Abstract
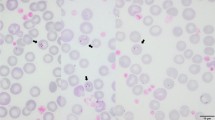
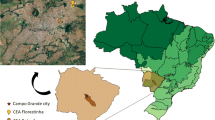
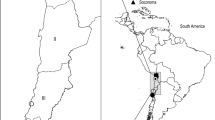
Xenarthra mammals can be found from southern North America to southern South America, including all Brazilian biomes. Although it has been shown that Xenarthra mammals can play a role as reservoirs for several zoonotic agents, few studies investigate the diversity of piroplasmids (Apicomplexa: Piroplasmida) in this group of mammals. Taking into account that piroplasmids can cause disease in animals and humans, understanding the prevalence and diversity of piroplasmids in Xenarthra mammals would contribute to conservation efforts for this group of animals as well as to infer risk areas for transmission of emergent zoonosis. The present study aimed to investigate the occurrence and molecular identity of piroplasmids in free-living mammals of the Superorder Xenarthra from four Brazilian states (Mato Grosso do Sul, São Paulo, Rondônia, and Pará). For this, DNA was extracted from blood or spleen samples from 455 animals. A nested PCR based on the 18S rRNA gene was used as screening for piroplasmids. Of the 455 samples analyzed, 25 (5.5%) were positive. Additionally, PCR assays based on 18S rRNA near-complete, cox-1, cox-3, hsp70, cytB, β-tubulin genes and the ITS-1 intergenic region were performed. Five out of 25 positive samples also tested positive for ITS-1-based PCR. The phylogenetic analysis positioned three 18S rRNA sequences detected in Priodontes maximus into the same clade of Babesia sp. detected in marsupials (Didelphis albiventris, Didelphis marsupialis, and Monodelphis domestica) and Amblyomma dubitatum collected from opossums and coatis in Brazil. On the other hand, the 18S rRNA sequence obtained from Dasypus novemcinctus was closely related to a Theileria sp. sequence previously detected in armadillos from Mato Grosso State, grouping in a subclade within the Theileria sensu stricto clade. In the phylogenetic analysis based on the ITS-1 region, the sequences obtained from Myrmecophaga tridactyla and Tamandua tetradactyla were placed into a single clade, apart from the other piroplasmid clades. The present study demonstrated the molecular occurrence of Piroplasmida in anteaters and Babesia sp. and Theileria sp. in armadillos from Brazil.



Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available in the NCBI GenBank Nucleotide platform (https://www.ncbi.nlm.nih.gov/genbank/) and can be accessed through accession numbers OQ969878-OQ969881 and OQ974563-OQ974565.
References
Acosta IC, Martins TF, Marcili A, Soares HS, Krawczak FS, Vieira FT, Labruna MB (2016) Ticks (Acari: Ixodidae, Argasidae) from humans, domestic and wild animals in the state of Espírito Santo, Brazil, with notes on rickettsial infection. Vet Parasitol Reg Stud Reports 3:66–69. https://doi.org/10.1016/j.vprsr.2016.08.001
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Alvarado-Rybak M, Solano-Gallego L, Millán J (2016) A review of piroplasmid infections in wild carnivores worldwide: importance for domestic animal health and wildlife conservation. Parasit Vectors 9:1–19. https://doi.org/10.1186/s13071-016-1808-7
André MR (2018) Diversity of Anaplasma and Ehrlichia/Neoehrlichia agents in terrestrial wild carnivores worldwide: implications for human and domestic animal health and wildlife conservation. Front Vet Sci 23(5):293. https://doi.org/10.3389/fvets.2018.00293
Arzua M, Onofrio VC, Barros-Battesti DM (2005) Catalogue of the tick collection (Acari Ixodida) of the Museu de História Natural Capão da Imbuia Curitiba Paraná Brazil. Rev Bras Zool 22(3):623–632. https://doi.org/10.1590/S0101-81752005000300015
ASASG (2023) The Xenarthrans. https://xenarthrans.org/species/. Accessed 11 May 2023
Barbosa AD, Austen J, Portas TJ, Amigo JÁ, Ahlstrom LA, Oskam CL et al (2019) Sequence analyses at mitochondrial and nuclear loci reveal a novel Theileria sp and aid in the phylogenetic resolution of piroplasms from Australian marsupials and ticks. PloS One 12:e0225822. https://doi.org/10.1371/journal.pone.0225822
Benson DA, Mizrachi IK, Lipman DJ, Ostell J, Rapp BA, Wheeler DI (2002) GenBank. Nucleic Acids Res 30:17–20. https://doi.org/10.1093/nar/28.1.15
Bernardes FCS, Martins TF, Ferreira SS, Rosa BF, Ruiz-Miranda CR, Giné GAF, Soffiati FL, Miranda FR (2022) Sloth’s giant tick (Amblyomma varium) parasitizing free-ranging maned sloth (Bradypus torquatus) in the Atlantic Forest biome. Brazil Braz J Vet Med 44:e004021. https://doi.org/10.29374/2527-2179.bjvm004021
Birkenheuer AJ, Neel J, Ruslander D, Levy MG, Breitschwerdt EB (2003) Detection and molecular characterization of a novel large Babesia species in a dog. Vet Parasitol 124:151–160. https://doi.org/10.1016/j.vetpar.2004.07.008
Calchi AC, Vultão JG, Alves MH, Yogui DR, Desbiez ALJ, De Santi M, Santana MS, da Silva TVM, Werther K, Teixeira MMG, Machado RZ, André MR (2020a) Ehrlichia spp. and Anaplasma spp. in Xenarthra mammals from Brazil, with evidence of novel ‘Candidatus Anaplasma spp.’. Sci Rep 10:12615. https://doi.org/10.1038/s41598-020-69263-w
Calchi AC, Vultão JG, Alves MH, Yogui DR, Desbiez ALJ, do Amaral RB, de Santi M, MMG T, Werther K, Machado RZ, André MR (2020) Multi-locus sequencing reveals a novel Bartonella in mammals from the Superorder Xenarthra. Transbound Emerg Dis 67:2020–2033. https://doi.org/10.1111/tbed.13545
Corduneanu A, Hrazdilová K, Sándor AD, Matei IA, Ionică AM, Barti L, Ciocanau MA, Mantoiu DS, Coroiu I, Hornok S, Fueher HP, Leitner N, Bagó Z, Stefke K, Modry D, Mihalca AD (2017) Babesia vesperuginis, a neglected piroplasmid: new host and geographical records, and phylogenetic relations. Parasit Vectors 10:598. https://doi.org/10.1186/s13071-017-2536-3
Dantas-Torres F, Ferreira DR, de Melo LM, Lima PAC, Siqueira DB, Rameh-de-Albuquerque LC, de Melo AV, Ramos JA (2010) Ticks on captive and free-living wild animals in northeastern Brazil. Exp Appl Acarol 50:181–189. https://doi.org/10.1007/s10493-009-9296-5
Dedet JP, Veilly M, Robin Y, Bonnevie O, Landau I (1988) Babesia choloepi n. sp. (Apicomplexa, Piroplasmida), parasite du paresseux à deux doigts, Choloepus didactylus (Linné, 1758)(Xenarthra, Bradypodidae), en Guyane française. Ann Parasitol Hum Comp 63:16–21. https://doi.org/10.1051/parasite/198863116
de Oliveira AFXD, Calchi AC, Mongruel ACB, Stocco AV, Stocco NV, Costa AC, Mureb EN, Pires JR, Guimarães A, Balthazar DA, Machado RZ, André MR, Baldani CD (2023) Expanding the universe of Piroplasmids: morphological detection and phylogenetic positioning of putative novel piroplasmids in black-eared opossums (Didelphis aurita) from southeastern Brazil, with description of “South American Marsupialis Group” of Piroplasmida. Parasitol Res. https://doi.org/10.1007/s00436-023-07852-0
de Oliveira LB, Calchi AC, Vultão JG, Yogui DR, Kluyber D, Alves MH, Desbiez ALJ, de Santi M, Soares AG, Soares JF, Werther K, Teixeira MMG, Machado RZ, André MR (2022) Molecular investigation of haemotropic mycoplasmas and Coxiella burnetii in free-living Xenarthra mammals from Brazil, with evidence of new haemoplasma species. Transbound Emerg Dis 69:e1877–e1891. https://doi.org/10.1111/tbed.14523
de Thoisy B, Michel JC, Vogel I, Vié JC (2000) A survey of hemoparasite infections in free-ranging mammals and reptiles in French Guiana. J Parasitol 86:1035–1040. https://doi.org/10.1645/0022-3395(2000)086[1035:asohii]2.0.co;2
Desbiez ALJ, Kluyber D (2013) The role of giant armadillos (Priodontes maximus) as physical ecosystem engineers. Biotropica 45:537–540. https://doi.org/10.1111/btp.12052
Ewing B, Green P (1998) Basecalling of automated sequencer traces using phred. II Error Probabilities. Genome Res 8:186–194. https://doi.org/10.1101/gr.8.3.186
Ewing B, Hillier L, Wendl MC, Green P (1998) Base-calling of automated sequencer traces using Phred. I Accuracy assessment. Genome Res 8:175–185. https://doi.org/10.1101/gr.8.3.175
Fava NM, Alves TS, Lopes MG, Labruna MB, Santos AQ, Cury MC (2022) Occurrence and molecular identification of hemoparasites in wild mammals kept in rehabilitation centers in Brazil. Acta Parasitol 67:1–11. https://doi.org/10.1007/s11686-021-00492-3
França RT, Da Silva AS, Loretti AP, Mazzanti CM, Lopes ST (2014) Canine rangeliosis due to Rangelia vitalii: from first report in Brazil in 1910 to current day–a review. Ticks Tick Borne Dis 5:466–474. https://doi.org/10.1016/j.ttbdis.2014.04.005
Furuta PI, Oliveira TMFS, Teixeira MCA, Rocha AG, Machado RZ, Tinucci-Costa M (2009) Comparison between a soluble antigen-based ELISA and IFAT in detecting antibodies against Babesia canis in dogs. Rev Bras Parasitol Vet 18:41–45. https://doi.org/10.4322/rbpv.01803007
Garcia MV, Silva DCD, Almeida RFCD, Cunha RC, Matias J, Barros JC, Andreotti R, Szabó MPJ (2013) Environmentally associated ticks (Acari: Ixodidae) in Campo Grande, Mato Grosso do Sul, Brazil. Rev Bras Parasitol Vet 22:124–128. https://doi.org/10.1590/s1984-29612013000100023
Gardner AL (2005) Order Pilosa. In: Wilson DE, Reeder DM (eds) Mammal species of the world. Johns Hopkins University Press, Baltimore, pp 94–99
Gonçalves LR, Paludo G, Bisol TB, Perles L, de Oliveira LB, de Oliveira CM, da Silva TMV, Nantes WAG, Duarte MA, Santos FM, Porfírio GEO, Hirano LQL, Herrera HM, Barros-Battesti DM, Machado RZ, André MR (2021) Molecular detection of piroplasmids in synanthropic rodents, marsupials, and associated ticks from Brazil, with phylogenetic inference of a putative novel Babesia sp. from white-eared opossum (Didelphis albiventris). Parasitol Res 120:3537–3546. https://doi.org/10.1007/s00436-021-07284-8
Gou H, Guan G, Liu A, Ma M, Xu Z, Liu Z, Ren Q, Li Y, Yang J, Chen Z, Yin H, Luo J (2012) A DNA barcode for Piroplasmea. Acta Trop 124:92–97. https://doi.org/10.1016/j.actatropica.2012.07.001
Guillemi EC, De La Fourniere S, Orozco M, Pena Martinez J, Correa E, Fernandez J, Arias LA, Paoletta M, Corona B, Pinarello V, Wilkowsky SE, Farber MD (2016) Molecular identification of Anaplasma marginale in two autochthonous South American wild species revealed an identical new genotype and its phylogenetic relationship with those of bovines. Parasit Vectors 9:1–9. https://doi.org/10.1186/s13071-016-1555-9
Greay TL, Zahedi A, Krige A, Owens JM, Rees RL, Ryan UM et al (2018) Endemic, exotic and novel apicomplexan parasites detected during a national study of ticks from companion animals in Australia. Parasite Vector 11:197. https://doi.org/10.1186/s13071-018-2775-y
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98 https://d1wqtxts1xzle7.cloudfront.net/29520866
Homer MJ, Aguilar-Delfin I, Telford SR III, Krause PJ, Persing DH (2000) Babesiosis. Clin Microbiol Rev 13:451–469. https://doi.org/10.1128/cmr.13.3.451
Hunfeld KP, Hildebrandt A, Gray JS (2008) Babesiosis: recent insights into an ancient disease. Int J Parasitol 38:1219–1237. https://doi.org/10.1016/j.ijpara.2008.03.001
Ikeda P, Menezes TR, Torres JM, de Oliveira CE, Lourenço EC, Herrera HM, Machado RZ, André MR (2021) First molecular detection of piroplasmids in non-hematophagous bats from Brazil, with evidence of putative novel species. Parasitol Res 120:301–310. https://doi.org/10.1007/s00436-020-06985-w
IUCN (2023) The IUCN red list of threatened species. https://www.iucnredlist.org/. Accessed 11 May 2023
Jalovecka M, Sojka D, Ascencio M, Schnittger L (2019) Babesia life cycle–when phylogeny meets biology. Trends Parasitol 35:356–368. https://doi.org/10.1016/j.pt.2019.01.007
Jefferies R, Ryan U, Jardine J, Broughton DK, Robertson ID, Irwin PJ (2007) Blood, bull terriers and babesiosis: further evidence for direct transmission of Babesia gibsoni in dogs. Aust Vet J 85:459–463. https://doi.org/10.1111/j.1751-0813.2007.00220.x
Katoh K, Rozewicki J, Yamada KD (2019) MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 20:1160–1111. https://doi.org/10.1093/bib/bbx108
Kawabuchi T, Tsuji M, Sado A, Matoba Y, Asakawa M, Ishihara C (2005) Babesia microti-like parasites detected in feral raccoons (Procyon lotor) captured in Hokkaido, Japan. J Vet Med Sci 67:825–827. https://doi.org/10.1292/jvms.67.825
Kjemtrup AM, Conrad PA (2000) Human babesiosis: an emerging tick-borne disease. Int J Parasitol 30:1323–1337. https://doi.org/10.1016/s0020-7519(00)00137-5
Kluyber D, Martins TF, Desbiez AJ, Amorim M, Massocato G, Labruna MB (2016) Ticks (Acari: Ixodidae) infesting armadillos (Cingulata: Dasypodidae) in the Pantanal wetland, Mato Grosso do Sul, Brazil. Syst Applied Acarol 21:1087–1091. https://doi.org/10.11158/saa.21.8.9
Labruna MB, de Paula CD, Lima TF, Sana DA (2002) Ticks (Acari: Ixodidae) on wild animals from the Porto-Primavera hydroelectric power station area, Brazil. Mem Inst Oswaldo Cruz 97:1133–1136. https://doi.org/10.1590/s0074-02762002000800012
Laison R, Shaw JJ, Fraiha H, Miles MA, Draper CC (1979) Chagas’s disease in the Amazon. Trypanosoma cruzi infections in sylvatic mammals, triatomine bugs and man in the State of Para, north Brazil. Trans R Soc Trop Med Hyg:193–204
Léger M, Mouzels P (1917) Piroplasma and microfilaria of an Edentate, Bradypus tridactylus, L. Bull Soc Pathol Exot 10:173–176
Leiby DA (2011) Transfusion-transmitted Babesia spp.: bull’s-eye on Babesia microti. Clin Microbiol Rev 24:14–28. https://doi.org/10.1128/cmr.00022-10
Luz HR, Costa Neto SFD, Weksler M, Gentile R, Faccini JLH (2018) Ticks parasitizing wild mammals in Atlantic Forest areas in the state of Rio de Janeiro, Brazil. Rev Bras Parasitol Vet 27:409–414. https://doi.org/10.1590/s1984-296120180027
Marques S, Barros-Battesti DM, Faccini JLH, Onofrio VC (2002) Brazilian distribution of Amblyomma varium Koch, 1844 (Acari: Ixodidae), a common parasite of sloths (Mammalia: Xenarthra). Mem Inst Oswaldo Cruz 97:1141–1146. https://doi.org/10.1590/s0074-02762002000800014
Martins TF, Yogui DR, Alves MH, Kluyber D, Massocato G, Labruna MB, Desbiez AL (2023) Surveillance of ticks (Acari: Ixodidae) on monitored free-ranging giant anteaters and on road-killed anteaters and armadillos in the State of Mato Grosso do Sul, Midwestern Brazil. Syst Appl Acarol 28:641–646. https://doi.org/10.11158/saa.28.4.1
Merino S, Martínez J, Martínez-de la Puente J, Criado-Fornelio Á, Tomás G, Morales J, Lobato E, García-Fraile S (2006) Molecular characterization of the 18S rDNA gene of an avian Hepatozoon reveals that it is closely related to Lankesterella. J Parasitol 92:1330–1335. https://doi.org/10.1645/ge-860r.1
Mongruel ACB, Medici EP, Canena AC, Calchi AC, Perles L, Rodrigues BCB, Soares JF, Machado RZ, André MR (2022) Theileria terrestris nov. sp.: a novel Theileria in lowland tapirs (Tapirus terrestris) from two different biomes in Brazil. Microorganisms 10:2319. https://doi.org/10.3390/microorganisms10122319
Ogrzewalska M, Uezu A, Jenkins CN, Labruna MB (2011) Effect of forest fragmentation on tick infestations of birds and tick infection rates by Rickettsia in the Atlantic Forest of Brazil. EcoHealth 8:320–331. https://doi.org/10.1007/s10393-011-0726-6
Perles L, Barreto WTG, de Macedo GC, Calchi AC, Bezerra-Santos M, Mendoza-Roldan JA, Otranto D, Herrera HM, Barros-Battesti DM, Machado RZ, André MR (2023) Molecular detection of Babesia spp. and Rickettsia spp. in coatis (Nasua nasua) and associated ticks from midwestern Brazil. Parasitol Res 122:1–8. https://doi.org/10.1007/s00436-023-07815-5
Quillfeldt P, Martinez J, Bugoni L, Mancini PL, Merino S (2014) Blood parasites in noddies and boobies from Brazilian offshore islands–differences between species and influence of nesting habitat. Parasitology 141:399–410. https://doi.org/10.1017/s0031182013001649
Ramos VN, Lemos FG, Azevedo FC, Arrais RC, Lima CFM, Candeias IZ, Martins MM, Sandrin ACLG, Siqueira SM, Szabó MPJ (2020) Wild carnivores, domestic dogs and ticks: shared parasitism in the Brazilian Cerrado. Parasitology 147:689–698. https://doi.org/10.1017/s0031182020000335
Sanches GS, André MR, Do Prado AP, Allegretti SM, Remedio RN, Nunes PH, Machado RZ, Bechara GH, Camargo-Mathias MI (2014) Oocyte maturation in the sloth’s giant tick Amblyomma varium (Acari: Ixodidae) in an ecological context. Exp Appl Acarol 64:519–531. https://doi.org/10.1007/s10493-014-9837-4
Schreeg ME, Marr HS, Tarigo JL, Cohn LA, Bird DM, Scholl EH, Levy MG, Wiegmann BM, Birkenheuer AJ (2016) Mitochondrial genome sequences and structures aid in the resolution of Piroplasmida phylogeny. PloS One 11:e0165702. https://doi.org/10.1371/journal.pone.0165702
Shaw SE, Birtles RJ, Day MJ (2001) Arthropod-transmitted infectious diseases of cats. J Feline Med Surg 3:193–209. https://doi.org/10.1053/jfms.2001.0149
Shock BC, Birkenheuer AJ, Patton LL, Olfenbuttel C, Beringer J, Grove DM, Peek M, Butfiloski JW, Hughes DW, Lockhart JM, Cunningham MW, Brown HM, Peterson DS, Yabsley MJ (2012) Variation in the ITS-1 and ITS-2 rRNA genomic regions of Cytauxzoon felis from bobcats and pumas in the eastern United States and comparison with sequences from domestic cats. Vet Parasitol 190:29–35. https://doi.org/10.1016/j.vetpar.2012.06.010
Silva VLDB, Almeida SLHD, Maia MO, Santos TÁ, Pavelegini LAD, Zaffalon GB, Marcili A, Morgado TO, Dutra V, Nakazato L, Pacheco RDC (2021) Post mortem protozoan hemoparasites detection in wild mammals from Mato Grosso state, Midwestern Brazil. Rev Bras Parasitol Vet 30:e013021. https://doi.org/10.1590/s1984-29612021083
Soares HS, Barbieri AR, Martins TF, Minervino AH, de Lima JT, Marcili A, Gennari SM, Labruna MB (2015) Ticks and rickettsial infection in the wildlife of two regions of the Brazilian Amazon. Exp ApplAcarol 65:125–140. https://doi.org/10.1007/s10493-014-9851-6
Soares HS, Marcili A, Barbieri AR, Minervino AH, Malheiros AF, Gennari SM, Labruna MB (2017) Novel Anaplasma and Ehrlichia organisms infecting the wildlife of two regions of the Brazilian Amazon. Acta Trop 174:82–87. https://doi.org/10.1016/j.actatropica.2017.07.006
Soares JF, Girotto A, Brandão PE, Da Silva AS, França RT, Lopes STA, Labruna MB (2011) Detection and molecular characterization of a canine piroplasm from Brazil. Vet Parasitol 180:203–208. https://doi.org/10.1016/j.vetpar.2011.03.024
Solano-Gallego L, Baneth G (2011) Babesiosis in dogs and cats—expanding parasitological and clinical spectra. Vet Parasitol 181:48–60. https://doi.org/10.1016/j.vetpar.2011.04.023
Stover BC, Muller KF (2010) TreeGraph 2: combining and visualizing evidence from different phylogenetic analyses. BMC Bioinformatics 11:01–09. https://doi.org/10.1186/1471-2105-11-7
Szabó MPJ, Pascoal JO, Martins MM, Ramos VN, Osava CF, Santos ALQ, Yokosawa J, Rezende LM, Tolesano-Pascoli GV, Torga K, de Castro MB, Suzin A, Barbieri ARM, Werther K, Silva JMM, Labruna MB (2019) Ticks and Rickettsia on anteaters from southeast and central-west Brazil. Ticks Tick Borne Dis 10:540–545. https://doi.org/10.1016/j.ttbdis.2019.01.008
Trifinopoulos J, Nguyen LT, Haeseler A, Minh BQ (2016) W-IQTREE: a fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235. https://doi.org/10.1093/nar/gkw256
Yabsley MJ, Vanstreels RE, Shock BC, Purdee M, Horne EC, Peirce MA, Parsons NJ (2017) Molecular characterization of Babesia peircei and Babesia ugwidiensis provides insight into the evolution and host specificity of avian piroplasmids. Int J Parasitol Parasites Wildl 6:257–264. https://doi.org/10.1016/j.ijppaw.2017.08.006
Zamoto A, Tsuji M, Wei Q, Cho SH, Shin EH, Kim TS, Leonova GN, Hagiwara K, Asakawa M, Kariwa H, Takashima I, Ishihara C (2004) Epizootiologic survey for Babesia microti among small wild mammals in northeastern Eurasia and a geographic deversity in the β-tubulin gene sequences. J Vet Med Sci 66:785–792. https://doi.org/10.1292/jvms.66.785
Zanella JRC (2016) Zoonoses emergentes e reemergentes e sua importância para saúde e produção animal. Pesqui Agropecu Bras 51:510–519. https://doi.org/10.1590/S0100-204X2016000500011
Zintl A, Mulcahy G, Skerrett HE, Taylor SM, Gray JS (2003) Babesia divergens, a bovine blood parasite of veterinary and zoonotic importance. Clin Microbiol Rev 16:622–636. https://doi.org/10.1128/cmr.16.4.622-636.2003
Acknowledgements
The authors would like to thank “Laboratório de Filogenia e Taxonomia Molecular de Tripanossomatídeos” Institute of Biology (IB) Institute of Biomedical Sciences (ICB), USP (São Paulo, SP, Brazil), ICAS (Instituto de Conservação de Animais Silvestres - Projeto Bandeiras e Rodovias e Projeto de Conservação do Tatu-Canastra), and Wildlife Pathology Service of the School of Agricultural and Veterinarian Sciences.
Funding
This work was supported by FAPESP (Fundação de Amparo à Pesquisa do Estado da São Paulo - Process #2020/12037-0) and CNPq (National Council for Scientific and Technological Development; Productivity Grant to MRA [CNPq Process #303701/2021-8]). A.C.C. and P.V.C.A. were supported by FAPESP (2020/07826-5; 2021/00353-7).
Author information
Authors and Affiliations
Contributions
Conceptualization: Ana Cláudia Calchi and Marcos Rogério André; Methodology: Ana Cláudia Calchi, Débora Regina Yogui, Mario Henrique Alves, Arnaud Leonard Jean Desbiez, Danilo Kluyber, Juliana Gaboardi Vultão, Paulo Vitor Cadina Arantes, Mariele de Santi, Karin Werther, and Marta Maria Geraldes Teixeira; Writing – original draft preparation: Ana Cláudia Calchi, Débora Regina Yougui, Mario Henrique Alves, Arnaud Leonard Jean Desbiez, Danilo Kluyber, Juliana Gaboardi Vultão, Mariele de Santi, Paulo Vitor Cadina Arantes, Karin Werther, Marta Maria Geraldes Teixeira, Rosangela Zacarias Machado, and Marcos Rogério André; Writing -review and editing: Ana Cláudia Calchi and Marcos Rogério André; Funding acquisition: Marcos Rogério André; Supervision: Marcos Rogério André.
Corresponding author
Ethics declarations
Ethical approval
Animal procedures and management protocols were approved by Institute Chico Mendes for Conservation of Biodiversity (SISBIO number 53798–5) and by the Ethics Committees on Animal use of the ICB—USP (protocol number 98) and the School of Agricultural and Veterinarian Sciences (FCAV/UNESP) (protocol number 9412/2022). The DNA samples were registered under SISGEN #A4C38D5 (“Sistema Nacional de Gestão do Patrimônio Genético e do Conhecimento Tradicional Associado”).
Consent to participate and consent for publication
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
Additional information
Handling Editor: Una Ryan
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Calchi, A.C., Yogui, D.R., Alves, M.H. et al. Molecular detection of piroplasmids in mammals from the Superorder Xenarthra in Brazil. Parasitol Res 122, 3169–3180 (2023). https://doi.org/10.1007/s00436-023-08008-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-023-08008-w