi.org/10.1186/s13071-016-1808-7
André MR, Calchi AC, Perles L, Gonçalves LR, Uccella L, Lemes JRB et al (2022) Novel Ehrlichia and Hepatozoon genotypes in white-eared opossums (Didelphis albiventris) and associated ticks from Brazil. Ticks Tick Borne Dis 13(6):102022. https://doi.org/10.1016/j.ttbdis.2022.102022
Barbosa AD, Austen J, Portas TJ, Amigo JÁ, Ahlstrom LA, Oskam CL et al (2019) Sequence analyses at mitochondrial and nuclear loci reveal a novel Theileria sp and AID in the phylogenetic resolution of piroplasms from Australian marsupials and ticks. PloS one. 12:e0225822. https://doi.org/10.1371/journal.pone.0225822
Benson DA, Mizrachi IK, Lipman DJ, Ostell J, Rapp BA, Wheeler DI (2002) GenBank. Nucleic Acids Res 30:17–20. https://doi.org/10.1093/nar/28.1.15
Bezerra-santos MA, Ramos RAN, Campos K, Dantas-torres F, Otranto D (2021) Didelphis spp. opossums and their parasites in the Americas: a One Health perspective. Parasitol Res 120:4091–4111. https://doi.org/10.1007/s00436-021-07072-4
Bilić P, Kuleš J, Barić Rafaj R, Mrljak V (2018) Canine babesiosis: where do we stand. Acta Vet Scand 68:127–160. https://doi.org/10.2478/acve-2018-0011
Birkenheuer AJ, Neel J, Ruslander D, Levy MG, Breitschwerdt EB (2004) Detection and molecular characterization of a novel large Babesia species in a dog. Vet Parasitol 124:151–160. https://doi.org/10.1016/j.vetpar.2004.07.008
Bordes F, Morand S (2011) The impact of multiple infections on wild animal hosts: a review. Infect Ecol 1:7346. https://doi.org/10.3402/iee.v1i0.7346
Cáceres NC (2004) Diet of three didelphid marsupials (Mammalia: Didelphimorphia) in southern Brazil. Mamm 69:430–433. https://doi.org/10.1078/1616-5047-00165
Cardoso L, Cortes HCE, Reis A, Rodrigues P, Simoes M, Lopes AP et al (2013) Prevalence of Babesia microti-like infection in red foxes (Vulpes vulpes) from Portugal. Vet Parasitol 196(1–2):90–95. https://doi.org/10.1016/j.vetpar.2012.12.060
Casagrande RA, de Oliveira CM, Horta MC, Rossi S, Teixeira RH, Matushima ER (2009) Perfil hematológico de gambás Didelphis aurita e D. albiventris do Estado de Sao Paulo, Brasil. Acta Sci Biol 31(2):185–189. https://doi.org/10.4025/actascibiolsci.v31i2.7007
Clements WA (1968) Origin and early evolution of marsupials. Evolution 22:1–18. https://doi.org/10.1111/j.1558-5646.1968.tb03444.x
Colle AC, Mendonça RFB, Maia MO, Freitas LC, Witter RW, Marcili A et al (2019) Molecular survey of tick-borne pathogens in small mammals from Brazilian Amazonia. Rev Bras Parasitol Vet 28:592–604. https://doi.org/10.1590/S1984-29612019086
Corduneanu A, Hrazdilová K, Sándor AD, Matei IA, Ionică AM, Barti L et al (2017) Babesia vesperuginis, a neglected piroplasmid: new host and geographical records, and phylogenetic relations. Parasit Vectors 10:598. https://doi.org/10.1186/s13071-017-2536-3
Da Silva MR, Fornazari F, de Castro Demoner L, Teixeira CR, Langoni H, O’Dwyer LH (2017) Didelphis albiventris naturally infected with Hepatozoon canis in southeastern Brazil. Ticks and Tick-borne Dis 8(6):878–81. https://doi.org/10.1016/j.ttbdis.2017.07.005
Devictor V, Julliard R, Jiguet F (2008) Distribution of specialist and generalist species along spatial gradients of habitat disturbance and fragmentation. Oikos 117:507–514. https://doi.org/10.1111/j.0030-1299.2008.16215.x
Donahoe SL, Peacock CS, Choo AY, Cook RW, O’Donoghue P, Crameri S et al (2015) A retrospective study of Babesia macropus associated with morbidity and mortality in eastern grey kangaroos (Macropus giganteus) and agile wallabies (Macropus agilis). Int J Parasitol Parasites Wildl. 4(2):268–76. https://doi.org/10.1016/j.ijppaw.2015.02.002
Eiras DF, Craviotto MB, Baneth G, Moré G (2014) First report of Rangelia vitalii infection (canine rangeliosis) in Argentina. Parasitol Int 63(5):729–734. https://doi.org/10.1016/j.parint.2014.06.003
Ewing B, Green P (1998) Basecalling of automated sequencer traces using phred. II Error Probabilities. Genome Res 8:186–194. https://doi.org/10.1101/gr.8.3.186
Faria MB, Lanes RO, Bonvicino CR (2019) Marsupiais do Brasil: guia de identifcação com base em caracteres morfológicos externos e cranianos. Amélie Editorial, São Caetano do Sul
Fava N, Alves TS, Lopes MG, Labruna MB, Santos AQ, Cury MC (2022) Occurrence and molecular identification of hemoparasites in wild mammals kept in rehabilitation centers in Brazil. Acta Parasit 67(1):476–486. https://doi.org/10.1007/s11686-021-00492-3
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Furuta PI, Oliveira TMFS, Teixeira MCA, Rocha AG, Machado RZ, Tinuccicosta M (2009) Comparison between a soluble antigen-based ELISA and IFAT in detecting antibodies against Babesia canis in dogs. Rev Bras De Parasitol Vet 18:41–45. https://doi.org/10.4322/rbpv.01803007
Gonçalves LR, Herrera HM, Nantes WAG et al (2020) Genetic diversity and lack of molecular evidence for hemoplasma cross-species transmission between wild and synanthropic mammals from Central-Western Brazil. Acta Tropica. 203:105303. https://doi.org/10.1016/j.actatropica.2019.105303
Gonçalves LR, Paludo G, Bisol TB, Perles L, De Oliveira LB, De Oliveira CM et al (2021) Molecular detection of piroplasmids in synanthropic rodents, marsupials, and associated ticks from Brazil, with the phylogenetic inference of a putative novel Babesia sp. from white-eared opossum (Didelphis albiventris). Parasitol Res 120:3537–3546. https://doi.org/10.1007/s00436-021-07284-8
Greay TL, Zahedi A, Krige A, Owens JM, Rees RL, Ryan UM et al (2018) Endemic, exotic and novel apicomplexan parasites detected during a national study of ticks from companion animals in Australia. Parasite Vector 11:197. https://doi.org/10.1186/s13071-018-2775-y
Guimarães A, Raimundo JM, Silva ATD, Carpintero FM, Pires JR, Benevenute JL, et al (2018) Detection of aputative novel genotype of Ehrlichia sp. from opossums (Didelphis aurita) from Brazil. Rev Bras Parasitol Vet 28:140–144. https://doi.org/10.1590/S1984-296120180068
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98. https://d1wqtxts1xzle7.cloudfront.net/29520866. Accessed Dec 2022
Harris DJ (2016) Naming no names: Comments on the taxonomy of small piroplasmids in canids. Parasites & vectors 9(1):289. https://doi.org/10.1186/s13071-016-1567-5
Herrera L, Urdaneta-Morales S (1991) Didelphis marsupialis: reservoir of Babesia brasiliensis in the valley of Caracas (Venezuela). Acta Cient Venez 42(1):45–46
Hersh MH, Tibbetts M, Strauss M, Ostfeld RS, Keesing F (2012) Reservoir competence of wildlife host species for Babesia microti. Emerging Infect Dis 18(12):1951–1957. https://doi.org/10.3201/eid1812.111392
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23:254–267. https://doi.org/10.1093/molbev/msj030
Jalovecka M, Sojka D, Ascencio M, Schnittger L (2019) Babesia life cycle–when phylogeny meets biology. Trends Parasitol 13:356–368. https://doi.org/10.1016/j.pt.2019.01.007
Jefferies R, Ryan U, Jardine J, Broughton DK, Robertson ID, Irwin PJ (2007) Blood, bull terriers and babesiosis: further evidence for direct transmission of Babesia gibsoni in dogs. Aust Vet J 85:459–463. https://doi.org/10.1111/j.1751-0813.2007.00220.x
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1211. https://doi.org/10.1093/bib/bbx108
Lin M, Xiong Q, Chung M, Daugherty SC, Nagaraj S, Sengamalay N et al (2021) Comparative analysis of genome of Ehrlichia sp. HF, a model bacterium to study fatal human ehrlichiosis. BMC Genomics 22:11. https://doi.org/10.1186/s12864-020-07309-z
Kruse H, Kirkemo AM, Handeland K (2004) Wildlife as source of zoonotic infections. Emerg Infect Dis 10:2067. https://doi.org/10.3201/eid1012.040707
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Lau BT, Malkus P, Paulsson J (2013) New quantitative methods for measuring plasmid loss rates reveal unexpected stability. Plasmid 9:353–361. https://doi.org/10.1016/j.plasmid.2013.07.007
Leigh JW, Bryant D (2015) Popart: full-feature sofware for haplotype network construction. Methods Ecol Evol 6:1110–1116. https://doi.org/10.1111/2041-210X.12410
Lewis RE (1993) Notes on the geographical distribution and host preferences in the order Siphonaptera. Part 8. New taxa described between 1984 and 1990, with a current classification of the order. J. Med. Entomol. 30(1):239–256. https://doi.org/10.1093/jmedent/30.1.239
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinform 25:1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Malta MCC, Luppi MM (2006) Marsupialia–Didelphimorphia (Gambá, Cuíca). In: Cubas Z, Silva JCR, Catão-Dias JL (eds). Tratado de animais selvagens—Medicina Veterinária. São Paulo: Rocca 2006:340–357
Massini PF, Drozino RN, Otomura FH, Mongruel ACB, Valente JDM, Toledo MJO et al (2019) Detection of Hemotropic Mycoplasma sp. in white-eared opossums (Didelphis albiventris) from Southern Brazil. Rev Bras Parasitol Vet 28:797–801. https://doi.org/10.1590/S1984-29612019058
Oliveira ÁFX, Calchi AC, Mongruel ACB, Stocco AV, Stocco NV, Costa AC et al (2023) Molecular detection of hemoplasmas in rescued black-eared opossums (Didelphis aurita Wied-Neuwied, 1826) from southeastern Brazil, with evidence of a novel genotype infecting marsupials. Rev Bras Parasitol Vet 32(2):e016422. https://doi.org/10.1590/S1984-29612023015
Pitel PH, Pronost S, Scrive T, Léon A, Richard E, Fortier G (2010) Molecular detection of Theileria equi and Babesia caballi in the bone marrow of asymptomatic horses. Vet Parasitol 170:182–184. https://doi.org/10.1016/j.vetpar.2010.01.043
Pitel PH, Pronost S, Scrive T, Léon A, Richard E, Fortier G et al (2010) Molecular detection of Theileria equi and Babesia caballi in the bone marrow of asymptomatic horses. Vet Parasitol 170(1–2):182–184. https://doi.org/10.1016/j.vetpar.2010.01.043
Rojero-vazquez E, Gordillo-Pérez G, Weber M (2017) Infection of Anaplasma phagocytophilum and Ehrlichia spp. in opossums and dogs in Campeche, Mexico: the role of tick infestation. Front Ecol Evol 5:161. https://doi.org/10.3389/fevo.2017.00161
Sanger F, Air GM, Barrell BG, Brown NL, Coulson AR, Fiddes JC (1977) Nucleotide sequence of bacteriophage phiX174 DNA. Nature 265:687–695. https://doi.org/10.1038/265687a0
Schnittger L, Rodriguez AE, Florin-Christensen M, Morrison DA (2012) Babesia: a world emerging. Infect Genet Evol 21:1788–1809. https://doi.org/10.1016/j.meegid.2012.07.004
Schreeg ME, Marr HS, Tarigo JL, Cohn LA, Bird DM, Scholl EH et al (2016) Mitochondrial genome sequences and structures aid in the resolution of Piroplasmida phylogeny. PloS one 11:e0165702. https://doi.org/10.1371/journal.pone.0165702
Scheer S, Macedo MRP, Soares T de AL, Muller G. (2022) Análise molecular de Theileria equi e Babesia sp. (Piroplasmida) em Didelphis albiventris Lund, 1840, no sul do Rio Grande do Sul, Brasil. Braz J Dev 8(6):47182–47189. https://doi.org/10.34117/bjdv8n5-292
Serra Freire NM (1979) Babesia ernestoi sp. n. in Didelphis marsupialis L., 1758, and D. albiventris Lund, 1841, in Brazil. Zentralbl Veterinarmed B 26:614–622. https://doi.org/10.1111/j.1439-0450.1979.tb00855.x
Shaw SE, Birtles RJ, Day MJ (2001) Review: arthropod-transmitted infectious diseases of cats. J Feline Med Surg 3:193-209. https://doi.org/10.1053/jfms.2001.0149
Soares JF, Girotto A, Brandão PE, Da Silva AS, França RT, Lopes STA et al (2011) Detection and molecular characterization of a canine piroplasm from Brazil. Vet Parasitol 180:203–208. https://doi.org/10.1016/j.vetpar.2011.03.024
Soares HS, Marcili A, Barbieri AR, Minervino AH, Moreira TR, Gennari SM et al (2017) Novel piroplasmid and Hepatozoon organisms infecting the wildlife of two regions of the Brazilian Amazon. Int J Parasitol Parasites Wildl 6:115–121. https://doi.org/10.1016/j.ijppaw.2017.05.002
Stover BC, Muller KF (2010) TreeGraph 2: combining and visualizing evidence from different phylogenetic analyses. BMC Bioinformatics 11:01–09. https://doi.org/10.1186/1471-2105-11-7
Tardieu L, Rollock W, Garcia GW (2020) Wildlife rehabilitation: a case study of the neo-tropical, opossum Didelphis marsupialis insularis, Allen 1902. Braz J Biol 80:529–534. https://doi.org/10.1590/1519-6984.214757
Trifinopoulos J, Nguyen LT, Haeseler A, Minh BQ (2016) W-IQTREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235. https://doi.org/10.1093/nar/gkw256
Wolf RW, Aragona M, Muñoz-Leal S, Pinto LB, Melo ALT, Braga IA et al (2016) Novel Babesia and Hepatozoon agents infecting non-volant small mammals in the Brazilian Pantanal, with the first record of the tick Ornithodoros guaporensis in Brazil. Ticks Tick Borne Dis 7:449–456. https://doi.org/10.1016/j.ttbdis.2016.01.005
Funding
This work was supported by CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) with the Productivity Scholarship for MRA (CNPq Process nº 303701/2021–8), by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Código 001) and by Research Support Foundation of the State of São Paulo (Process No. 2020/12037–0 granted to the MRA).
Author information
Authors and Affiliations
Contributions
ÁFXO: writing of the original article and collecting samples. CBD: supervision and revision of the writing of the original article. ACC, MRA, and RZM: supervision and performance of molecular and phylogenetic analyses. NVS, ENM, ACC, and ABRG: support in carrying out hematological and morphometric analyses. AVS and DAB: supervision and performance of sample collections and animal clinical management. AG and JMR: support in DNA extractions, writing, and adequacy to the norms for article submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
Biological samples in this study were obtained following the directions of the Instituto Chico Mendes de Conservação da Biodiversidade (license number 76330–1) and the Ethics Committee of Universidade Estácio de Sá (number 009/ 2021).
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: Daniel Howe
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oliveira, Á.F.X., Calchi, A.C., Stocco, A.V. et al. Expanding the universe of Piroplasmids: morphological detection and phylogenetic positioning of putative novel piroplasmids in black-eared opossums (Didelphis aurita) from southeastern Brazil, with description of “South American Marsupialia Group” of Piroplasmida. Parasitol Res 122, 1519–1530 (2023). https://doi.org/10.1007/s00436-023-07852-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-07852-0