Abstract
Purpose
To evaluate the role of RUNX3 in breast cancer pathogenesis, we examined the RUNX3 expression in breast cancer tissues and analyzed the correlation between RUNX3 expression and clinicopathologic variables and patients survival.
Methods
We evaluated the RUNX3 expression by immunohistochemistry using a tissue microarray containing 256 specimens of breast cancer patients. We also studied the role of RUNX3 in cell migration and invasion by performing cell migration and invasion assay. Differential expression of metastasis-related genes after RUNX3 restoration was analyzed using the Human Tumor Metastasis PCR Array.
Results
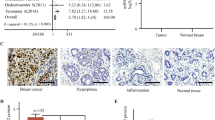
The RUNX3 expression was significantly correlated with breast cancer histology grade (P = 0.000), and low RUNX3 expression strongly correlated with worse 5-year overall and disease-specific survival rates (P = 0.000 and P = 0.001, respectively). Furthermore, we found that RUNX3 restoration suppressed breast cancer metastasis by controlling cell migration and invasion capacity. Finally, gene expression profiles of RUNX3-549 and Ctrl-549 cells showed matrix metalloproteinase-2 (MMP-2) was the most significant gene among the 84 metastasis-related genes influenced by RUNX3 reintroduction.
Conclusions
Reduced RUNX3 expression is significantly correlated with breast cancer progression and predicts worse survival. RUNX3 regulates breast cancer cell migration and invasion through the MMP-2 pathway.




Similar content being viewed by others
References
Al-Mehdi AB, Tozawa K, Fisher AB, Shientag L, Lee A, Muschel RJ (2000) Intravascular origin of metastasis from the proliferation of endothelium-attached tumor cells: a new model for metastasis. Nat Med 6(1):100–102. doi:10.1038/71429
Bai J, Zhang J, Wu J, Shen L, Zeng J, Ding J, Wu Y, Gong Z, Li A, Xu S, Zhou J, Li G (2010) JWA regulates melanoma metastasis by integrin alphaVbeta3 signaling. Oncogene 29(8):1227–1237. doi:10.1038/onc.2009.408
Bai J, Mei PJ, Liu H, Li C, Li W, Wu YP, Yu ZQ, Zheng JN (2012) BRG1 expression is increased in human glioma and controls glioma cell proliferation, migration and invasion in vitro. J Cancer Res Clin Oncol 138(6):991–998. doi:10.1007/s00432-012-1172-8
Bernardini C, Barba M, Tamburrini G, Massimi L, Di Rocco C, Michetti F, Lattanzi W (2012) Gene expression profiling in human craniosynostoses: a tool to investigate the molecular basis of suture ossification. Childs Nerv Syst 28(9):1295–1300. doi:10.1007/s00381-012-1780-2
Blaichman J, Marcus JC, Alsaadi T, El-Khoury M, Meterissian S, Mesurolle B (2012) Sonographic appearance of invasive ductal carcinoma of the breast according to histologic grade. AJR Am J Roentgenol 199(3):W402–W408. doi:10.2214/AJR.11.7374
Chen LF (2012) Tumor suppressor function of RUNX3 in breast cancer. J Cell Biochem 113(5):1470–1477. doi:10.1002/jcb.24074
Chen W, Salto-Tellez M, Palanisamy N, Ganesan K, Hou Q, Tan LK, Sii LH, Ito K, Tan B, Wu J, Tay A, Tan KC, Ang E, Tan BK, Tan PH, Ito Y, Tan P (2007) Targets of genome copy number reduction in primary breast cancers identified by integrative genomics. Genes Chromosomes Cancer 46(3):288–301. doi:10.1002/gcc.20411
Coradini D, Daidone MG (2004) Biomolecular prognostic factors in breast cancer. Curr Opin Obstet Gynecol 16(1):49–55
Deryugina EI, Quigley JP (2006) Matrix metalloproteinases and tumor metastasis. Cancer Metastasis Rev 25(1):9–34. doi:10.1007/s10555-006-7886-9
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19(5):403–410
Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thurlimann B, Senn HJ (2003) Meeting highlights: updated international expert consensus on the primary therapy of early breast cancer. J Clin Oncol 21(17):3357–3365. doi:10.1200/JCO.2003.04.576
Hanai J, Chen LF, Kanno T, Ohtani-Fujita N, Kim WY, Guo WH, Imamura T, Ishidou Y, Fukuchi M, Shi MJ, Stavnezer J, Kawabata M, Miyazono K, Ito Y (1999) Interaction and functional cooperation of PEBP2/CBF with Smads. Synergistic induction of the immunoglobulin germline Calpha promoter. J Biol Chem 274(44):31577–31582
Hayashi N, Nakamura S, Tokuda Y, Shimoda Y, Yagata H, Yoshida A, Ota H, Hortobagyi GN, Cristofanilli M, Ueno NT (2012) Prognostic value of HER2-positive circulating tumor cells in patients with metastatic breast cancer. Int J Clin Oncol 17(2):96–104. doi:10.1007/s10147-011-0260-0
Huang BQZ, Ong CW, Tsang YH, Xiao G, Shapiro D, Salto-Tellez M, Ito K, Ito Y, Chen LF (2011) RUNX3 acts as a tumor suppressor in breast cancer by targeting estrogen receptor alpha. Oncogene 31:527–534
Hwang KT, Han W, Bae JY, Hwang SE, Shin HJ, Lee JE, Kim SW, Min HJ, Noh DY (2007) Downregulation of the RUNX3 gene by promoter hypermethylation and hemizygous deletion in breast cancer. J Korean Med Sci 22(Suppl):S24–S31
Hynes NE, Lane HA (2005) ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5(5):341–354. doi:10.1038/nrc1609
Jemal A, Siegel R, Xu J (2010) Ward E (2010) Cancer statistics. CA Cancer J Clin 60(5):277–300. doi:10.3322/caac.20073
Jensen JD, Knoop A, Ewertz M, Laenkholm AV (2012) ER, HER2, and TOP2A expression in primary tumor, synchronous axillary nodes, and asynchronous metastases in breast cancer. Breast Cancer Res Treat 132(2):511–521. doi:10.1007/s10549-011-1610-3
Jiang Y, Tong D, Lou G, Zhang Y, Geng J (2008) Expression of RUNX3 gene, methylation status and clinicopathological significance in breast cancer and breast cancer cell lines. Pathobiology 75(4):244–251. doi:10.1159/000132385
Lau QC, Raja E, Salto-Tellez M, Liu Q, Ito K, Inoue M, Putti TC, Loh M, Ko TK, Huang C, Bhalla KN, Zhu T, Ito Y, Sukumar S (2006) RUNX3 is frequently inactivated by dual mechanisms of protein mislocalization and promoter hypermethylation in breast cancer. Cancer Res 66:6512–6520
Lee B, Thirunavukkarasu K, Zhou L, Pastore L, Baldini A, Hecht J, Geoffroy V, Ducy P, Karsenty G (1997) Missense mutations abolishing DNA binding of the osteoblast-specific transcription factor OSF2/CBFA1 in cleidocranial dysplasia. Nat Genet 16(3):307–310. doi:10.1038/ng0797-307
Li QL, Ito K, Sakakura C, Fukamachi H, Inoue K, Chi XZ, Lee KY, Nomura S, Lee CW, Han SB, Kim HM, Kim WJ, Yamamoto H, Yamashita N, Yano T, Ikeda T, Itohara S, Inazawa J, Abe T, Hagiwara A, Yamagishi H, Ooe A, Kaneda A, Sugimura T, Ushijima T, Bae SC, Ito Y (2002) Causal relationship between the loss of RUNX3 expression and gastric cancer. Cell 109(1):113–124
Look AT (1997) Oncogenic transcription factors in the human acute leukemias. Science 278(5340):1059–1064
Mei PJ, Bai J, Liu H, Li C, Wu YP, Yu ZQ, Zheng JN (2011) RUNX3 expression is lost in glioma and its restoration causes drastic suppression of tumor invasion and migration. J Cancer Res Clin Oncol 137(12):1823–1830. doi:10.1007/s00432-011-1063-4
Okuda T, van Deursen J, Hiebert SW, Grosveld G, Downing JR (1996) AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell 84(2):321–330
Pennison M, Pasche B (2007) Targeting transforming growth factor-beta signaling. Curr Opin Oncol 19(6):579–585. doi:10.1097/CCO.0b013e3282f0ad0e
Roberts AB, Anzano MA, Wakefield LM, Roche NS, Stern DF, Sporn MB (1985) Type beta transforming growth factor: a bifunctional regulator of cellular growth. Proc Natl Acad Sci USA 82(1):119–123
Rundhaug JE (2005) Matrix metalloproteinases and angiogenesis. J Cell Mol Med 9(2):267–285
Sakakura CMK, Fukuda KI, Nakashima S, Yoshikawa T, Kin S, Nakase Y, Ida H, Yazumi S, Yamagishi H, Okanoue T, Chiba T, Ito K, Hagiwara A, Ito Y (2007) Frequent silencing of RUNX3 in esophageal squamous cell carcinomas is associated with radioresistance and poor prognosis. Oncogene 26(40):5927–5938
Schwab M, Praml C, Amler LC (1996) Genomic instability in 1p and human malignancies. Genes Chromosomes Cancer 16(4):211–229. doi:10.1002/(SICI)1098-2264(199608)16:4<211:AID-GCC1>3.0.CO;2-0
Subramaniam MM, Chan JY, Soong R, Ito K, Ito Y, Yeoh KG, Salto-Tellez M, Putti TC (2009a) RUNX3 inactivation by frequent promoter hypermethylation and protein mislocalization constitute an early event in breast cancer progression. Breast Cancer Res Treat 113(1):113–121. doi:10.1007/s10549-008-9917-4
Subramaniam MM, Chan JY, Yeoh KG, Quek T, Ito K, Salto-Tellez M (2009b) Molecular pathology of RUNX3 in human carcinogenesis. Biochim Biophys Acta 1796(2):315–331. doi:10.1016/j.bbcan.2009.07.004
Wei D, Gong W, Oh SC, Li Q, Kim WD, Wang L, Le X, Yao J, Wu TT, Huang S, Xie K (2005) Loss of RUNX3 expression significantly affects the clinical outcome of gastric cancer patients and its restoration causes drastic suppression of tumor growth and metastasis. Cancer Res 65(11):4809–4816
Weigelt B, Peterse JL, van’t Veer LJ (2005) Breast cancer metastasis: markers and models. Nat Rev Cancer 5(8):591–602. doi:10.1038/nrc1670
Weith A, Brodeur GM, Bruns GA, Matise TC, Mischke D, Nizetic D, Seldin MF, van Roy N, Vance J (1996) Report of the second international workshop on human chromosome 1 mapping 1995. Cytogenet Cell Genet 72(2–3):114–144
Yu G, Wang J, Chen Y, Wang X, Pan J, Li G, Jia Z, Li Q, Yao JC, Xie K (2009) Overexpression of phosphorylated mammalian target of rapamycin predicts lymph node metastasis and prognosis of Chinese patients with gastric cancer. Clin Cancer Res 15(5):1821–1829. doi:10.1158/1078-0432.CCR-08-2138
Zhang Z, Chen G, Cheng Y, Martinka M, Li G (2011) Prognostic significance of RUNX3 expression in human melanoma. Cancer 117(12):2719–2727. doi:10.1002/cncr.25838
Acknowledgments
Grant support: This project is supported by grants from the National Natural Science Foundation of China (No. 81201636), the Science and Technology Department of Jiangsu province (No. BK2012139), and China Postdoctoral Science Foundation (No. 2012M511323).
Conflict of interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Jin Bai, Hong-Mei Yong and Fei-Fei Chen contributed equally to this paper.
Rights and permissions
About this article
Cite this article
Bai, J., Yong, HM., Chen, FF. et al. RUNX3 is a prognostic marker and potential therapeutic target in human breast cancer. J Cancer Res Clin Oncol 139, 1813–1823 (2013). https://doi.org/10.1007/s00432-013-1498-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-013-1498-x